Abstract
Background
PPAR-γ has an integrative role in the management of insulin resistance; ligands of this receptor have emerged as potent insulin sensitizers and may modulate proteins involved in the pathogenesis of diabetes mellitus. Hence the present study is aimed to identify PPAR-γ modulators from the plant Cassia glauca and predict the ontology enrichment analysis utilizing various in-silico tools.
Methods
ChEBI database was used to mine the phytoconstituents present in the plant C. glauca, SwissTargetPrediction database was used to identify the targets, and scrutinizing of phytoconstituents modulating PPAR-γ was performed. Autodock4.0 was used to dock phytoconstituent ligands with the target PPAR-γ. Multiple open-source databases and in-silico tools were utilized to predict the drug-likeness characters and predict side effects of the phytoconstituents modulating PPAR-γ and STRING database was used to construct a network between the modulated genes.
Results
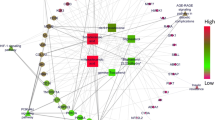
Twenty-four phytoconstituents were identified from the plant Cassia glauca from which four were found to modulate PPAR-γ, sennoside was predicted to have the greatest drug-likeness score and a significantly less side effect whereas diphenyl sulfone was predicted to show hepatotoxicity with the greatest pharmacological activity of 0.815. [epicatechin-(4beta- > 8)]5-epicatechin showed the lowest binding affinity with target PPAR-γ i.e. -8.6 kcal/mol and possessing a positive drug-likeness score with no side effect data.
Conclusion
Bioctives were found free from probable side effects leaving out diphenyl sulfone having a prediction of hepatotoxicity, the anti-diabetic property of the plant may be due to the presence of [epicatechin-(4beta- > 8)]5-epicatechin which needs further validation by in-vitro and in-vivo protocols.



Similar content being viewed by others
References
Mellitus D. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2005;28(S37):S5–10.
Forouhi NG, Wareham NJ. Epidemiology of diabetes. Medicine. 2010;38(11):602–6.
Deshpande AD, Harris-Hayes M, Schootman M. Epidemiology of diabetes and diabetes-related complications. Phys Ther. 2008;88(11):1254–64.
Wagenmakers AJ, Frayn KN, Arner P, Yki-Järvinen H. Fatty acid metabolism in adipose tissue, muscle and liver in health and disease. Essays Biochem. 2006;27(42):89–103.
Kadhe G, Arasan RE. Advances in drug delivery of oral hypoglycemic agents. Curr Sci. 2002;83(12):1539–43.
Hays NP, Galassetti PR, Coker RH. Prevention and treatment of type 2 diabetes: current role of lifestyle, natural product, and pharmacological interventions. Pharmacol Ther. 2008;118(2):181–91.
Chen L, Magliano DJ, Zimmet PZ. The worldwide epidemiology of type 2 diabetes mellitus—present and future perspectives. Nat Rev Endocrinol. 2012;8(4):228–36.
Rangwala SM, Lazar MA. Peroxisome proliferator-activated receptor γ in diabetes and metabolism. Trends Pharmacol Sci. 2004;25(6):331–6.
Cho N, Momose Y. Peroxisome proliferator-activated receptor γ agonists as insulin sensitizers: from the discovery to recent progress. Curr Top Med Chem. 2008;8(17):1483–507.
Janani C, Kumari BR. PPAR gamma gene–a review. Diabetes Metab Syndr. 2015;9(1):46–50.
Martens FM, Visseren FL, Lemay J, de Koning EJ, Rabelink TJ. Metabolic and additional vascular effects of thiazolidinediones. Drugs. 2002;62(10):1463–80.
Chinetti G, Fruchart JC, Staels B. Peroxisome proliferator-activated receptors (PPARs): nuclear receptors at the crossroads between lipid metabolism and inflammation. J Inflamm Res. 2000;49(10):497–505.
Bassaganya-Riera J, Misyak S, Guri AJ, Hontecillas R. PPAR γ is highly expressed in F4/80hi adipose tissue macrophages and dampens adipose-tissue inflammation. Cell Immunol. 2009;258(2):138–46.
Cha BS, Ciaraldi TP, Carter L, Nikoulina SE, Mudaliar S, Mukherjee R, Paterniti JR Jr, Henry RR. Peroxisome proliferator-activated receptor (PPAR) γ and retinoid X receptor (RXR) agonists have complementary effects on glucose and lipid metabolism in human skeletal muscle. Diabetologia. 2001;44(4):444–52.
Wanjari MM, Mishra S, Dey YN, Sharma D, Gaidhani SN, Jadhav AD. Antidiabetic activity of Chandraprabha vati - a classical Ayurvedic formulation. J Ayurveda Integr Med. 2016;7(3):144–50. https://doi.org/10.1016/j.jaim.2016.08.010.
Duyu T, Khanal P, Dey YN, Jha SK. Network pharmacology of Withania somnifera against stress associated neurodegenerative diseases. Adv Tradit Med (ADTM). 2021. https://doi.org/10.1007/s13596-020-00530-x.
