Abstract
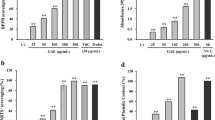
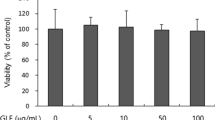
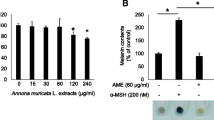
We evaluated the antioxidant activity and anti-melanogenic effects of Oenothera laciniata methanol extract (OLME) in vitro by using melan-a cells. The total polyphenol and flavonoid content of OLME was 66.3 and 19.0 mg/g, respectively. The electron-donating ability, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) radical-scavenging activity, and superoxide dismutase (SOD)-like activity of OLME (500 µg/mL) were 94.5%, 95.6%, and 63.6%, respectively. OLME and arbutin treatment at 50 µg/ mL significantly decreased melanin content by 35.5% and 14.2%, respectively, compared to control (p < 0.05). OLME and arbutin treatment at 50 µg/mL significantly inhibited intra-cellular tyrosinase activity by 22.6% and 12.6%, respectively, compared to control (p < 0.05). OLME (50 µg/mL) significantly decreased tyrosinase, tyrosinase-related protein-1 (TRP-1), TRP-2, and microphthalmia-associated transcription factor-M (MITF-M) mRNA expression by 57.1%, 67.3%, 99.0%, and 77.0%, respectively, compared to control (p < 0.05). Arbutin (50 µg/mL) significantly decreased tyrosinase, TRP-1, and TRP-2 mRNA expression by 24.2%, 42.9%, and 48.5%, respectively, compared to control (p < 0.05). However, arbutin (50 µg/mL) did not affect MITF-M mRNA expression. Taken together, OLME showed a good antioxidant activity and anti-melanogenic effect in melan-a cells that was superior to that of arbutin, a well-known skin-whitening agent. The potential mechanism underlying the anti-melanogenic effect of OLME was inhibition of tyrosinase activity and down-regulation of tyrosinase, TRP-1, TRP-2, and MITF-M mRNA expression.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kang, H.Y., Yoon, T.J. and Lee, G.J. (2011) Whitening effects of marine Pesudomonas extract. Ann. Dermatol., 23, 144–149.
Hearing, V.J. (2005) Biogenesis of pigment granules: a sensitive way to regulatemelanocyte function. J. Dermatol. Sci., 37, 3–14.
Huang, Y.H., Lee, T.H., Chan, K.J., Hsu, F.L., Wu, Y.C. and Lee, M.H. (2008) Anemonin is a natural bioactive compound that can regulate tyrosinase-related proteins and mRNA in human melanocytes. J. Dermatol. Sci., 49, 115–123.
Sturm, R.A., Teasdale, R.D. and Box, N.F. (2001) Human pigmentation genes: identification, structure and consequences of polymorphic variation. Gene, 277, 49–62.
Mallick, S., Singh, S.K., Sarkar, C., Saha, B. and Bhadra, R. (2005) Human placental lipid induces melanogenesis by increasing the expression of tyrosinase and itsrelated proteins in vitro. Pigment Cell Res., 18, 25–33.
Udono, T., Yasumoto, K., Takeda, K., Amae, S., Watanabe, K., Saito, H., Fuse, N., Tachibana, M., Takahashi, K., Tamai, M. and Shibahara, S. (2000) Structural organization of the human microphthalmia-associated transcription factor gene containingfour alternative promoters. Biochim. Biophys. Acta, 1491, 205–219.
Fuse, N., Yasumoto, K., Suzuki, H., Takahashi, K. and Shibahara, S. (1996) Identificationof a melanocyte-type promoter of the microphthalmia-associated transcription factor gene. Biochem. Biophys. Res. Commun., 219, 702–707.
Draelos, Z.D. (2007) Skin lightening preparations and the hydroquinone controversy. Dermatol. Ther., 20, 308–313.
Lim, J.T. (1999) Treatment of melasma using kojic acid in a gel containing hydroquinone and glycolic acid. Dermatol. Surg., 25, 282–284.
Varro, E.T., Lynn, R.B. and James, E.R. (1988) Pharmacognosy (9th edition), Lea and Febriger, Philadelphia, p. 471.
Hatano, T., Yasuhara, T., Fukuda, T., Noro, T. and Okuda, T. (1989) Phenolic constituents of licorice. II: Structures of licopyranocoumarin, licoarylcoumarin and glisoflavone, and inhibitory effects of licorice phenolics on xanthine oxidase. Chem. Pharm. Bull., 37, 3005–3009.
