Abstract
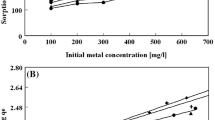
A granular composite material (GCM) consisting of beta-zeolite-/powder activated carbon (PAC), organic binder (MC) and inorganic binder (bentonite) was prepared from powdered samples. GCMs were prepared successfully at a constant concentration of MC (10 wt%) and bentonite (30 wt%) and at various ratios of β-zeolite/PAC (0.05–0.5). The results indicated that the GCMs had high iodine values (981.9–1170.1 mg/g) and suitable mechanical strength (180.55–323.86 N). A series of batch sorption experiments for removal of Zn2+, Cd2+, Cu2+ and Pb2+ from aqueous solutions were performed with two selected composites of GCM2 (β-zeolite/PAC ratio = 0.05) and GCM9 (β-zeolite/PAC ratio = 0.4) under various conditions (contact time and pH). Maximum sorptions of Zn, Cd and Pb at pH 5.5 and that of Cu at pH 6 were attained at equilibrium time of 80 min and initial metal ions concentration of 20 mg/L. The results showed that the sorption of Zn2+, Cu2+, Cd2+ and Pb2+ by GCMs increased by increasing the β-zeolite/PAC ratio. The maximum sorption occurred at the β-zeolite/PAC ratio of 0.4. The maximum sorption capacity order of GCM2 in unit of mg g−1 was: Pb2+ (13.684) > Cu2+ (13.300) > Cd2+ (12.717) > Zn2+ (12.590). The maximum sorption capacity order of GCM9 in unit of mg g−1 was: Pb2+ (15.983) > Cu2+ (15.066) > Cd2+ (14.760) > Zn2+ (14.481). Three equations of Morris–Weber, pseudo-first order and pseudo-second order were investigated to study the kinetics of sorption process. The kinetic data showed that sorption of heavy metal ions was well described by pseudo-second-order equation.






Similar content being viewed by others
References
Ali I, Alothman ZA, Al-Warthan A (2015) Sorption, kinetics and thermodynamics studies of atrazine herbicide removal from water using iron nano-composite material. Int J Environ Sci Technol 13:733–742. https://doi.org/10.1007/s13762-015-0919-6
Alslaibi TM, Abustan I, Ahmad MA, Abu Foul AA (2013) Application of response surface methodology (RSM) for optimization of Cu, Cd, Ni, Pb, Fe, and Zn removal from aqueous solution using microwaved olive stone activated carbon. J Chem Technol Biotechnol 88:2141–2151. https://doi.org/10.1002/jctb.4073
Alslaibi TM, Abustan I, Ahmad MA, Abu Foul AA (2014a) Comparison of activated carbon prepared from olive stones by microwave and conventional heating for iron (II), lead(II), and copper(II) removal from synthetic wastewater. Environ Prog Sustain Energy 33:1074–1085. https://doi.org/10.1002/ep.11877
Alslaibi TM, Abustan I, Ahmad MA, Abu Foul AA (2014b) Preparation of activated carbon from olive stone waste: optimization study on the removal of Cu, Cd, Ni, Pb, Fe, and Zn from aqueous solution using response surface methodology. J Dispers Sci Technol 35:913–925. https://doi.org/10.1080/01932691.2013.809506
Apiratikul R, Pavasant P (2008) Sorption of Cu, Cd, and Pb using modified zeolite from coal fly ash. J Chem Eng 144:245–258. https://doi.org/10.1016/j.cej.2008.01.038
Babic B, Kokunesoski M, Gulicovski J, Prekajski M, Pantic J, Radosavljevic-Mihajlovic A, Matovic B (2011) Synthesis and characterization of carbon cryogel/zeolite composites. J Process Appl Ceram 5:91–96. https://doi.org/10.2298/PAC1102091B
Bunhu T, Tichagwa L, Chaukura N (2017) Competitive sorption of Cd and Pb from a binary aqueous solution by poly (methyl methacrylate)-grafted montmorillonite clay nanocomposite. Appl Water Sci 7:2287–2295. https://doi.org/10.1007/s13201-016-0404-5
Chen JP, Wu S, Chong KH (2003) Surface modification of a granular activated carbon by citric acid for enhancement of copper sorption. Carbon 41:1979–1986. https://doi.org/10.1016/S0008-6223(03)00197-0
Esfandian H, Samadi-Maybodi A, Parvini M, Khoshandam B (2016) Development of a novel method for the removal of diazinon pesticide from aqueous solution and modeling by artificial neural networks (ANN). J Ind Eng Chem 35:295–308. https://doi.org/10.1016/j.jiec.2016.01.011
Foo KY, Hameed BH (2011) The environmental applications of activated carbon/zeolite composite materials. J Adv Colloid Interface Sci 162:22–28. https://doi.org/10.1016/j.cis.2010.09.003jhk
Gadhave A, Waghmare J (2014) Removal of heavy metal ions from wastewater by carbon nanotubes. Int J Chem Sci Appl 5:56–67
Ghasemi M, Ghasemi N, Zahedi G, Alwi SRW, Goodarzi M, Javadian H (2014) Kinetic and equilibrium study of Ni(II) sorption from aqueous solutions onto Peganum harmala-L. Int J Environ Sci Technol 11:1835–1844. https://doi.org/10.1007/s13762-014-0617-9
Ghasemian-Lemraski E, Sharafinia S (2016) Kinetics, equilibrium and thermodynamics studies of Pb2+ adsorption onto new activated carbon prepared from Persian mesquite grain. J Mol Liq 219:482–492. https://doi.org/10.1016/j.molliq.2016.03.031
