Abstract
Purpose of Review
This review summarizes the anesthesiologist’s role in transferring critically ill surgical patients at different phases of care.
Recent Findings
Early recognition of patients at high intraoperative and postoperative risk is one of the most important first steps, followed by preoperative and intraoperative stabilization measures depending on the individual needs. It mainly is the anesthesiologist’s responsibility to decide on postoperative ICU admission. The transfer of the critically ill should be planned; the ICU staff has to be informed as early as possible. Locally developed checklists should be used during the preparation of patient transport. Trained, dedicated staff should be made available in every institution. A detailed handover using dedicated institutional flowcharts should ensure patient safety upon arrival to the ICU.
Summary
Transfer of critically ill patients from the OR to the ICU is an interdisciplinary task with a high probability of eventual incidents. Anesthesiologists should play a key role in all phases of the procedure to improve patient outcomes.
Similar content being viewed by others
Introduction
Perioperative complications in high-risk surgical patients remain the leading causes of death and prolonged hospitalization. Although high-risk patients make up approximately 9–15% of surgical procedures they might be responsible for as high as 80% of deaths [1, 2]. The availability of hospital critical care beds shows a large variation worldwide [3], and it is clear that hospital mortality for patients admitted to critical care following surgery is significantly influenced by critical care bed utilization [4•]. On the other hand, several important factors may significantly influence patients’ outcome, especially in the first 24 h postoperatively. These factors are related to the anesthesia team and include preoperative risk stratification, preparation of high-risk patients for elective surgeries, and intraoperative and postoperative measures. In the present review, we intend to summarize the key issues that may be essential from the anesthesiological point of view to ensure a better outcome for high-risk patients and prepare them for intensive care unit (ICU) transfer.
The most important tasks of the anesthesiologist can be summarized as follows:
-
1.
Preoperative identification and optimization of high-risk patients
-
2.
Intraoperative monitoring and therapeutic measures
-
3.
Prevention, recognition, and treatment of intraoperative causes leading to unplanned ICU admission
-
4.
Organizing transport and handover
Preoperative Identification of the “High-Risk” Patient
Different definitions exist for describing high-risk surgical patients. Age, underlying diseases, the complexity of the surgical procedure, and elective or urgent interventions have to be taken into account. In a most generally accepted term, a high-risk surgical patient is defined as a patient having an individual mortality risk of > 5%, the surgical procedure itself poses a mortality risk of > 5%, or both.
For anesthesia providers, the most simple and most widely used risk stratification scoring system is American Society of Anesthesiologists Physical Status (ASA-PS) scoring. According to the report of Wolters et al., the risk of postoperative mortality exceeds 5% in ASA IV grade; thus, per definition, they should be considered high-risk patients, especially if they undergo emergency procedures [5].
Boyd and associates [6••] described a more specific list of criteria for high-risk surgical patients:
-
Previous severe cardiorespiratory illness—acute myocardial infarction, chronic obstructive pulmonary disease, or stroke
-
Late-stage vascular disease involving the aorta
-
Age > 70 years with limited physiological reserve in one or more vital organs
-
Extensive surgery for carcinoma (e.g., oesophagectomy, gastrectomy cystectomy)
-
Acute abdominal catastrophe with hemodynamic instability (e.g., peritonitis, perforated viscus, pancreatitis)
-
Acute massive blood loss > 8 units
-
Septicemia
-
Positive blood culture or septic focus
-
Respiratory failure: PaO2 < 8.0 kPa on FIO2 > 0.4 or mechanical ventilation > 48 h
-
Acute renal failure: urea > 20 mmol/l or creatinine > 260 mmol/l
Another widely used, surgery-related score, the Portsmouth Physiological and Operative Severity Score for the enUmeration of Mortality and morbidity (P-POSSUM) includes 12 basic preoperative physiologic factors and 6 operative factors. Operative factors may be calculated before surgery, thus enabling a rough estimation of the perioperative risk, but may also be modified after surgery. As some of the parameters are available in the postoperative setting, this score should be used for preoperative risk stratification with caution.
