Abstract
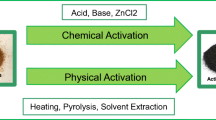
Phenolic pollutants are very toxic and their removal from aquatic resources is very important. Adsorption by activated carbon, AC, is the best method for removal of phenols from solutions. However, the high cost of AC and difficulty of its regeneration after phenol adsorption puts high demand on low price AC materials. Therefore, sugarcane bagasse as a sustainable, bulky and fibrous biomass was selected for the purpose of low-price AC formation for phenols adsorption. Sugarcane bagasse derived activated carbon, BAC, was achieved via an environmental thermo-chemical activation process using ZnCl2 followed by pyrolysis at different temperatures (400–600 °C). The formed BAC materials were characterized by elemental analysis, simultaneous TGA–DTA, ATR-FTIR, XRD, Raman spectroscopy, SEM, and nitrogen adsorption/desorption techniques. The BAC materials showed several enhanced characteristics including extra high specific surface area (up to 2046 m2/g), improved meso-/microporosity dual system and nanostructured graphitic-like structure composed of few graphene layers. Adsorption removal of phenol as an industrial waste pollutant was investigated from solutions of wide range of concentrations (50–1000 mg/L). The adsorption processes were characterized by (L2) class of adsorption isotherm, Langmuir isotherm model, physical-adsorption thermochemical parameters and pseudo-second-order kinetics. Adsorptions of two other substituted phenols (resorcinol and pyrogallol) were investigated. The adsorption capacity was increased with increasing of intramolecular bonding of the adsorbate in the order of phenol < resorcinol < pyrogallol. The present results emphasized the versatility of the formed BACs as environmentally sustainable adsorbent for phenolic pollutants.










Similar content being viewed by others
References
Abo ELNaga AO, El Saied M, Shaban SA, El Kady FY (2019) Fast removal of diclofenac sodium from aqueous solution using sugar cane bagasse-derived activated carbon. J Mol Liquids 285:9–19. https://doi.org/10.1016/j.molliq.2019.04.062
Açıkyıldız M, Gürses A, Karaca S (2014) Preparation and characterization of activated carbon from plant wastes with chemical activation. Microporous Mesoporous Mater 198:45–49
Adinaveen T, Kennedy LJ, Vijaya JJ, Sekaran G (2013) Studies on structural, morphological, electrical and electrochemical properties of activated carbon prepared from sugarcane bagasse. J Ind Eng Chem 19:1470–1476. https://doi.org/10.1016/j.jiec.2013.01.010
Al-Malack MH, Dauda M (2017) Competitive adsorption of cadmium and phenol on activated carbon produced from municipal sludge. J Environ Chem Eng 5:2718–2729
Angin D (2014) Production and characterization of activated carbon from sour cherry stones by zinc chloride. Fuel 115:804–811
Bernal V, Giraldo L, Moreno-Piraján JC (2020) Adsorption of pharmaceutical aromatic pollutants on heat-treated activated carbons: effect of carbonaceous structure and the adsorbent-adsorbate interactions. ACS Omega 5:15247–15256. https://doi.org/10.1021/acsomega.0c01288
Boontham W, Habaki H, Egashira R (2020) Removal of phenol from oil mill effluent using activated carbon prepared from Kernel Shell in Thailand’s Palm Industry. J Chem Eng Jpn 53:682–688
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. Am Chem Soc 60(1):309–319
Cai Y, Liu L, Tian H, Yang Z, Luo X (2019) Adsorption and desorption performance and mechanism of tetracycline hydrochloride by activated carbon-based adsorbents derived from sugar cane bagasse activated with ZnCl2. Molecules 24:4534. https://doi.org/10.3390/molecules24244534
Chai X, He H, Fan H, Kang X, Song X (2019) A hydrothermal-carbonization process for simultaneously production of sugars, graphene quantum dots, and porous carbon from sugarcane bagasse. Bioresour Technol 282:142–147. https://doi.org/10.1016/j.biortech.2019.02.126
