Abstract
Purpose
Intravenous lipid emulsion (IVLE) was first used to prevent essential fatty acids deficiency. IVLE with α-tocopherol was reported to provide protection against parenteral nutrition-associated liver disease. This study aims to determine the optimal parameters and conditions in developing a physically stable IVLE from superolein palm oil (SoLE 20%) and its effect on lipid and liver profiles in an animal model.
Methods
SoLE 20% was prepared using superolein oil and MCT oil (1:1), stabilized with egg lecithin and homogenized using a high pressure homogenizer. Mean droplet size was used as the response variable and was measured using laser diffraction and dynamic light scattering method. Physical stability at 4 °C, 25 °C and 40 °C storage temperatures were determined based on particle size and distribution, polydispersity index, zeta potential, viscosity, vitamin E contents and pH. Sterility and pyrogenicity were also investigated. Rabbits were administered with 1.0 g/kg SoLE 20% for 5 h and repeated daily for 3 days to investigate its effect on blood lipid and liver enzymes profile.
Results
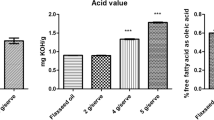
SoLE 20% was succesfully prepared using the optimized parameters of 800 psi, 7 cycles and 1.2 g lecithin. The IVLE prepared had a particle size of 252.60 ± 4.88 nm and was physically stable for 4 weeks at different storage temperatures. SoLE 20% had a high content of natural vitamin E, remained sterile and pyrogen free. It was also safe for intravenous administration and did not alter the blood lipid (p > 0.05) and liver enzymes profiles (p > 0.05) of the rabbits.
Conclusion
The optimal parameters to develop a stable superolein based IVLE are 800 psi homogenization pressure, 7 homogenization cycles and using 1.2 g lecithin as the emulsifier. SoLE 20% is safe for intravenous administration and does not significantly alter lipid and liver enzymes profiles of the rabbits.


Similar content being viewed by others
References
Vanek VW, Seidner DL, Bistrian B, Gura K, Valentine CJ. Novel nutrient task force, intravenous fat emulsions workgroup, American Society for Parenteral and Enteral Nutrition (ASPEN) Board of Directors. ASPEN position paper: clinical role for alternative intravenous fat emulsions. Nutr Clin Pract. 2012;27(2):150–92.
Driscoll DF. Commercial lipid emulsions and all-in-one mixtures for intravenous infusion – composition and physicochemical properties. In: Calder PC, Waitzberg DL, Koletzko B, editors. Intravenous lipid emulsions vol. 112. World Rev Nutr Diet. Basel: Karger; 2015. p. 48–56. https://doi.org/10.1159/000365430.
Akbari H, Dalir-Naghadeh B. The effects of short term intravenous infusion of a soybean based lipid emulsion on some blood constituents in sheep: a preliminary study. Vet Res Forum. 2014;5(1):35–41.
Fettiplace MR, Weinberg G. Past, present and future of lipid resuscitation therapy. JPEN J Parenter Enteral Nutr. 2015;39(1):72S–83S.
Muller RH, Harden D, Keck CM. Development of industrially feasible concentrated 30% and 40% nanoemulsions for intravenous drug delivery. Drug Dev Ind Pharm. 2012;38(4):420–30.
Ren T, Cong L, Wang Y, Tang Y, Tian B, Lin X, et al. Lipid emulsions in parenteral nutrition: current applications and future developments. Expert Opin Drug Deliv. 2013;10(11):1533–49.
Mba OI, Dumont MJ, Ngadi M. Palm oil: processing, characterization and utilization in the food industry-a review. Food Biosci. 2015;10:26–41.
Narang D, Subeena Sood S, Thomas MK, Dinda AK, Maulik SK. Effect of dietary palm olein oil on oxidative stress associated with ischemic-reperfusion injury in isolated rat heart. BMC Pharmacol. 2004;4:29. https://doi.org/10.1186/1471-2210-4-29.
Ng KDO, Stoll B, Chacko S, de Pipaon MS, Lauridsen C, Gray M, et al. Vitamin E in new-generation lipid emulsions protects against parenteral nutrition-associated liver disease in parenteral nutrition-fed preterm pigs. JPEN J Parenter Enteral Nutr. 2016;40(5):656–71.
Alayoubi A, Abu-Fayyad A, Rawas-Qalaji MM, Slyvester PW, Nazzal S. Effect of lipid viscosity and lipid viscosity of “vitamin E” enriched emulsion. Pharm Dev Technol. 2015;20(5):555–61.