It has to be noted that not all high-risk patients will be transferred to the ICU after surgery. ICU admissions from the operating theater may be categorized as planned and unplanned admissions. Planned ICU admissions in high-risk patients may occur in 5–30% of the cases depending on the local availability of perioperative critical care beds, hospital organization, and local regulations [1, 3, 4•]. Although the availability of perioperative ICU beds may improve outcomes [4•], data are suggesting that adding more ICU beds just adds lower priority ICU admissions, without changing overall hospital mortality [7]. The key here is the proper selection of patients who will benefit from ICU admissions. According to international priority criteria for admission to the ICU, these patients may be priority 1 (= needing immediate organ-supporting treatment, such as ventilatory support, vasoactive medications, renal support) and priority 2 patients (= needing advance monitoring and immediate intervention when needed). One has to realize that monitoring opportunities and the number of qualified staff members may be low in surgical wards; thus, admitting priority 2 patients may result in late ICU admissions. In a large study [1], admission of standard-risk patients resulted in high mortality when they had to be transferred later to the ICU. Proper patient selection and timing of ICU admission are of critical importance because delayed admission is associated with increased mortality [8]. Thus, besides preoperative risk assessment and defining patients who might be at higher risk, it is the anesthesiologist’s responsibility to make a decision at the end of surgery about ICU admission. High-risk surgical patients have increased risk to get decompensated usually in the first 24–48 h postoperatively and thus the effectiveness of the anesthesiologist’s activity extends well beyond what happens in the operating rooms. Several factors help optimize the patient’s condition and thus either avoid ICU admissions or to contribute to a more stable condition before transport. The list of potential measures to be taken by the anesthesiologist in high-risk surgical patients is summarized in Table 1.
The admittance rate of high-risk patients to ICU after emergency and urgent surgical interventions is higher than that after elective, planned surgeries. In these patients, the opportunities for hemodynamic stabilization and preoperative preparation of the patients are limited and therefore the role of the anesthesiologist’s work to achieve stable vital parameters during surgery is of paramount importance.
The Decision About ICU Admission and Organization of Transport
It is the anesthesiologist’s responsibility to decide the level of required care for the patient after surgery, with the operating surgeons involved. Ideally, the necessity of ICU admission can be unequivocally decided before starting surgery and the critical care team can be informed about the demand on the bed. This advance notification is the anesthesiologist’s responsibility. According to several studies, ICU team members require this notification because then they have the opportunity to prepare necessary equipment and supplies and thus handoff may be more patient-focused [16, 17••, 18]. For unanticipated ICU admissions (due to emergency high-risk surgeries or intraoperative complications), it is also required that the senior anesthesiologist of the team warns the ICU members about the demand on critical care beds as early as possible. Ideally, this includes pre-handoff communication 30–60 min before the end of surgery and an immediate warning before the transport [16]. It has been proven that patient transport by the anesthesiologist team is as safe as transporting patients by ICU specialists. ICU service can improve turnover times and workflow in the operating room (OR) [19].
Organizing Transport
There are different structures of OR and critical care worldwide. In some countries, anesthesiologists and intensive care specialist share a common specialization and thus perioperative and postoperative care occurs in the frame of one department. In other countries, anesthesia and intensive care are split into two specializations. Thus, in some institutions, a dedicated team of the critical care unit is responsible for the transport, while in others it is the anesthesia team’s responsibility. Whatever the case is, it is important to have a local protocol describing the members of teams and their responsibilities during transport from the OR to the ICU.
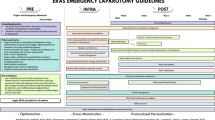
Most important serious adverse events during transport make up approximately 4.2–8.9% of critically ill cases. The majority of them are cardiovascular (severe alterations in blood pressure, arrhythmias, cardiac arrest), neurological (agitation or elevation of intracranial pressure), respiratory (patient-ventilator asynchrony, hypercapnia, hypoxia, bronchospasm, accidental extubation), and hypothermia [19]. Some of the incidents are related to organizational or human errors, such as equipment malfunction due to battery/electric or oxygen tank failure. To avoid them, every institution should work out a checklist of devices that have to be readily available for the transport of critically ill patients. In some institutions, one of the biggest concerns with transitioning care from the OR to the ICU is when the OR team boluses vasoactive medications just before handoff to prevent suboptimal hemodynamics during handoff. While this can improve the clinical picture, the effects are often transient and the patient becomes hypotensive shortly after handoff has occurred once the medication effect has ceased. Although bolus doses of vasopressors administered before transport may result in a temporary elevation of blood pressure in critically ill hypotensive patients, there are results indicating worse outcomes and increased mortality [20]. To avoid this suboptimal practice, vasoactive drugs should be continuously administered through perfusion during transportation and attention should be paid to the proper volume replacement therapy. The necessary drugs and devices are summarized in Fig. 1. It is the dedicated transport team’s responsibility to control the readiness of all types of equipment for transport (recharging batteries, checking oxygen vials) and the completeness of the drug lists. It has been proven that the use of locally developed checklists and regular training of the staff being involved in the transport procedure may largely improve patient safety [21].