Chen B, Chen Z (2009) Sorption of naphthalene and 1-naphthol by biochars of orange peels with different pyrolytic temperatures. Chemosphere 76:127–133. https://doi.org/10.1016/j.chemosphere.2009.02.004
Coates J (2000) Interpretation of infrared spectra, a practical approach. Encycl Anal Chem. https://doi.org/10.1002/9780470027318
Congsomjit D, Areeprasert C (2020) Hydrochar-derived activated carbon from sugar cane bagasse employing hydrothermal carbonization and steam activation for syrup decolorization. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-020-00635-y
Dabrowski A, Podkoscielny P, Hubicki Z, Barczak M (2005) Adsorption of phenolic compounds by activated carbon-acritical review. Chemosphere 58(8):1049–1070
Derylo-marczewska A, Buczek B, Swiatkowski A (2011) Effect of oxygen surface groups on adsorption of benzene derivatives from aqueous solutions onto active carbon samples. Appl Surf Sci 257:9466–9472. https://doi.org/10.1016/j.apsusc.2011.06.036
Dubinin MM, Radushkevich LV (1947) Equation of the characteristic curve of activated charcoal. Proc Acad Sci USSR Phys Chem Sect 55:331
Eslami A, Borghei SM, Rashidi A, Takdastan A (2018) Preparation of activated carbon dots from sugarcane bagasse for naphthalene removal from aqueous solutions. Sep Sci Technol 53:2536–2549. https://doi.org/10.1080/01496395.2018.1462832
Ferrari AC (2007) Raman spectroscopy of graphene and graphite: disorder, electron–phonon coupling, doping and nonadiabatic effects. Solid State Commun 143:47–57
Freundlich H (1909) einedarstellung der chemie der kolloide und verwantergebiete. Kapillarchemie. AcademisheBibliotek, Leipzig
Ge X, Wu Z, Manzoli M, Wu Z, Cravotto G (2020) Feasibility and the mechanism of desorption of phenolic compounds from activated carbons. Ind Eng Chem Res 59:12223–12231
Giles CH, D’Silva AP, Easton IA (1974a) A general treatment and classification of the solute adsorption isotherm, II. Experimental interpretation. J Colloid Interface Sci 47:766–778
Giles CH, Smith D, Huitson A (1974b) A general treatment and classification of the solute adsorption isotherm. I. Theoretical. J Colloid Interface Sci 47:755–765
Girods P, Dufour A, Fierro V, Rogaume Y, Rogaume C, Zoulalian A, Celzard A (2009) Activated carbons prepared from wood particleboard wastes: characterisation and phenol adsorption capacities. J Hazard Mater 166:491–501
Gonçalves R, Martins C, Mendes N, Farias L, Sousa D, Cristina R et al (2014) Preparation of activated carbons from cocoa shells and siriguela seeds using H 3 PO 4 and ZnCL 2 as activating agents for BSA and α -lactalbumin adsorption. Fuel Process Technol 126(2014):476–486. https://doi.org/10.1016/j.fuproc.2014.06.001
Greish AA, Sokolovskiy PV, Finashina ED, Kustov LM, Vezentsev AI, Chien ND (2021) Adsorption of phenol and 2,4-dichlorophenol on carbon-containing sorbent produced from sugar cane bagasse. Mendeleev Commun 31(1):121–122. https://doi.org/10.1016/j.mencom.2021.01.038
Gundogdu A, Duran C, Senturk HB, Soylak M, Ozdes D, Serencam H, Imamoglu M (2012) Adsorption of phenol from aqueous solution on a low-cost activated carbon produced from tea industry waste: equilibrium, kinetic, and thermodynamic study. J Chem Eng Data 57:2733–2743. https://doi.org/10.1021/je300597u
Halsey G (1948) Physical adsorption on non-uniform surfaces. J Chem Phys 16(10):931
Hameed BH, Rahman AA (2008) Removal of phenol from aqueous solutions by adsorption onto activated carbon prepared from biomass material. J Hazard Mater 160:576–581
Hazzaa R, Hussein M (2015) Adsorption of cationic dye from aqueous solution onto activated carbon prepared from olive stones. Environ Technol Innov 4:36–51
