Abstract
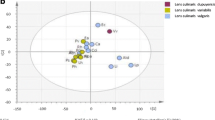
The lipophilic compounds in nine vegetables consumed in Korea were characterized for diversity in phytochemical content. We also analyzed the relationships among these compounds in terms of their contents. The profiles of 18 lipophilic compounds in the leaves were subjected to data-mining processes, including principal component analysis (PCA), Pearson’s correlation analysis, hierarchical clustering analysis (HCA), and partial least squares discriminant analysis (PLS-DA). These species could be distinguished by means of the PCA results. HCA of these phytochemicals resulted in clusters derived from closely related biochemical pathways. PLS-DA showed significant separation among extracts from the following four families: Amaranthaceae, Asteraceae, Brassicaceae, and Malvaceae. The major metabolites that facilitated differentiation in the PLS-DA model were campesterol, β-sitosterol, β-amyrin, stigmasterol, cholesterol, octacosanol (c28), α-amyrin, and hexacosanol (c26). Chard contained high levels of triacontanol (c30), which was positively correlated with the c28 content (r = 0.746, p < 0.0001). Stigmasterol, α-amyrin, and β-amyrin contents were higher in Asteraceae species including chicon, lettuce, and oak than other plant families. These results demonstrate the utility of metabolic profiling, combined with multivariate analysis, for discrimination of vegetable species as well as evaluation of food quality.





Similar content being viewed by others
References
Adhikari P, Hwang KT, Park JN, Kim CK (2006) Policosanol content and composition in perilla seeds. J Agric Food Chem 54:5359–5362
Agren JJ, Tvrzicka E, Nenonen MT, Helve T, Hänninen O (2001) Divergent changes in serum sterols during a strict uncooked vegan diet in patients with rheumatoid arthritis. Br J Nutr 85:137–139
Arruzazabala ML, Carbajal D, Mas R, Garcia M, Fraga V (1993) Effects of policosanol on platelet aggregation in rats. Thromb Res 69:321–327
Arruzazabala ML, Molina V, Mas R, Fernández L, Carbajal D, Valdés S, Castaño G (2002) Antiplatelet effects of policosanol (20 and 40 mg/day) in healthy volunteers and dyslipidaemic patients. Clin Exp Pharmacol Physiol 29:891–897
Bai S (1997) Effects of octacosanol in food physiological parameters in tail-suspended rats. Space Med Med Eng 10:450–452
Eriksson L, Johansson E, Kettaneh-Wold N, Wold S (2001) Multi- and megavariate data analysis principles and applications. Umetrics AB, Umeå
Goodacre R, Roberts L, Ellis DI, Thorogood D, Reader SM, Ougham H, King I (2007) From phenotype to genotype: whole tissue profiling for plant breeding. Metabolomics 3:489–501
Gouni-Berthold I, Berthold HK (2002) Policosanol: clinical pharmacology and therapeutic significance of a new lipid-lowering agent. Am Heart J 143:356–365
Haffner SM (2000) Clinical relevance of the oxidative stress concept. Metabolism 49:30–34
Hwang KT, Weller CL, Cuppett SL, Hanna MA (2004) Policosanol contents and composition of grain sorghum kernels and dried distillers grains. Cereal Chem 81:345–349
Irmak S, Dunford NT (2005) Policosanol contents and compositions of wheat varieties. J Agric Food Chem 53:5583–5586
Kim HE, Choi YH, Choi KH, Park JS, Kim HS, Jeon JH, Heu MS, Shin DS, Lee JH (2012a) Metabolic classification of herb plants by NMR-based metabolomics. J Korean Magn Reson Soc 16:91–102
Kim JK, Ha SH, Park SY, Lee SM, Kim HJ, Lim SH, Suh SC, Kim DH, Cho HS (2012b) Determination of lipophilic compounds in genetically modified rice using gas chromatography time-of-flight-mass spectrometry. J Food Compos Anal 25:31–38