Patient Handover
Several studies indicated that approximately half of the critical incidents occurring during transport are human-related [22]. Among them, information transfer during patient handover is one of the most important. It has been shown that poor communication is the second most common cause of critical incidents, corresponding to 14% [23], and patients whose transport team exhibited less detailed information sharing at ICU handover were at higher risk for critical complications [24]. It has to be emphasized that an adequate time span and quiet environment have to be ensured for an appropriate handover to avoid incomplete information transfer [25]. Optimally, a detailed handover has to include basic patient-related information, including age, previous diseases, and indications for surgery followed by information related to anesthesia and the surgical intervention. A very important part of patient handover should be related to the transport itself; all eventual incidents that occur during transportation have to be documented and verbally shared. The status of the patient at the time of handover has to be checked and documented in common by the transport and the ICU team members. The most important information about patient handover is summarized in Table 2. In many countries, the flowchart of handover communication is based on the SBAR (Situation, Background, Assessment, Recommendation) technique [26].
It has to be underlined that it is not enough to have a detailed description of handover at the institutions, a regular teaching and simulation practice program are necessary for the staff involved in the procedure, and it has to become an integral part of both the gradual and residency teaching programs [27••, 28, 29].
Conclusion
Preparing critically ill patients for ICU transfer starts in the preoperative phase by optimization of the patient’s status. Anesthesiologists are involved in the intraoperative and postoperative phases of stabilization. An important task is to organize transport and handover, which need interdisciplinary communication and cooperation.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Jhanji S, Thomas B, Ely A, Watson D, Hinds CJ, Pearse RM. Mortality and utilisation of critical care resources amongst high-risk surgical patients in a large NHS trust. Anaesthesia. 2008;63(7):695–700.
Pearse RM, Harrison DA, James P, Watson D, Hinds C, Rhodes A, Grounds RM, Bennett ED. Identification and characterisation of the high-risk surgical population in the United Kingdom. Crit Care. 2006;10(3):R81.
Pearse RM, Moreno RP, Bauer P, Pelosi P, Metnitz P, Spies C, Vallet B, Vincent JL, Hoeft A, Rhodes A; European Surgical Outcomes Study (EuSOS) group for the Trials groups of the European Society of Intensive Care Medicine and the European Society of Anaesthesiology. Mortality after surgery in Europe: a 7 day cohort study. Lancet. 2012;380(9847):1059–65.
Gillies MA, Power GS, Harrison DA, Fleming A, Cook B, Walsh TS, Pearse RM, Rowan KM. Regional variation in critical care provision and outcome after high-risk surgery. Intensive Care Med. 2015;41(10):1809–16. Detailed description on local differences and barriers of ICU-bed availability after high risk surgery.
Wolters U, Wolf T, Stutzer H, Schroder T. ASA classification and perioperative variables as predictors of postoperative outcome. Br J Anaesth. 1996;77:217–22.
Boyd O, Jackson N. How is risk defined in high-risk surgical patient management? Crit Care. 2005;9:390–6. A good description on how high risk surgery should be defined.
Howell MD, Stevens JP. ICUs after surgery, mortality, and the Will Rogers effect. Intensive Care Med. 2015;41(11):1990–2.
Bing-Hua YU. Delayed admission to intensive care unit for critically surgical patients is associated with increased mortality. Am J Surg. 2014;208(2):268–74.
Pearse R, Dawson D, Fawcett J, Rhodes A, Grounds RM, Bennett ED. Early goal-directed therapy after major surgery reduces complications and duration of hospital stay. A randomised, controlled trial [ISRCTN38797445]. Crit Care. 2005;9(6):R687-93.
Clevenger B, Mallett SV, Klein AA, Richards T. Patient blood management to reduce surgical risk. Br J Surg. 2015;102(11):1325–37.
Wick EC, Grant MC, Wu CL. Postoperative multimodal analgesia pain management with nonopioid analgesics and techniques: A review. JAMA Surg. 2017;152:691–7.
Sessler DI, Sigl JC, Kelley SD, Chamoun NG, Manberg PJ, Saager L, Kurz A, Greenwald S. Hospital stay and mortality are increased in patients having a “triple low” of low blood pressure, low bispectral index, and low minimum alveolar concentration of volatile anesthesia. Anesthesiology. 2012;116(6):1195–203.