Ternikar SG, Patil MB, Pasha I, Khanal P. Gene set enrichment analysis of α-amylase and α-glucosidase inhibitors of Cassia glauca. J Diabetes Metab Disord. 2020;19(2):683–9.
Khanal P, Patil BM. Consolidation of network and experimental pharmacology to divulge the antidiabetic action of Ficus benghalensis L. bark. 3 Biotech. 2021;11:238.
Khanal P, Patil BM. Integration of in silico, in vitro and ex vivo pharmacology to decode the anti-diabetic action of Ficus benghalensis L. bark. J Diabetes Metab Disord. 2020;19(2):1325–37.
Veerapur VP, Pratap V, Thippeswamy BS, Marietta P, Bansal P, Kulkarni PV, Kulkarni VH. Polyphenolic enriched extract of Cassia glauca Lamk, improves streptozotocin-induced type-1 diabetes linked with partial insulin resistance in rats. J Ethnopharmacol. 2017;198:489–98.
Srinivas Y. Phytochemical and in vitro anticancer activity of Cassia glauca leaves extract. Int J Green Pharm. 2019;13(04):354–8.
Kim S, Thiessen PA, Bolton EE, Chen J, Fu G, Gindulyte A, Han L, He J, He S, Shoemaker BA, Wang J. PubChem substance and compound databases. Nucleic Acids Res. 2016;44(D1):D1202–13.
Biovia DS. Discovery studio visualizer. San Diego, CA, USA. 2017;936.
Drug-Likeness and molecular property prediction. molsoft molecules in silico. Available at: https://molsoft.com/mprop/.
Ivanov SM, Lagunin AA, Rudik AV, Filimonov DA, Poroikov VV. ADVERPred–Web service for prediction of adverse effects of drugs. J Chem Inf Model. 2018;58(1):8–11.
Halgren TA. Merck molecular force field. I. Basis, form, scope, parameterization, and performance of MMFF94. J Comput Chem. 1996;17(5–6):490–519.
Khanal P, Dey YN, Patil R, Chikhale R, Wanjari MM, Gurav SS, Patil BM, Srivastava B, Gaidhani SN. Combination of system biology to probe the anti-viral activity of andrographolide and its derivative against COVID-19. RSC Advances. 2021;11(9):5065–79.
Duyu T, Khanal P, Khatib NA, Patil BM. Mimosa pudica Modulates Neuroactive Ligand-Receptor Interaction in Parkinson’s Disease. Indian J Pharm Educ Res. 2020;54(3):732–9.
Khanal P, Patil BM. In vitro and in silico anti-oxidant, cytotoxicity and biological activities of Ficus benghalensis and Duranta repens. Chin Herb Med. 2020;12(4):406–13.
Duyu T, Khatib NA, Khanal P, Patil BM, Hullatti KK. Network pharmacology-based prediction and experimental validation of Mimosa pudica for Alzheimer’s disease. J Phytopharmacol. 2020;9(1):46–53.
Khanal P, Patil BM. Integration of network and experimental pharmacology to decipher the antidiabetic action of Duranta repens L. J Integr Med. 2021;19(1):66–77.
Zeven AC, De Wet JM. Dictionary of cultivated plants and their regions of diversity: excluding most ornamentals, forest trees and lower plants. Pudoc; 1982.
Khan ZI, Nahar B, Jakaria A, Rahman S, Chowdhury MH, Rahmatullah M. An evaluation of antihyperglycemic and antinociceptive effects of methanol extract of Cassia fistula L. (Fabaceae) leaves in Swiss albino mice. Adv Nat Appl Sci. 2010;4(3):305–11.
Pletz J, Enoch SJ, Jais DM, Mellor CL, Pawar G, Firman JW, Madden JC, Webb SD, Tagliati CA, Cronin MT. A critical review of adverse effects to the kidney: mechanisms, data sources, and in silico tools to assist prediction. Expert Opin Drug Metab Toxicol. 2018;14(12):1225–53.
Cordero-Herrera I, Chen X, Ramos S, Devaraj S. (−)-Epicatechin attenuates high-glucose-induced inflammation by epigenetic modulation in human monocytes. Eur J Nutr. 2017;56(3):1369–73.
Ursu O, Rayan A, Goldblum A, Oprea TI. Understanding drug-likeness. Wiley Interdiscip Rev Comput Mol Sci. 2011;1(5):760–81.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical statement
This work did not include any human subjects and animals to retrieve the data.
Conflict of interest
None declared.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ternikar, S.G., Patil, M.B., Pasha, I. et al. Gene ontology enrichment analysis of PPAR-γ modulators from Cassia glauca in diabetes mellitus. J Diabetes Metab Disord 20, 1239–1246 (2021). https://doi.org/10.1007/s40200-021-00848-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-021-00848-6