Abstract
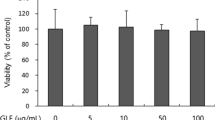
Three single compounds were isolated by the fractionation from Pruni persicae Flos, and its chemical structure was analyzed using 1H-, 13C-nuclear magnetic resonance, fast atom bombardment -Mass, and fourier transform infrared spectrometer analyses. The compounds were determined as quercetin 3-O-rhamnoside, kaempferol 3-O-β-neohesperidoside, and kaempferol 3-O-β-d-glucoside. To prove the whitening effect of these compounds, B16F10 melanoma was treated with quercetin-3-O-rhamnoside, kamferol-3-O-β-neohesperidoside or kaemferol-3-O-β-d-glucoside to confirm cell cytotoxicity using 3-[4,5-dimethylthiazol-2yl]-2,5-diphenyl-tetrazolium-bromide assay. As a result, cell cytotoxicity was examined at more than 20% in 20 μg/mL of compounds isolated from Pruni persicae Flos. Tyrosinase activity and melanin production were examined at 10 μg/mL of each compounds. Results showed quercetin-3-O-rhamnoside, kamferol-3-O-β-neohesperidoside or kaemferol-3-O-β-d-glucoside reduced tyrosinase activity in a dose-dependent manner. Moreover, quercetin-3-O-rhamnoside showed reduction of tyrosinase activity by 37.6% at 10 μg/mL of quercetin-3-O-rhamnoside as well as the reduction of melanin contents by 58.8%. In addition, quercetin-3-O-rhamnoside, kamferol-3-O-β-neohesperidoside, and kaemferol-3-O-β-d-glucoside reduced tyrosinase, tyrosinase-related proteins 1, and 2, microphthalmia associated transcription factor expressions, which were related to whitening effect, as well as both of mRNA and protein level of cyclic adenosine monophosphate as upstream signals. More interestingly, quercetin-3-O-rhamnoside strongly reduced both mRNA and protein expressions than other compounds. Therefore, we determined the whitening effect from isolated compound fractionated from Pruni persicae Flos. Moreover, strong whitening activity of quercetin-3-O-rhamnoside was examined, which suggested the potential application of this compound in functional cosmetic development.
Similar content being viewed by others
References
Abou-Zaid MM, Helson BV, Nozzolillo C, and Arnason JT (2001) Ethyl mdigallate from red maple, Acer rubrum L., as the major resistance factor to forest tent caterpillar, Malacosoma disstria Hbn. J Chem Ecol 27, 2517–27.
Akiu S, Suzuki Y, Asahara T, Fujinuma Y, and Fukuda M (1991) Inhibitory effect of arbutin on melanogenesis-biochemical study using cultured B16 melanoma cells. Nihon Hifuka Gakkai Zasshi 101, 609–13.
Alfonso C, Vincenzo DF, Ernesto F, Virginia L, Silvana M, and Carla C (1996) The flavonoids of Allium ursinum. Phytochemistry 41, 531–6.
Anil KK and Krishnaswamy NR (2000) Constituents of the flowers of Persea gratissima. Fitoterapia 71, 94–6.
Busca R and Ballotti R (2000) Cyclic AMP a key messenger in the regulation of skin pigmentation. Pigment Cell Res 13, 60–9.
Carmichael J, DeGraff WG, Gazdar AF, Minna JD, and Mitchell JB (1987) Evaluation of a tetrazolium based semiautomated colorimetric assay: assessment of chemosensitivity testing. Cancer Res 47, 936–42.
Chin JE and Kim KC (2005) Effect of Chestnut bark extracts on tyrosinase gene expression. Kor J Sanitation 20, 10–6.
Goding CR (2000) Mitf from neural crest to melanoma: signal transduction and transcription in the melanocyte lineage. Genes Dev 14, 1712–28.
Hunt G, Todd C, Cresswell JE, and Thody AJ (1994) Alpha-melanocyte stimulating hormone and its analogue Nle4DPhe7 alpha-MSH affect morphology, tyrosinase activity and melanogenesis in cultured human melanocytes. J Cell Sci 107, 205–11.
Kameyama K, Sakai C, Kuge S, Nishiyama S, Tomita Y, Ito S, Wakamatsu K, and Hearing VJ (1995) The expression of tyrosinase, tyrosinase-related proteins 1 and 2 (TRP1 and TRP2), the silver protein, and a melanogenic inhibitor in human melanoma cells of differing melanogenic activities. Pigment Cell Res 8, 97–104.
Kligman LH (1996) The hairless mouse model for photoaging. Clin Dermatol 14, 183–95.
Kobayashi T, Urabe K, Winder A, Jiménez-Cervantes C, Imokawa G, Brewington T, Solano F, García-Borrón JC, and Hearing VJ (1994) Tyrosinase related protein 1 (TRP1) functions as a DHICA oxidase in melanin biosynthesis. EMBO J 13, 5818–25.
Mishima Y, Hatta S, Ohyama Y, and Inazu M (1988) Induction of melanogenesis suppression: cellular pharmacology and mode of differential action. Pigment Cell Res 1, 367–74.
Mohamed SM, Sayed ET, Irmgard M, and Mahmoud AMN (1999) Polyphenolic metabolites of Rhamnus disperma. Phytochemistry 52, 943–6.
Nielsen AH, Olsen CE, and Moller BL (2005) Flavonoids in flowers of 16 Kalanchoe blossfeldiana varieties. Phytochemistry 66, 2829–35.
Yao C, Oh JH, Oh IG, Park CH, and Chung JH (2013) [6]-Shogaol inhibits melanogenesis in B16 mouse melanoma cells through activation of the ERK pathway. Acta Pharmacol Sin 34, 289–94.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, JY. Down-regulation of MITF, TRP-1, TRP-2, and tyrosinase expressions by compounds isolated from Pruni persicae Flos in murine B16F10 melanoma. J Korean Soc Appl Biol Chem 57, 827–834 (2014). https://doi.org/10.1007/s13765-014-4288-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13765-014-4288-5