Xu Z, Harvey KA, Pavlina TM, Zaloga GP, Siddiqui RA. Tocopherol and tocotrienol homologs in parenteral lipid emulsions. Eur J Lipid Sci Technol. 2015;117:15–22.
Jufri M, Azmi N, Shamsuddin AF. Effects of intravenous palm oil-based lipid nanoemulsion on fat metabolism in rabbits. Asian Pac J Trop Dis. 2012;2:S944–8.
Zainol S, Basri M, Basri H, Shamsuddin AF, Abdul-Ghani SS, Abedi-Karjiban R, et al. Formulation optimization of a palm-based Nanoemulsion system containing levodopa. Int J Mol Sci. 2012;13:13049–64.
Tan SK, Masoumi HRF, Karjiban RA, Stanslas J, Kirby BP, Basri M, et al. Ultrasonic emulsification of parenteral valproic acid-loaded nanoemulsion with response surface methodology and evaluation of its stability. Ultrason Sonochem. 2016;29:299–308.
Said KAM, Amin MAM. Overview on the response surface methodology (RSM) in extraction process. Journal of Applied Science & Process Engineering. 2015;2(1):8–17. http://www.jaspe.unimas.my/images/jaspe/Vol2_No1_2015/V21_Paper2.pdf. Accessed 26 Feb 2019.
Bouchoud L, Sadeghipour F, Klingmuller M, Fonzo-Christe C, Bonnabry P. Long-term physico-chemical stability of standard parenteral nutritions for neonates. Clin Nutr. 2010;29:808–12.
Tadros TF. Emulsion formation, stability and rheology. In: Tadros TF, editors. Emulsion formation and stability. First edition. Weinheim: Wiley-VCH Verlag GmbH & Co. KGaA; 2013. p. 1–75. https://doi.org/10.1002/9783527647941.
WHO. Stability testing of active pharmaceutical ingredients and finished pharmaceutical products. In: WHO technical report series; 2009. http://apps.who.int/medicinedocs/documents/s19133en/s19133en.pdf. Accessed 6 Feb 2019.
Garcia-Moreno PJ, Guadix A, Guadix EM, Jacobsen C. Physical and oxidative stability of fish oil-in-water emulsions stabilized with fish protein hydrolysates. Food Chem. 2016;203:124–35.
Staven V, Wang S, Gronlie I, Tho I. Development and evalution of a test program for Y-site compatibility testing of total perenteral nutrition and intravenous drugs. Nutr J. 2016;15. https://doi.org/10.1186/s12937-016-0149-x.
Anez-Bustillos L, Dao DT, Baker MA, Fell GL, Puder M, Gura KM. Intravenous fat emulsion formulation for the adult and pediatric patient: understanding the differences. Nutr Clin Pract. 2016;31(5):596–609.
Peng J, Dong WJ, Li L, Xu JM, Jin DJ, Xia XJ, et al. Effect of high-pressure homogenization preparation on mean globule size and large-diameter tail of oil-in-water injectable emulsions. J Food Drug Anal. 2015;23:828–35.
Gong H, Geng S, Zheng Q, Wang P, Luo L, Wang X, et al. An intravenous claritnromycin lipid emulsion with a high drug loading, H-bonding and a hydrogen-bonded ion pair complex exhibiting excellent antibacterial activity. Asian J Pharm Sci. 2016;11(5):618–30.
Salama GSA, Kaabneh MAF, Almasaeed MN, Alquran MIA. Intravenous lipids for preterm infants: a review. Pediatrics. 2015;9:25–36.
Memisoglu-Bilensoy E, Hincal AA. Sterile, injectable cyclodextrin nanoparticles: effects of gamma irradiation and autoclaving. Int J Pharm. 2006;311(1–2):203–8.
Driscoll DF. Globule-size distribution in injectable 20% lipid emulsions: compliance with USP requirements. Am J Health-System Pharm. 2007;64:2032–6.
Driscoll DF, Ling PR, Bistrian BR. Pharmacopeial compliance of fish oil-containing parenteral lipid emulsionmixtures: globule size distribution (GSD) and fatty acid analyses. Int J Pharm. 2009;379:125–30.
WHO. Test for sterility. International pharmacopoeia. 4th ed; 2012. https://www.who.int/medicines/publications/pharmacopoeia/TestForSterility-RevGenMethod_QAS11-413FINALMarch2012.pdf. Accessed 6 Feb 2019
Akers MJ. Introduction, scope and history of sterile products. In: Sterile drug products: formulation, packaging, manufacturing and quality. New York: Informa Healthcare; 2010. p. 1–10.
