Abstract
Introduction
This study aimed to evaluate the effect of pre-operative versus pre-operative plus post-operative intravitreal conbercept (IVC) injection on severe proliferative diabetic retinopathy (PDR).
Methods
This was a prospective, comparative and randomised study. A total of 84 patients who underwent vitrectomy for severe PDR were included in this study. Patients were randomly divided into control (41 eyes) and experiment (43 eyes) groups. Patients in the experiment group received adjunctive pre-operative and post-operative IVC injection, whereas patients in the control group only received pre-operative IVC injection. The incidence of post-operative vitreous haemorrhage (POVH), best-corrected visual acuity (BCVA) and central retinal thickness (CRT) were determined.
Results
The incidence of early POVH was significantly different between the two groups, but no significant difference was observed between groups at 3 and 6 months. In the experiment group, the BCVA was significantly improved 1 month after surgery when compared with the control group (p 0.019). There was no marked difference in the mean post-operative BCVA at 3 and 6 months between groups (p 0.063 and 0.082). CRT was significantly lower in the experiment group than in the control group at 1 and 3 months after surgery (p 0.037 and 0.041), but there was no significant difference at 6 months (p 0.894).
Conclusion
Additional IVC injected at the end of surgery improves the POVH and BCVA at the early stage after surgery in severe PDR, but this benefit is absent at 6 months. Further studies are needed to investigate the effect of IVC at the end of vitrectomy.
Trial Registration
chictr.org.cn identifier: ChiCTR2200060735. Retrospectively registered, register date: 9 June 2022.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This study investigated the effects of pre-operative and post-operative IVC on severe PDR. |
The adjunctive use of pre-operative and post-operative IVC is a safe and effective method. |
The adjunctive use of pre-operative and post-operative IVC can reduce early POVH recurrence and improve visual acuity quickly. |
Further studies are needed to investigate the effect of IVC at the end of vitrectomy. |
Introduction
Proliferative diabetic retinopathy (PDR) is an advanced stage of diabetic retinopathy (DR), which can lead to neovascular glaucoma, vitreous haemorrhage (VH), fibrovascular proliferation and tractional retinal detachment (TRD), eventually resulting in severe vision loss [1]. Although vitrectomy surgery has contributed significantly to the successful treatment of PDR, its effectiveness should be further improved. Recurrent non-clearing post-operative VH (POVH) after pars plana vitrectomy (PPV) is a major adverse event [2]. Some studies have reported that the incidence of POVH is 15–75% [3]. The non-clearing POVH not only affects patients vision acuity, but also hinders retina examination, which may delay treatments such as photocoagulation [2, 4].
Conbercept (Chengdu Kanghong Biotech Co., Ltd., Sichuan, China) is an anti-vascular endothelial growth factor (VEGF) agent that has been approved by the China Food and Drug Administration for the treatment of age-related macular degeneration and macular oedema [5,6,7,8]. Recently, numerous studies have focused on the adjunctive use of conbercept in vitrectomy to reduce POVH for severe DR [9, 10]. Some studies have reported that pre-operative intravitreal conbercept (IVC) injection is effective to reduce the incidence of pre-operative and post-operative haemorrhage, shorten the mean operation time and improve the best-corrected visual acuity (BCVA) [11, 12]. However, the incidence of post-operative complications, such as VH, is still high [13]. This may be explained as anti-VEGF drugs are removed following vitrectomy in PPV, and simultaneously, PPV increases VEGF expression in response to surgical trauma and inflammation [14, 15]. Therefore, an additional injection of conbercept after vitrectomy may be needed. Some studies have reported that IVC, or intravitreal bevacizumab (IVB), after vitrectomy can improve BCVA and reduce POVH recurrence [16,17,18]. We hypothesised that a lower level of anti-VEGF drug and a high level of VEGF after surgery may contribute to the POVH recurrence, and pre-operative and post-operative intravitreal injection of conbercept may be effective to prevent POVH.
This study aimed to investigate the role of pre-operative and post-operative IVC injection in preventing POVH in patients undergoing vitrectomy for severe PDR.
Methods
This was a prospective, randomised clinical trial. The study was conducted according to the Declaration of Helsinki and approved by the Ethics Committee of Tongji Hospital, Tongji University. All patients provided informed consent before surgery. This study was registered in the Chinese Clinical Trial Registry.