Gwenzi W, Musarurwa T, Nyamugafata P, Chaukura N, Chaparadza A, Mbera S (2014) Adsorption of Zn2+ and Ni2+ in a binary aqueous solution by biosorbents derived from sawdust and water hyacinth (Eichhornia crassipes). Water Sci Technol 70(8):1419–1427. https://doi.org/10.2166/wst.2014.391
Jha VK, Matsuda M, Miyake M (2008) Sorption properties of the activated carbon-zeolite composite prepared from coal fly ash for Ni2+, Cu2+, Cd2+ and Pb2+. J Hazard Mater 160:148–153. https://doi.org/10.1016/j.jhazmat.2008.02.107
Komkiene J, Baltrenaite E (2015) Biochar as adsorbent for removal of heavy metal ions [Cadmium(II), Copper(II), Lead(II), Zinc(II)] from aqueous phase. Int J Environ Sci Technol 13:471–482. https://doi.org/10.1007/s13762-015-0873-3
Lee JS, Kim JH, Kim JT, Suh JK, Lee JM, Lee CH (2002) Adsorption equilibria of CO2 on zeolite 13X and zeolite X/activated carbon composite. J Chem Eng Data 47:1237–1242. https://doi.org/10.1021/je020050e
Liang C, Lee PH (2012) Granular activated carbon/pyrite composites for environmental application: synthesis and characterization. J Hazard Mater 231–232:120–126. https://doi.org/10.1016/j.jhazmat.2012.06.048
Ma J, Sun H, Su S, Cheng W, Li R (2008) A novel double-function porous material: zeolite-activated carbon extrudates from elutrilithe. J Porous Mater 15:289–294. https://doi.org/10.1007/s10934-006-9080-4
Ma J, Si C, Li Y, Li R (2012) CO2 adsorption on zeolite X/activated carbon composites. J Adsorpt 18:503–510. https://doi.org/10.1007/s10450-012-9440-0
Miyake M, Kimura Y, Ohashi T, Matsuda M (2008) Preparation of activated carbon–zeolite composite materials from coal fly ash. J Microporous Mesoporous Mater 112:170–177. https://doi.org/10.1016/j.micromeso.2007.09.028
Okada K, Shimizu YI, Kameshima Y, Nakajima A (2005) Preparation and properties of carbon/zeolite composites with corrugated structure. J Porous Mater 12:281–291. https://doi.org/10.1007/s10934-005-3126-x
Purna Chandra Rao G, Satyaveni S, Ramesh A, Seshaiah K, Murthy KSN, Choudary NV (2006) Sorption of cadmium and zinc from aqueous solutions by zeolite 4A, zeolite 13X and bentonite. J Environ Manage 81:265–272. https://doi.org/10.1016/j.jenvman.2005.11.003
Rivera-Utrilla J, Bautista-Toledo I, Ferro-Garcia MA, Moreno-Castilla C (2001) Activated carbon surface modifications by adsorption of bacteria and their effect on aqueous lead adsorption. J Chem Technol Biotechnol 76:1209–1215. https://doi.org/10.1002/jctb.506
Ruziwa D, Chaukura N, Gwenzi W, Pumure I (2015) Removal of Zn and Pb ions from aqueous solution using sulphonated waste polystyrene. J Environ Chem Eng 3:2528–2537. https://doi.org/10.1016/j.jece.2015.08.006
Saeidi N, Lotfollahi MN (2015a) Effects of powder activated carbon particle size on adsorption capacity and mechanical properties of the semi activated carbon fiber. J Fibers Poly 16:543–549. https://doi.org/10.1007/s12221-015-0543-6
Saeidi N, Lotfollahi MN (2015b) A procedure to form powder activated carbon into activated carbon monolith. Int J Adv Manuf Technol 81:1281–1288. https://doi.org/10.1007/s00170-015-7311-z
Saeidi N, Lotfollahi MN (2016) Effects of powder activated carbon particle size on activated carbon monoliths properties. J Mater Manuf Process 31:1634–1638. https://doi.org/10.1080/10426914.2015.1117630
Thamilarasu P, Karunakaran K (2011) Kinetic, equilibrium and thermodynamic studies on removal of Cr(VI) by activated carbon prepared from Ricinus communis seed shell. Can J Chem Eng 91:9–18. https://doi.org/10.1002/cjce.20675
Ucer A, Uyanik A, Aygun SF (2006) Adsorption of Cu(II), Cd(II), Zn(II), Mn(II) and Fe(III) ions by tannic acid immobilized activated carbon. Sep Purif Technol 47:113–118. https://doi.org/10.1016/j.seppur.2005.06.012
Zhu L, Ye S, Asghar A, Bang SH, Oh WC (2016) Additional effect of zeolite based on bactericidal activated carbon spheres with enhanced adsorption effect and higher ignition temperature. J Korean Ceram Soc 53:68–74. https://doi.org/10.4191/kcers.2016.53.1.68
Acknowledgements
The authors tend to appreciate the partial financial support from the research office of Semnan University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: BV Thomas.
Rights and permissions
About this article
Cite this article
Seyedein Ghannad, S.M.R., Lotfollahi, M.N. Preparation of granular composite materials as novel sorbents and their application for removal of heavy metals from solution. Int. J. Environ. Sci. Technol. 16, 3697–3706 (2019). https://doi.org/10.1007/s13762-018-1772-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-018-1772-1