Sessler DI. Perioperative thermoregulation and heat balance. Lancet. 2016;387(10038):2655–64.
Naguib M, Brull SJ, Kopman AF, Hunter JM, Fülesdi B, Arkes HR, Elstein A, Todd MM, Johnson KB. Consensus Statement on Perioperative Use of Neuromuscular Monitoring. Anesth Analg. 2018;127(1):71–80. A very important consensus statement on the necessity and importance of neuromuscular monitoring.
Prielipp RC, Fulesdi B, Brull SJ. Postoperative opioid-induced respiratory depression: 3 steps forward. Anesth Analg. 2020;131(4):1007–11.
McElroy LM, Macapagal KR, Collins KM, Abecassis MM, Holl JL, Ladner DP, Gordon EJ. Clinician perceptions of operating room to intensive care unit handoffs and implications for patient safety: a qualitative study. Am J Surg. 2015;210:629–35.
Segall N, Bonifacio AS, Schroeder RA, Barbeito A, Rogers D, Thornlow DK, Emery J, Kellum S, Wright MC, Mark JB; Durham VA Patient Safety Center of Inquiry. Can we make postoperative patient handovers safer? A systematic review of the literature. Anesth Analg. 2012;115:102–15. An important systematic review on patient handover.
Caruso TJ, Marquez JLS, Gipp MS, Kelleher SP, Sharek PJ. Standardized ICU to OR handoff increases communication without delaying surgery. Int J Health Care Qual Assur. 2017;30(4):304–11.
Waydhas C. Intrahospital transport of critically ill patients. Crit Care. 1999;3:R83-89.
Guyette FX, Martin-Gill C, Galli G, McQuaid N, Elmer J. Bolus Dose Epinephrine Improves Blood Pressure but is Associated with Increased Mortality in Critical Care Transport. Prehosp Emerg Care. 2019;23:764–71.
Fanara B, Manzon C, Barbot O, Desmettre T, Capellier G. Recommendations for the intra-hospital transport of critically ill patients. Crit Care. 2010;14:R87.
Beckmann U, Gillies DM, Berenholtz SM, Wu AW, Pronovost P. Incidents relating to the intra-hospital transfer of critically ill patients. An analysis of the reports submitted to the Australian Incident Monitoring Study in Intensive Care. Intensive Care Med. 2004;30:1579–85.
Kluger MT, Bullock MF. Recovery room incidents: a review of 419 reports from the Anaesthetic Incident Monitoring Study (AIMS). Anaesthesia. 2002;57:1060–6.
Mazzocco K, Petitti DB, Fong KT, Bonacum D, Brookey J, Graham S, Lasky RE, Sexton JB, Thomas EJ. Surgical team behaviors, and patient outcomes. Am J Surg. 2009;197:678–85.
Dusse F, Pütz J, Böhmer A, Schieren M, Joppich R, Wappler F. Completeness of the operating room to intensive care unit handover: a matter of time? BMC Anesthesiol. 2021;21:38.
Müller M, Jürgens J, Redaèlli M, Klingberg K, Hautz WE, Stock S. Impact of the communication and patient hand-off tool SBAR on patient safety: a systematic review. BMJ Open. 2018;8(8):e022202.
Lane-Fall MB, Brooks AK, Wilkins SA, Davis JJ, Riesenberg LA. Addressing the mandate for hand-off education: a focused review and recommendations for anesthesia resident curriculum development and evaluation. Anesthesiology. 2014;120:218–29. On the importance of education in the handover of patients.
Allen S, Caton C, Cluver J, Mainous AG 3rd, Clyburn B. Targeting improvements in patient safety at a large academic center: an institutional handoff curriculum for graduate medical education. Acad Med. 2014;89:1366–9.
Dupont FW, Tung A, Shahul S, Pohlman A, Silas J, Gottlieb O, O’Connor FO, Cutter TW. Transport of critically ill patients by the anesthesia versus the intensive care unit service: a before-after study of operating room workflows. Anesth Analg. 2019;129:671–8.
Funding
Open access funding provided by University of Debrecen.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they do not have any conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Patient Safety in Anesthesia
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
László, I., Végh, T., Szántó, D. et al. Preparing the Patient for ICU Transfer: What Is the Anesthesiologist’s Role?. Curr Anesthesiol Rep 12, 461–466 (2022). https://doi.org/10.1007/s40140-022-00543-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40140-022-00543-z