Karri RR, Sahub JN, Jayakumar NS (2017a) Optimal isotherm parameters for phenol adsorption from aqueous solutions onto coconut shell based activated carbon: error analysis of linear and non-linear methods. J Taiwan Inst Chem Eng 80(2017):472–487
Karri RR, Jayakumar NS, Sahu JN (2017b) Modelling of fluidised-bed reactor by differential evolution optimization for phenol removal using coconut shells based activated carbon. J Mol Liq 231:249–262
Karri RR, Sahu JN, Meikap BC (2020) Improving efficacy of Cr (VI) adsorption process on sustainable adsorbent derived from waste biomass (sugarcane bagasse) with help of ant colony optimization. Ind Crops Product 143:111927 https://doi.org/10.1016/j.indcrop.2019.111927
Khalil KMS, Allam OAS, Khairy M, Mohamed KMH, Elkhatib RM, Hamed MA (2017) High surface area nanostructured activated carbons derived from sustainable sorghum stalk. J Mol Liq 247:386–396
Khenniche L, Benissad-Aissani F (2010) Adsorptive removal of phenol by coffee residue activated carbon and commercial activated carbon: equilibrium, kinetics, and thermodynamics. J Chem Eng Data 55(11):4677–4686. https://doi.org/10.1021/je100302e
Khraisheh M, Al-Ghouti MA, AlMomani F (2020) P. putida as biosorbent for the remediation of cobalt and phenol from industrial waste waste waters. Environ Technol Innov 20:101148. https://doi.org/10.1016/j.eti.2020.101148
Kumar A, Jena MH (2016) Removal of methylene blue and phenol onto prepared activated carbon from Fox nutshell by chemical activation in batch and fixed-bed column. J Clean Prod 137:1246–1259
Langmuir I (1918) The adsorption of gases on plane surface of glass, mica and olatinum. J Am Chem Soc 40:1361–1403. https://doi.org/10.1021/ja02242a004
Liou T (2010) Development of mesoporous structure and high adsorption capacity of biomass-based activated carbon by phosphoric acid and zinc chloride activation. Chem Eng J 158:129–142. https://doi.org/10.1016/j.cej.2009.12.016
Long S, Du Q, Wang S, Tang P, Li D (2019) Graphene two-dimensional crystal prepared from cellulose two-dimensional crystal hydrolysed from sustainable biomass sugarcane bagasse. J Clean Prod 241:118209. https://doi.org/10.1016/j.jclepro.2019.118209
Lorenc-Grabowska E, Diez MA, Gryglewicz G (2016) Influence of pore size distribution on the adsorption of phenol on PET-based activated carbons. J Colloid Interface Sci 469:205–212
Luo X (2019) Cr (VI) adsorption performance and mechanism of an effective activated carbon prepared from bagasse with a one-step pyrolysis and ZnCl2 activation method. Cellulose 26:4921–4934. https://doi.org/10.1007/s10570-019-02418-9
Magne P, Walker PL Jr (1986) Phenol adsorption on activated carbons: application to the regeneration of activated carbons polluted with phenol. Carbon 24(1986):101–107
Mattson JS, Mark HB Jr, Malbin MD, Weber WJ Jr, Crittenden JC (1969) Surface chemistry of active carbon: specific adsorption of phenols. J Colloid Interface Sci 31:116–130
Michailof C, Stavropoulos GG, Panayiotou C (2008) Enhanced adsorption of phenolic compounds, commonly encountered in olive mill wastewaters, on olive husk derived activated carbons. Bioresour Technol 99(14):6400–6408
Mohammed NAS, Abu-Zurayk RA, Hamadneh I, Al-Dujaili AH (2018) Phenol adsorption on biochar prepared from the pine fruit shells: equilibrium, kinetic and thermodynamics studies. J Environ Manag 226:377–385
Okolo B, Park C, Keane MA (2000) Interaction of phenol and chlorophenols with activated carbon and synthetic zeolites in aqueous media. J Colloid Interface Sci 226:308. https://doi.org/10.1006/jcis.2000.6796
Pezoti O, Cazetta AL, Souza IP, Bedin KC, Martins AC, Silva TL, Almeida VC (2014) Adsorption studies of methylene blue onto ZnCl2-activated carbon produced from buriti shells (Mauritia flexuosa L.). Ind Eng Chem 20:4401–4407