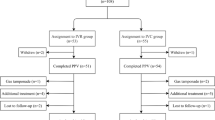
Randomisation was carried out using computer-generated random number table. A total of 86 eyes from 86 patients who received vitrectomy for severe PDR were included in the study. Only one eye of each patient was included in the study. Patients with the indications for PPV, including non-clearing VH and fibrovascular proliferation without TRD, were enrolled. The exclusion criteria included a history of PPV, intra-operative use of C3F8 or silicon oil, history of macular-involved TRD, history of intravitreal injection of any anti-VEGF drug within 3 months, ocular disease other than DR and uncontrolled hypertension.
Before surgery, all patients underwent BCVA measurement, slit-lamp biomicroscopy, intraocular pressure (IOP) examination, fundus examination and eye ultrasonography. Visual acuity was converted to logarithm of the minimum angle of resolution (LogMAR) for analysis, ‘counting fingers’ was defined as 2.0 LogMAR and ‘hand motion’ was defined as 3.0 LogMAR [19, 20]. All patients received an IVC (conbercept, 10 mg/0.05 ml) injection before surgery (3–5 days before surgery). Then, a standard three-port 25-gauge trans-conjunctival vitrectomy was performed under local anaesthesia. The surgical procedures were performed by the same surgeon using the 25-gauge vitrectomy system (constellation surgical system, Alcon Surgical Inc., Fort Worth, TX). The maximum removal of the vitreous body was achieved by using a wide-angle visualisation system and pressing on the peripheral sclera. Triamcinolone acetonide was used to visualise the vitreous body, which was completely removed from the vitreous cavity after vitrectomy. Fibrovascular tissues were peeled and dissected, internal diathermy was applied if necessary and pan-retinal photocoagulation was performed in every patient. The IOP reduced at the end of surgery to ensure that any potential bleeding sites were identified and blocked. The leaking sclerotomy sites were sutured to prevent post-operative hypotony. At the end of surgery, patients in the experiment group received IVC (conbercept, 10 mg/0.05 ml) injection through the pars plana and patients in the control group did not receive IVC injection.
All patients were examined 1 day, 1 week, 1 month, 3 months and 6 months after surgery. At each post-operative visit, examinations of BCVA, IOP, optical coherence tomography, ultrasonography and colour fundus photography were performed. The VH severity was graded by indirect ophthalmoscopic fundus examination according to the grading system (Table 1). Early POVH was defined as VH occurring between 1 week and 1 month after surgery, and delayed POVH was defined as VH occurring between 1 and 6 months after surgery.
Data analysis was performed using the SPSS software package (version 23.0). Data are presented as mean ± standard deviation (SD). Intergroup differences were assessed using a t-test, Pearson chi-square test, chi-square test or Fisher’s exact test. After normal distribution test, one-way analysis of variance was used for data with normal distribution, and Kruskal–Wallis test was used for data without normal distribution. A value of p < 0.05 was considered statistically significant.
Results
A total of 86 eyes of 86 patients were included in this study, with 2 eyes being excluded. The two eyes underwent silicone oil tamponade because of retinal detachment after surgery. Thus, 84 eyes of 84 patients were included for final analysis. They all had IVC injection 3–5 days before surgery. Patients in the control group (41 eyes) did not receive an additional IVC injection at the end of the surgery, whereas patients in the experiment group (43 eyes) received a second IVC injection at the end of surgery. Table 2 presents the demographics of patients in two groups. Results showed that there were no marked differences in the age, sex, disease, lens status, pre-operative BCVA and systemic disease status between both groups.
The incidence of early POVH was 19.5% and 9.3% in the control group and experiment group, respectively, showing significant difference between groups (p 0.046). The incidence of delayed POVH was similar between groups (p 0.710) (Table 3).
In both groups, the BCVA was improved within 6 months (Table 3). The mean post-operative BCVA increased significantly from 1.37 ± 0.34 at baseline to 0.39 ± 0.13 at 6 months in the control group (p < 0.001). The mean post-operative BCVA increased significantly from 1.41 ± 0.41 at baseline to 0.34 ± 0.16 at 6 months in the experiment group (p < 0.001). The mean post-operative BCVA at 1 month was 0.71 ± 0.15 LogMAR in the control group and 0.57 ± 0.11 LogMAR in the experiment group, showing significant difference between groups (p = 0.019). However, the mean post-operative BCVA at 3 and 6 months was comparable between two groups (p = 0.063 and 0.082).