Kim, J.Y., Lee, J.A. and Park, S.Y. (2007) Antibacterial activities of Oenothera laciniata extracts. J. Korean Soc. Food Sci. Nutr., 36, 255–259.
Lee, J.A., Kim, J.Y., Yoon, W.J., Oh, D.J., Jung, Y.H., Lee, W.J. and Park, S.Y. (2006) Biological activities of Oenothera laciniata extracts (Onagraceae, Myrtales). Korean J. Food Sci. Technol., 38, 810–815.
Kim, S.E., Lee, C.M. and Kim Y.C. (2016) Anti-wrinkle efficacy of Oenothera laciniata methanol extract in human dermal fibroblasts. J. Invest. Cosmetol., 12, 197–203.
Folin, O. and Denis, W. (1912) On phosphotungstic-phosphomolybdic compounds as color reagents. J. Biol. Chem., 12, 239–243.
Davies, R., Massey, R.C. and Mcweeny, D.J. (1980) The catalisis of the N-nitrosation of secondary amines by nitrosophenols. Food Chem., 6, 115–122.
Blois, M.S. (1958) Antioxidant determinations by the use of a stable freeradical. Nature, 181, 1199–1200.
Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M. and Rice-Evans, C. (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med., 26, 1231–1237.
Marklund, S. and Marklund, G. (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem., 47, 469–474.
Rice-Evans, C.A., Miller, N.J., Bolwell, P.G., Bramley, P.M. and Pridham, J.B. (1995) The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic. Res., 22, 375–383.
Boudet, A.M. (2007) Evolution and current status of research in phenolic compounds. Phytochemistry, 68, 2722–2735.
Seyoum, A., Asres, K. and El-Fiky, F.K. (2006) Structure-radical scavenging activity relationships of flavonoids. Phytochemistry, 67, 2058–2070.
Bondet, V., Brand-Williams, W. and Berset, C. (1997) Kinetics and mechanisms of antioxidant activity using the DPPH•free radical method. Food Sci. Technol., 30, 609–615.
Kondo, S. (2000) The roles of cytokines in photoaging. J. Dermatol. Sci., 23, S30–S36.
Kang, H.Y., Yoon, T.J. and Lee, G.J. (2011) Whitening effects of marine Pesudomonas extract. Ann. Dermatol., 23, 144–149.
Chae, G.Y. and Ha, B.J. (2011) The comparative evaluation of fermented and non-fermented soybean extract on antioxidation and whitening. Toxicol. Res., 27, 205–209.
Park, H.Y., Kosmadaki, M., Yaar, M. and Gilchrest, B.A. (2009) Cellularmechanisms regulating human melanogenesis. Cell. Mol. Life Sci., 66, 1493–1506.
Kwon, B.S., Haq, A.K., Pomerantz, S.H. and Halaban, R. (1987) Isolation and sequence of a cDNA clone for human tyrosinase that maps at the mouse c-albino locus. Proc. Natl. Acad. Sci. U.S.A., 84, 7473–7477.
Schallreuter, K.U., Kothari, S., Chavan, B. and Spencer, J.D. (2008) Regulation of melanogenesis-controversies and new concepts. Exp. Dermatol., 17, 395–404.
del Marmol, V. and Beermann, F. (1996) Tyrosinase and related proteins in mammalian pigmentation. FEBS Lett., 381, 165–168.
Boissy, R.E., Zhao, H., Oetting, W.S., Austin, L.M., Wildenberg, S.C., Boissy, Y.L., Zhao, Y., Sturm, R.A., Hearing, V.J., King, R.A. and Nordlund, J.J. (1996) Mutation in and lack of expression of tyrosinase-related protein-1(TRP-1) in melanocytes from an individual with brown oculocutaneous albinism: a new subtype of albinism classified as “OCA3”. Am. J. Hum. Genet., 58, 1145–1156.
Levy, C., Khaled, M. and Fisher, D.E. (2006) MITF: master-regulator of melanocyte development and melanoma oncogene. Trends Mol. Med., 12, 406–414.
Park, H.Y., Wu, C., Yonemoto, L., Murphy-Smith, M., Wu, H., Stachur, C.M. and Gilchrest, B.A. (2006) MITF mediates cAMP-induced protein kinase C-β expression in human melanocytes. Biochem. J., 395, 571–578.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This is an Open-Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://doi.org/creativecommons.org/licenses/by/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Kim, S.E., Lee, C.M. & Kim, Y.C. Anti-Melanogenic Effect of Oenothera laciniata Methanol Extract in Melan-a Cells. Toxicol Res. 33, 55–62 (2017). https://doi.org/10.5487/TR.2017.33.1.055
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2017.33.1.055