Priya DS, Sureshkumar MV (2020) Synthesis of Borassus flabellifer fruit husk activated carbon flter for phenol removal from wastewater. Int J Environ Sci Technol 17:829–842. https://doi.org/10.1007/s13762-019-02325-3
Rambabu N, Rao BVSK, Surisetty VR, Das U, Dalai AK (2014) Production, characterization, and evaluation of activated carbons from de-oiled canola meal for environmental applications. Ind Crops Prod 65:572–581
Rangabhashiyam S, Anu N, Giri Nandagopal MS, Selvaraju N (2014) Relevance of isotherm models in biosorption of pollutants by agricultural, byproducts. J Environ Chem Eng 2:398–414
Shi J, Wang Y, Du W, Hou Z (2016) Synthesis of graphene encapsulated Fe3C in carbon nanotubes from biomass and its catalysis application. Carbon 99:330–337
Sing KSW, Everett DH, Haul RAW, Moscou L, Pierotti RA, Rouquerol J, Siemieniewska T (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl Chem 57(4):603–619
Solís-fuentes JA, Galán-méndez F, Hernández-medel MR, García-gómez RS, Bernal-gonzález M, Mendoza-pérez S et al (2019) Effectiveness of bagasse activated carbon in raw cane juice clarification. Food Biosci 32:100437. https://doi.org/10.1016/j.fbio.2019.100437
Srivastava VC, Swamy MM, Mall I, Prasad B, Mishra I (2006) Adsorptive removal of phenol by bagasse fly ash and activated carbon: equilibrium, kinetics and thermodynamics. Colloids Surf 272:89–104
Strachowski P, Bystrzejewski M (2015) Comparative studies of sorption of phenolic compounds onto carbon-encapsulated iron nanoparticles, carbon nanotubes and activated carbon. Colloids Surf A 467:113–123. https://doi.org/10.1016/j.colsurfa.2014.11.044
Temkin MJ, Phyzev V (1940) Recent modifications to Langmuir isotherms. Acta Physiochim USSR 12:217–222
Velasco LF, Carmona RJ, Matos J, Ania CO (2014) Performance of activated carbons in consecutive phenol photooxidation cycles. Carbon 73:206–215
Villegas LGC, Mashhadi N, Chen M, Mukherjee D, Taylor KE, Biswas N (2016) A short review of techniques for phenol removal from wastewater. Curr Pollut Rep. https://doi.org/10.1007/s40726-016-0035-3
Wang Y, Su F, Wood CD, Lee JY, Zhao XS (2008) Preparation and characterization of carbon nanospheres as anode materials in lithium-ion secondary batteries. Ind Eng Chem Res 47:2294–2300
Wassie AB, Srivastava VC (2016) (2016) Chemical treatment of teff straw by sodium hydroxide, phosphoric acid and zinc chloride: adsorptive removal of chromium. Int J Environ Sci Technol 13:2415–2426. https://doi.org/10.1007/s13762-016-1080-6
Yagmur E, Turkoglu S, Banford A, Aktas Z (2017) The relative performance of microwave regenerated activated carbons on the removal of phenolic pollutants. J Clean Prod 149:1109–1117
Yang T, Lua AC (2006) Textural and chemical properties of zinc chloride activated carbons prepared from pistachio-nut shells. Mater Chem Phys 100:438–444
Yang J, Qiu K (2010) Preparation of activated carbons from walnut shells via vacuum chemical activation and their application for methylene blue removal. Chem Eng J 165:209–217
Zhu G, Deng X, Hou M, Sun K, Zhang Y, Li P, Liang F (2016) Comparative study on characterization and adsorption properties of activated carbons by phosphoric acid activation from corncob and its acid and alkaline hydrolysis residues. Fuel Process Technol 144:255–261
Acknowledgements
The authors are greatly acknowledged the fund received during this project sponsored by STDF (Egypt). Grant number: TC Water ID 20709, PI: Prof. Kamal M.S. Khalil.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: Binbin Huang.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khalil, K.M.S., Khairy, M., Allam, O.A.S. et al. Formation of improved activated carbons from sugarcane bagasse as environmental materials for adsorption of phenolic pollutants. Int. J. Environ. Sci. Technol. 19, 3103–3116 (2022). https://doi.org/10.1007/s13762-021-03382-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-021-03382-3