Kim JK, Park SY, Jung JY, Ha SH, Lim SH, Lee SM, Woo HJ, Park SU, Suh SC (2012c) The policosanol content and composition of Korean rice (Oryza sativa L.) cultivars. Cereal Chem 89:151–154
Kim JK, Park SY, Na JK, Seong ES, Yu CY (2012d) Metabolite profiling based on lipophilic compounds for quality assessment of perilla (Perilla frutescens) cultivars. J Agric Food Chem 60:2257–2263
Kim YB, Park SY, Thwe AA, Seo JM, Suzuki T, Kim SJ, Kim JK, Park SU (2013) Metabolomic analysis and differential expression of anthocyanin biosynthetic genes in white- and red-flowered buckwheat cultivars (Fagopyrum esculentum). J Agric Food Chem 61:10525–10533
Lechner M, Reiter B, Lorbeer E (1999) Determination of tocopherols and sterols in vegetable oils by solid-phase extraction and subsequent capillary gas chromatographic analysis. J Chromatogr A 857:231–238
Lin Y, Rudrum M, van der Wielen RP, Trautwein EA, McNeill G, Sierksma A, Meijer GW (2004) Wheat germ policosanol failed to lower plasma cholesterol in subjects with normal to mildly elevated cholesterol concentrations. Metabolism 53:1309–1314
Pan O, Dai Y, Nuringtyas TR, Rianika RM, Schulte AE, Verpoorte R, Choi YH (2012) Investigation of the chemomarkers correlated with flower color in different organs of Catharanthus roseus using NMR-based metabolomics. Phytochem 25:66–74
Park SY, Choi SR, Lim SH, Yeo Y, Kweon SJ, Bae YS, Kim KW, Im KH, Ahn SK, Ha SH, Park SU, Kim JK (2014) Identification and quantification of carotenoids in paprika fruits and cabbage, kale, and lettuce leaves. J Korean Soc Appl Biol Chem 57:355–358
Piironen V, Lindsay DG, Miettinen TA, Toivo J, Lampi AM (2000) Plant sterols: biosynthesis, biological function and their importance to human nutrition. J Agric Food Chem 80:939–966
Pongsuwan W, Fukusaki E, Bamba T, Yonetani T, Yamahara T, Kobayashi A (2007) Prediction of Japanese green tea ranking by gas chromatography/mass spectrometry-based hydrophilic metabolite fingerprinting. J Agric Food Chem 55:231–236
Rusak DA, Brown LM, Martin SD (2003) Classification of vegetable oils by principal component analysis of FTIR spectra. J Chem Educ 80:541–543
Schummer C, Delhomme O, Appenzeller BMR, Wennig R, Millet M (2009) Comparison of MTBSTFA and BSTFA in derivatization reactions of polar compounds prior to GC/MS analysis. Talanta 77:1473–1482
Shepherd T, Dobson G, Verrall SR, Conner S, Griffiths DW, McNicol JW, Davies HV, Stewart D (2007) Potato metabolomics by GC–MS: what are the limiting factors? Metabolomics 3:475–488
Steuer R, Kurths J, Fiehn O, Weckwerth W (2003) Interpreting correlations in metabolomic networks. Biochem Soc Trans 31:1476–1478
Taylor JC, Rapport L, Lockwood GB (2003) Octacosanol in human health. Nutrition 19:192–195
Van Pelt CK, Haggarty P, Brenna T (1998) Quantitative subfemtomole analysis of α-tocopherol and deuterated isotopomers in plasma using tabletop GC/MS/MS. J Agric Food Chem 70:4369–4375
Acknowledgments
This work was supported by a grant from the Next-Generation BioGreen 21 Program (SSAC, PJ011094042015), Rural Development Administration, Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kim, T.J., Lee, K.B., Baek, SA. et al. Determination of lipophilic metabolites for species discrimination and quality assessment of nine leafy vegetables. J Korean Soc Appl Biol Chem 58, 909–918 (2015). https://doi.org/10.1007/s13765-015-0119-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13765-015-0119-6