Central retinal thickness (CRT) significantly decreased at 6 months compared with that at 1 month in both groups (Table 3). The CRT was significantly different between two groups at 1 month and 3 months (p 0.037 and 0.041), whereas no marked difference was observed at 6 months (p 0.894).
Discussion
This study compared the effect of pre-operative and post-operative IVC injection with that of a single pre-operative IVC injection. Our results showed that adjunctive use of pre-operative and post-operative IVC for severe PDR achieved thinner CRT, low incidence of POVH and better BCVA at the early stage after surgery. However, the outcomes at 6 months were comparable to those after a single IVC before surgery.
POVH is a severe complication of PPV in patients with PDR, and its treatment is still a challenge in clinical practice [4]. POVH may occur at any time after surgery [13]. POVH may become recurrent despite a successful vitrectomy [21]. The causes of POVH are complex, including the fibrovascular proliferation at the sclerotomy site and the retraction of fibrovascular tissue remnants [13].
VEGF is an important angiogenic factor and is involved in the proliferation of DR [22]. Nevertheless, many studies have reported that the VEGF expression increases in patients with DR who receive ocular surgery, which may be ascribed to the surgical trauma [23, 24]. Patel et al. reported an increase of VEGF expression in the aqueous humour of patients with DR undergoing phacoemulsification [14]. Funatsu et al. [25] found that high VEGF expression in the vitreous fluid was a significant risk factor for the progression and outcome of PDR after vitreous surgery. VEGF induced by surgery can cause some complications such as iris neovascularisation and fibrovascular proliferation, leading to POVH.
Since IVC or IVB injection can diminish the VEGF level in the eye, some studies have reported that anti-VEGF therapy pre-operatively as an adjunct to vitrectomy for severe PDR can significantly reduce the incidence of POVH [17, 26, 27]. However, other studies show that anti-VEGF therapy cannot prevent POVH [28,29,30]. Of note, almost all studies confirm that pre-operative intravitreal anti-VEGF injection makes surgery easier and can shorten the operation time and reduce post-operative bleeding [31, 32].
The half-life of conbercept in the vitreous body is about 4–5 days in rabbit and monkey eyes [5, 33]. Pre-operative intravitreal anti-VEGF injection is not effective to prevent POVH, which may be explained as follows: the anti-VEGF drug is removed during PPV, which limits the anti-angiogenic effect. Therefore, a second IVC injection at the end of the operation may prolong the anti-angiogenic effect and inhibit VEGF elevation and neovascularisation. The main cause of early POVH is the bleeding of scleral incision sites and fibrovascular tissue remnants [24, 34]. VEGF inhibition can inhibit the regression of neovascularisation, which was supported by our findings: additional IVC injection at the end of vitrectomy decreased the incidence of early POVH, especially within 1 week. However, the pharmacokinetics of intravitreal drugs in the vitrectomised eyes are different from that in normal eyes. The clearance rate in the normal eyes is about three times that in the non-vitrectomised eyes [35]. The anti-VEGF drugs may exert anti-angiogenic effect during the early post-operative period, and they are insufficient to exert long term inhibitory effects on POVH as demonstrated by the absence of significant difference between groups at 6 months.
Our results showed that patients who received IVC injection at the end of surgery had better BCVA at the early post-operative visit, which may be related to the decreased macular oedema, because macular oedema can significantly affect visual acuity [36,37,38]. In this study, CRT was measured post-operatively. CRT decreased gradually in both groups. IVC injection at the end of surgery significantly reduced CRT at the early post-operative visit.
However, this study had several limitations. First, the sample size was relatively small. Second, the follow-up duration was short. Therefore, more studies are needed to confirm the effect of IVC at the end of vitrectomy.
Conclusions
In conclusion, this study investigates the effects of pre-operative and post-operative IVC on the severe PDR. Our results indicate that the adjunctive use of pre-operative and post-operative IVC is a safe and effective method. It can reduce early POVH recurrence and improve visual acuity quickly. Further studies are needed to investigate the effect of IVC at the end of vitrectomy.