Testing AKS. Pharmaceutical product. In: Pharmaceutical microbiology. New Delhi: New Age International (P) Limited; 2008. p. 231–45.
John BA, Jalal KCA, Kamaruzzaman YB, Zaleha K. Mechanism in the clot formation of horseshoe crab blood during bacterial endotoxin invasion. J Appl Sci. 2010;10(17):1930–6.
Sandle T. Depyrogenation and endotoxin. In: Sterility, sterilisation and sterility assurance for pharmaceutical. Oxford: Woodhead Publishing; 2013. p. 171–88.
Hormann K, Zimmer A. Drug delivery and drug targeting with parenteral lipid nanoemulsions - a review. J Control Release. 2016;223:85–98.
Kaur K, Kumar R, Arpita GS, Uppal S, Bhatia A, Mehta SK. Physicochemical and cytotoxicity study of TPGS stabilized nanoemulsion designed by ultrasonication method. Ultrason Sonochem. 2017;34:173–82.
Cheong AM, Nyam KL. Improvement of physical stability of kenaf seed oil-in-water nanoemulsions by addition of β-cyclodextrin to primary emulsion containing sodium caseinate and Tween 20. J Food Eng. 2016;183:24–31.
Roche LD, Medina EA, Perez AF, Viart MAB, Lopez YS, Cama VF, et al. Lipofundin induced hyperlipidemia promotes oxidative stress and atherosclerotic lesions in New Zealand white rabbits. Int J Vasc Med. 2012a;2012:1–7. https://doi.org/10.1155/2012/898769.
Roche LD, Perez AF, Viart MAB, Lopez AMV. Lipofundin 20% induces hepatic lipid peroxidation in New Zealand white rabbits. Rev MVZ Cordoba. 2012b;17(3):3113–7.
Shahid U, Ahmed Khan D, Noor U, Ahmed Butt S. Effect of tocotrienols on lipid profile in rabbits. JRMC. 2012;16(1):68–9. https://www.journalrmc.com/index.php/JRMC/article/view/627/479. Accessed 26 February 2019.
Mayada RF, Taghred MS, Haytham AA. Boldenone-induced apoptotic, structural and functional alterations in the liver of rabbits. World Rabbit Sci. 2015;23:39–46.
Meredith A, Rayment L. Liver disease in rabbits. Seminars in Avian and Exotic Pet Medicine. 2000;9(3):146–52.
Onyesom I, Anosike EO. Changes in rabbit liver function markers after chronic exposure to ethanol. J Biochem Mol Biol Biophys. 2007;2(5):337–42. https://doi.org/10.3923/ajb.2007.337.342.
Ozkan C, Kaya A, Akgul Y. Normal values of haematological and some biochemical parameters in serum and urine of New Zealand white rabbits. World Rabbit Sci. 2012;20:253–9.
Shousha SM, Mahmoud MA, Hameed K. Some haemato-biochemical value in white New Zealand rabbits. IOSR J Agric Vet Sci. 2017;10(7):40–4.
Jufri M, Azmi A, Shamsuddin AF. Effect of intravenous palm oil-based lipid nanoemulsion on fat metabolism in rabbits. Asian Pac J Trop Dis. 2012;2:S944–8.
Mirtallo JM, Dasta JF, Klienschmidt KC, Varon J. State of the art review: intravenous fat emulsion: current applications, safety profile and clinical Implicatios. Ann Pharmacother. 2010;44:688–700.
Acknowledgements
This work has been supported by Universiti Kebangsaan Malaysia and the superolein oil has been supplied by FELDA IFFCO Sdn. Bhd. (Klang, Malaysia).
Funding
This study was funded by Universiti Kebangsaan Malaysia (Grant No. ETP-2013-073).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Animal study was approved by Universiti Kebangsaan Malaysia Animal Ethic Committee with approval number of FF/2017/AHMAD FUAD/24-MAY/847-JULY-2017-FEB.-2018.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Amran, M.H.H., Zulfakar, M.H., Danik, M.F. et al. A new alternative for intravenous lipid emulsion 20% w/w from superolein oil and its effect on lipid and liver profiles in an animal model. DARU J Pharm Sci 27, 191–201 (2019). https://doi.org/10.1007/s40199-019-00262-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-019-00262-7