Abstract
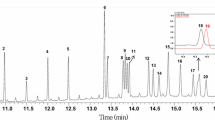
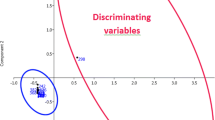
Food legumes (Fabaceae) form an important part of the human diet; besides, several Fabaceae species are acknowledged for their high levels of bioactive compounds, among which are isoflavones, being recognised for their varied types of biological activity. The aim of this work was to classify different varieties of three types of legumes (chickpeas, lentils and beans) according to their isoflavone contents. The analysis of isoflavones was carried out using high-performance liquid chromatography coupled to triple quadrupole tandem mass spectrometry (HPLC-MS/MS). To extract the analytes, a modified QuEChERS approach was used. The chromatographic peak areas obtained, after scaling in Pareto mode, were used to build statistical models. Both supervised and unsupervised techniques were applied for the classification of the different types of pulses analysed in the study: principal component analysis (PCA), hierarchical cluster analysis (HCA) and partial least squares discriminant analysis (PLS-DA). The statistical models were validated by internal validation, obtaining satisfactory results for the different matrices. PCA models allowed the differentiation between subspecies, but not subspecies, varieties or ecotypes. The results provided by HCA and PLS-DA revealed that the different species and subspecies of beans and the different varieties and subvarieties of lentils can be distinguished, and even the different ecotypes of the same variety in the case of chickpeas. This study revealed that it was possible to differentiate among species, subspecies, varieties and even ecotypes of different types of legumes based on their isoflavone content.




Similar content being viewed by others
References
Adlercreutz H (1998) Epidemiology of phytoestrogens. Baillière’s Clin Endocrinol Metab 12:605–623
Brereton R (2009) Chemometrics for pattern recognition. Wiley, New York, pp. 47–74
Bustamante-Rangel M, Pérez-Martín L, Delgado-Zamarreño MM (2014) Comparative study of the methodology used in the extraction of isoflavones from legumes applying a modified QuEChERS approach. Phytochem Anal 25:170–177
Cano A, García-Pérez MA, Tarín JJ (2010) Isoflavones and cardiovascular disease. Maturitas 67:219–226
Cassidy A, Albertazzi P, Nielsen IL, Hall W, Williamson G, Tetens I, Atkins S, Cross H, Manios Y, Wolk A, Steiner C, Branca F (2006) Critical review of health effects of soyabean phyto-oestrogens in post-menopausal women. Proc Nutr Soc 65:76–92
Delgado-Zamarreño MM, Pérez-Martín L, Bustamante-Rangel M, Carabias-Martínez R (2012) A modified QuEChERS method as sample treatment before the determination of isoflavones in foods by ultra-performance liquid chromatography–triple quadrupole mass spectrometry. Talanta 100:320–328
Eriksson L, Johansson E, Kettaneh-Wold N, Tryggs J, Wikstrom C, Wold S (2006) Multi- and megavariate data analysis. Part 1: basic principles and applications. Umetrics Academy, Umea, pp 361–380.
Frederick M, McCarty MF (2006) Isoflavones made simple—Genistein’s agonist activity for the beta-type estrogen receptor mediates their health benefits. Med Hypotheses 66:1093–1114
Hale GE, Hughes CL, Cline JM (2002) Endometrial cancer: hormonal factors, the perimenopausal “window of risk,” and isoflavones. J Clin Endocrinol Metab 87:3–15
Hwang CS, Kwak HS, Lim HJ, Lee SH, Kang YS, Choe TB, Hur HG, Han KO (2006) Isoflavone metabolites and their in vitro dual functions: they can act as an estrogenic agonist or antagonist depending on the estrogen concentration. J Steroid Biochem Mol Biol 101:246–253
Jackson JE (2004) A user’s guide to principal components. Wiley, New York, pp. 4–25
Jacobs A, Wegewitz U, Sommerfeld C, Grossklaus LA (2009) Efficacy of isoflavones in relieving vasomotor menopausal symptoms—a systematic review. Mol Nutr Food Res 53:1084–1097
Kumar V, Rani A, Dixit AK, Pratap D, Bhatnagar D (2010) A comparative assessment of total phenolic content, ferric reducing-anti-oxidative power, free radical-scavenging activity, vitamin C and isoflavones content in soybean with varying seed coat colour. Food Res Int 43:323–328
Lee SJ, Seguin P, Kim JJ, Moond HI, Ro HM, Kim EH, Seo SH, Kang EY, Ahn JK, Chung IM (2010) Isoflavones in Korean soybeans differing in seed coat and cotyledon color. J Food Compos Anal 23:160–165
Martin LM, Castilho MC, Silveira MI, Abreu JM (2006) Liquid chromatographic validation of a quantitation method for phytoestrogens, biochanin-A, coumestrol, daidzein, formononetin, and genistein, in lucerne. J Liq Chromatogr Rel Technol 29:2875–2884
Mazur WM, Duke JA, Wähälä K, Rasku S, Adlercreutz H (1998) Isoflavonoids and lignans in legumes: nutritional and health aspects in humans. J Nutr Biochem 9:193–200
Morris C, Thorpe J, Ambrosio L, Santin M (2006) The soybean isoflavone genistein induces differentiation of MG63 human osteosarcoma osteoblasts. J Nutr 136:1166–1170
Morrison MJ, Cober ER, Saleem MF, McLaughlin NB, Frégeau-Reid J, Ma BL, Woodrow L (2010) Seasonal changes in temperature and precipitation influence isoflavone concentration in short-season soybean. Field Crops Res 117:113–121
Ramis Ramos G, García Álvarez-Coque C (2001) Quimiometría. Síntesis S.A., Madrid.
Raschke M, Wähälä K, Pool-Zobel BL (2006) Reduced isoflavone metabolites formed by the human gut microflora suppress growth but do not affect DNA integrity of human prostate cancer cells. J Nutr 96:426–434
Rimbach G, Boesch-Saadatmandi C, Frank J, Fuchs D, Wenzel U, Daniel H, Hall WL, Weinberg PD (2008) Dietary isoflavones in the prevention of cardiovascular disease—a molecular perspective. Food Chem Toxicol 46:1308–1319
Sjostrom M, Wold S, Soderstrom B (1986) PLS discriminant plots. In: Pattern recognition in practice II. Elsevier, Amsterdam, p. 486
Trock BJ, Hilakivi-Clarke L, Clarke R (2006) Meta-analysis of soy intake and breast cancer risk. J Natl Cancer I 98:459–471
Vamerali T, Barion G, Hewidy M, Mosca G (2012) Soybean isoflavone patterns in main stem and branches as affected by water and nitrogen supply. Eur J Agron 41:1–10
Wood CE, Register TC, Franke AA, Anthony MS, Cline JM (2006) Dietary soy isoflavones inhibit estrogen effects in the postmenopausal breast. Cancer Res 66:1241–1249
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of Interest
Lara Pérez-Martín declares that she has no conflict of interest. Myriam Bustamante-Rangel declares that she has no conflict of interest. M. M. Delgado-Zamarreño declares that she has no conflict of interest.
Informed Consent
Not applicable.
Funding
This study was funded by the “Programa Propio de Financiación de Grupos de Investigación de la Universidad de Salamanca” Ref: USAL2015-16.
Rights and permissions
About this article
Cite this article
Pérez-Martín, L., Bustamante-Rangel, M. & Delgado-Zamarreño, M.M. Classification of Lentils, Chickpeas and Beans Based on Their Isoflavone Content. Food Anal. Methods 10, 1191–1201 (2017). https://doi.org/10.1007/s12161-016-0675-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-016-0675-3