References
Ozone D, et al. Outcomes and complications of 25-gauge transconjunctival sutureless vitrectomy for proliferative diabetic retinopathy. Ophthalmologica. 2011;226(2):76–80.
Al-Khersan H, et al. Pars plana vitrectomy reoperations for complications of proliferative diabetic retinopathy. Clin Ophthalmol. 2020;14:1559–63.
Schachat AP, et al. Complications of vitreous surgery for diabetic retinopathy. II Postoperative complications. Ophthalmology. 1983;90(5):522–30.
Khuthaila MK, et al. Postoperative vitreous hemorrhage after diabetic 23-gauge pars plana vitrectomy. Am J Ophthalmol. 2013;155(4):757-763.e1-2.
Li H, et al. Pharmacokinetics of a long-lasting anti-VEGF fusion protein in rabbit. Exp Eye Res. 2012;97(1):154–9.
Guo J, et al. Clinical effect of vitreoretinal surgery combined with intravitreal injection of conbercept or ranibizumab on severe proliferative diabetic retinopathy. Chin J Exp Ophthalmol. 2017;35(10):914–9.
Lu X, Sun X. Profile of conbercept in the treatment of neovascular age-related macular degeneration. Drug Des Dev Ther. 2015;9:2311–20.
Liu K, et al. Conbercept for treatment of neovascular age-related macular degeneration: results of the randomized phase 3 PHOENIX study. Am J Ophthalmol. 2019;197:156–67.
Su L, et al. Intravitreal conbercept (KH902) for surgical treatment of severe proliferative diabetic retinopathy. Retina. 2016;36(5):938–43.
Gao S, et al. Intravitreal conbercept injection as an adjuvant in vitrectomy with silicone oil infusion for severe proliferative diabetic retinopathy. J Ocul Pharmacol Ther. 2020;36(5):304–10.
Mao JB, et al. Effect of intravitreal conbercept treatment before vitrectomy in proliferative diabetic retinopathy. Int J Ophthalmol. 2018;11(7):1217–21.
Pranata R, Vania A. Intravitreal conbercept improves outcome in patients undergoing vitrectomy for proliferative diabetic retinopathy: a systematic review and meta-analysis. J Evid Based Med. 2020;13(2):116–24.
Shi L, Huang YF. Postvitrectomy diabetic vitreous hemorrhage in proliferative diabetic retinopathy. J Res Med Sci. 2012;17(9):865–71.
Patel JI, Hykin PG, Cree IA. Diabetic cataract removal: postoperative progression of maculopathy—growth factor and clinical analysis. Br J Ophthalmol. 2006;90(6):697–701.
Stefansson E. Physiology of vitreous surgery. Graefes Arch Clin Exp Ophthalmol. 2009;247(2):147–63.
Ren X, et al. Safety and efficacy of intravitreal conbercept injection after vitrectomy for the treatment of proliferative diabetic retinopathy. Eye (Lond). 2019;33(7):1177–83.
Cheema RA, et al. Role of intravitreal bevacizumab (Avastin) injected at the end of diabetic vitrectomy in preventing postoperative recurrent vitreous hemorrhage. Retina. 2010;30(10):1646–50.
Liang X, et al. Intravitreal ranibizumab injection at the end of vitrectomy for diabetic vitreous hemorrhage (observational study). Medicine. 2019;98(20):e15735.
Holladay JT. Proper method for calculating average visual acuity. J Refract Surg. 1997;13(4):388–91.
Holladay JT. Visual acuity measurements. J Cataract Refract Surg. 2004;30(2):287–90.
Sato T, et al. Early vitreous hemorrhage after vitrectomy with preoperative intravitreal bevacizumab for proliferative diabetic retinopathy. Middle East Afr J Ophthalmol. 2013;20(1):51–5.
Wong TY, et al. Diabetic retinopathy. Nat Rev Dis Primers. 2016;2:16012.
Petrovič MG, et al. Association of preoperative vitreous IL-8 and VEGF levels with visual acuity after vitrectomy in proliferative diabetic retinopathy. Acta Ophthalmol. 2010;88(8):e311–6.
Ding Y, et al. Multiple factors in the prediction of risk of recurrent vitreous haemorrhage after sutureless vitrectomy for non-clearing vitreous haemorrhage in patients with diabetic retinopathy. BMC Ophthalmol. 2020;20(1):292.
