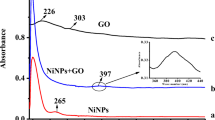
Abstract
Cu2O/nitrogen-doped grapheme(NG) nanocomposite material was prepared via a facile one step chemical reduction and characterized by means of X-ray diffraction(XRD) and scanning electron microscopy(SEM). A new electrochemical sensor was then fabricated by coating Cu2O/nitrogen-doped graphene nanocomposite with Nafion on glassy carbon electrode(Cu2O/NG/Nafion/GCE). The electrochemical response of this modified electrode toward ofloxacin was examined by cyclic voltammetry. The results indicate that Cu2O/NG/Nafion composite-modified electrode exhibits higher catalytic activity in the electrochemical oxidation of ofloxacin compared with glassy carbon electrode(GCE), Cu2O/Nafion modified electrode(Cu2O/Nafion/GCE), and N-doped graphene/Nafion modified electrode( NG/Nafion/GCE). Under optimal conditions, the peak current was found to be linearly proportional to the concentration of ofloxacin in the 0.5―27.5 μmol/L and 27.5―280 μmol/L ranges with a lower detection limit of 0.34 μmol/L, higher sensitivity of 39.32 μA·L·mmol–1 and a shorter reaction time of less than 2 s. In addition, Nafion can enhance the stability of the modified electrode and prevent some negative species. Thus the modified electrode exhibits good selectivity and a long working life. The Cu2O/NG/Nafion composite modified electrode shows promising application in electrochemical sensors, biosensors, and other related fields because of its excellent properties.
Similar content being viewed by others
References
Fierens C., Hillaert S., Bossche W. V., J. Pharm. Biomed. Anal., 2000, 22(5), 763
Cheng G. W., Wu H. L., Huang Y. L., Anal. Chim. Acta, 2008, 616(2), 230
Shao B., Sun X. J., Zhang J., Hu J. Y., Dong H. R., Yang Y., J. Chromatogr. A, 2008, 1182, 77
Paul S. F., Jacqui L. A., Anal. Chim. Acta, 2005, 541, 3
Garciía M. S., Albero M. I., Sánchez-Pedreño C., Abuherba M. S.. Eur. J. Pharm. Biopharm., 2005, 61, 87
Huang K. J., Liu X., Xie W. Z., Yuan H. X., Colloids. Surf. B, 2008, 64(2), 269
Zhang F. F., Gu S. Q., Ding Y. P., Li L., Liu X., Bioelectrochem., 2013, 89, 42
Chen D., Tang L. H., Li J. H., Chem. Soc. Rev., 2010, 39, 3157
Chen D., Feng H. B., Li J. H., Chem. Rev., 2012, 112, 6027
Tang L. H., Wang Y., Li Y. M., Feng H. B., Lu J., Li J. H., Adv. Funct. Mater., 2009, 19, 2782
Wang Y., Li Y. M., Tang L. H., Lu J., Li J. H., Electrochem. Commun., 2009, 11, 889
Yang G. H., Li Y. J., Rana R. K., Zhu J. J., J. Mater. Chem. A, 2013, 1(5), 1754
Briskman R. N., Sol. Energy Mater. Sol. Cells, 1992, 27(4), 361
Wang M. Y., Huang J. R., Tong Z. W., Li W. H., Chen J. B., J. Alloys Compd., 2013, 568, 26
Ye X. L., Gu Y., Wang C. M., Sens. Actuators B, 2012, 173, 530
Yan X. Y., Tong X. L., Zhang Y. F., Han X. D., Wang Y. Y., Jin G. Q., Qin Y., Guo X. Y., Chem. Commun., 2012, 48(13), 1892
Liu M. M., Liu R., Chen W., Biosens. Bioelectron., 2013, 45, 206
Li J., Guo S. J., Zhai Y. M., Wang E. K., Anal. Chim. Acta, 2009, 649, 196
Li X. L., Wang H. L., Robinson J. T., Sanchez H., Diankov G., Dai H. J., J. Am. Chem. Soc., 2009, 131, 15939
Zen J. M., Chung H. H., Kumar A. S., Analyst, 2000, 125, 1633
Huang K. J., Liu X., Xie W. Z., Yuan H. X., Microchim. Acta, 2008, 162(1/2), 227
Laviron E., J. Electroanal. Chem., 1979, 101, 19
Ding S. N., Xu J. J., Zhang W. J., Chen H. Y., Talanta, 2006, 70, 572
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Natural Science Foundation of China(Nos.21071005, 21271006), the Natural Science Foundation of the Education Bureau of Anhui Province, China(No.KJ2015A024) and the Graduate Student Innovation Fund of Anhui University of Technology, China(No.2014033).
Rights and permissions
About this article
Cite this article
Wu, F., Xu, F., Chen, L. et al. Cuprous oxide/nitrogen-doped graphene nanocomposites as electrochemical sensors for ofloxacin determination. Chem. Res. Chin. Univ. 32, 468–473 (2016). https://doi.org/10.1007/s40242-016-5367-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-016-5367-4