Funatsu H, et al. Risk evaluation of outcome of vitreous surgery based on vitreous levels of cytokines. Eye (Lond). 2007;21(3):377–82.
Park DH, Shin JP, Kim SY. Intravitreal injection of bevacizumab and triamcinolone acetonide at the end of vitrectomy for diabetic vitreous hemorrhage: a comparative study. Graefes Arch Clin Exp Ophthalmol. 2010;248(5):641–50.
Ahn J, et al. The effect of adjunctive intravitreal bevacizumab for preventing postvitrectomy hemorrhage in proliferative diabetic retinopathy. Ophthalmology. 2011;118(11):2218–26.
Jirawison C, Ittipunkul N. Intravitreal bevacizumab at the end of diabetic vitrectomy for prevention of postoperative vitreous hemorrhage: a comparative study. J Med Assoc Thai. 2012;95(Suppl 4):S136–42.
Göncü T, Özdek S, Ünlü M. The role of intraoperative bevacizumab for prevention of postoperative vitreous hemorrhage in diabetic vitreous hemorrhage. Eur J Ophthalmol. 2014;24(1):88–93.
Jiang T, et al. The effect of adjunctive intravitreal conbercept at the end of diabetic vitrectomy for the prevention of post-vitrectomy hemorrhage in patients with severe proliferative diabetic retinopathy: a prospective, randomized pilot study. BMC Ophthalmol. 2020;20(1):43.
Castillo J, et al. Preoperative bevacizumab administration in proliferative diabetic retinopathy patients undergoing vitrectomy: a randomized and controlled trial comparing interval variation. Am J Ophthalmol. 2017;183:1–10.
Arevalo JF, et al. Preoperative bevacizumab for tractional retinal detachment in proliferative diabetic retinopathy: a prospective randomized clinical trial. Am J Ophthalmol. 2019;207:279–87.
Zhang M, et al. The pharmacology study of a new recombinant human VEGF receptor-fc fusion protein on experimental choroidal neovascularization. Pharm Res. 2009;26(1):204–10.
Taskintuna I, et al. Comparison of outcomes of four different treatment modalities for diabetic vitreous haemorrhage. Sci Rep. 2020;10(1):3674.
Ficker L, et al. Cefazolin levels after intravitreal injection. Effects of inflammation and surgery. Investig Ophthalmol Vis Sci. 1990;31(3):502–5.
Baker CW, et al. Effect of initial management with aflibercept vs laser photocoagulation vs observation on vision loss among patients with diabetic macular edema involving the center of the macula and good visual acuity: a randomized clinical trial. JAMA. 2019;321(19):1880–94.
Glassman AR, et al. Assessment of the DRCR retina network approach to management with initial observation for eyes with center-involved diabetic macular edema and good visual acuity: a secondary analysis of a randomized clinical trial. JAMA Ophthalmol. 2020;138(4):341–9.
Zafar S, et al. Real-world outcomes among eyes with center-involving diabetic macular edema and good visual acuity. Curr Eye Res. 2020;45(7):879–87.
Acknowledgments
Funding
No funding or sponsorship was received for this study or publication of this article. The journal’s Rapid Service Fee was funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Houshuo Li, Yunli Niu, Ao Rong, Yanlong Bi, Wei Xu and Hongping Cui. The manuscript was drafted by Houshuo Li and all authors commented on the manuscript. All authors read and approved the final manuscript.
Disclosures
Houshuo Li, Yunli Niu, Ao Rong, Yanlong Bi, Wei Xu and Hongping Cui declare that they have no conflict of interest.
Compliance with Ethics Guidelines
This study has been approved by the Research Ethics Committee of Tongji Hospital, Tongji University. All subjects provided informed consent to participate in the study, which was conducted according to the Declaration of Helsinki.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Li, H., Niu, Y., Rong, A. et al. Effect of Adjunctive Intravitreal Conbercept Injection at the End of 25G Vitrectomy on Severe Proliferative Diabetic Retinopathy: 6-Month Outcomes of a Randomised Controlled Trial. Ophthalmol Ther 12, 1173–1180 (2023). https://doi.org/10.1007/s40123-023-00664-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-023-00664-6