Abstract
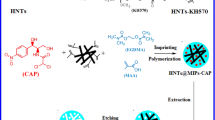
In this research, hydrophilic molecularly imprinted polymers (HMIPs) were synthesized through a facile one-step green precipitation polymerization in ethanol, which was non-toxic, green and environmentally friendly. HMIPs were tested by FT-IR, SEM, water contact angle studies and water dispersion stability test. HMIPs exhibited a good water-compatibility property. Compared with non-imprinted process, template molecule tetracycline (TC) had a significant influence on morphology of polymer particles. Batch binding experiments were carried out in water to evaluate adsorption equilibrium, kinetics and selectivity. Equilibrium adsorption amount increased with the increase of the temperature, and the data were well described by Freundlich model. Kinetics properties of HMIPs were better fitted to pseudo-second-order rate equations. The thermodynamic analysis indicated that the adsorption process of TC was spontaneous and endothermic. HMIPs exhibited the high affinity and selectivity toward TC over structurally analogous antibiotics than that of non-imprinted polymers. Also, the HMIPs had good reuse ability.








Similar content being viewed by others
References
A. Önal, Food Chem. 127, 197 (2011)
I. Yahiaoui, F. Aissani-Benissad, F. Fourcade, A. Amrane, Chem. Eng. J. 221, 418 (2013)
Y. Luo, L. Xu, M. Rysz, Y.Q. Wang, H. Zhang, P.J.J. Alvarez, Environ. Sci. Technol. 45, 1827 (2011)
Q.Q. Zhang, A. Jia, Y. Wan, H. Liu, K.P. Wang, H. Peng, Z.M. Dong, J.Y. Hu, Environ. Sci. Technol. 48, 14317 (2014)
M. Renkecz, G. Mistlberger, M. Pawlak, V. Horváth, E. Bakker, ACS Appl. Mater. Inter. 5, 8537 (2013)
J.E. Lofgreen, I.L. Moudrakovski, G.A. Ozin, ACS Nano 5, 2277 (2011)
X.F. Zhang, X.Z. Du, X. Huang, Z.P. Lv, J. Am. Chem. Soc. 135, 9248 (2013)
F.J. Ning, H.L. Peng, J.H. Li, L.X. Chen, H. Xiong, J. Agric, Food Chem. 62, 7436 (2014)
L.X. Chen, S.F. Xu, J.H. Li, Chem. Soc. Rev. 40, 2922 (2011)
Y. Li, M.J. Ding, S. Wang, R.Y. Wang, X.L. Wu, T.T. Wen, L.H. Yuan, P. Dai, Y.H. Lin, X.M. Zhou, A.C.S. Appl, Mater. Inter. 3, 3308 (2011)
H. Niu, Y.Q. Yang, H.Q. Zhang, Biosens. Bioelectron. 74, 440 (2015)
S.J. Li, Y. Ge, A. Tiwari, S. Wang, A.P.F. Turner, S.A. Piletsky, J. Catal. 278, 173 (2011)
L.G. Chen, J. Liu, Q.L. Zeng, H. Wang, A.M. Yu, H.Q. Zhang, L. Ding, J. Chromatogr. A 1216, 3710 (2009)
W.S. Cai, R.B. Gupta, Purif. Technol 35, 215 (2004)
J.D. Dai, Z.P. Zhou, X. Wei, J.M. Pan, T.B. Zou, C.X. Li, RSC Adv. 4, 7967 (2014)
J.D. Dai, Z.P. Zhou, C.Y. Zhao, X. Wei, X.H. Dai, L. Gao, Z.J. Cao, Y.Y. Yan. Ind. Eng. Chem. Res. 53, 7157 (2014)
J.D. Dai, J.M. Pan, L.C. Xu, X.X. Li, Z.P. Zhou, R.X. Zhang, Y.S. Yan, J. Hazard. Mater. 205–206, 179 (2012)
Y. Kanekiyo, R. Naganawa, H. Tao, Angew. Chem. Int. Ed. 42, 3014 (2003)
C. Gong, K.L. Wong, M.H.W. Lam, Chem. Mater. 20, 1353 (2008)
Z. Hua, Z. Chen, Y. Li, M. Zhao, Langmuir 24, 5773 (2008)
Z. Chen, L. Xu, Y. Liang, M. Zhao, Adv. Mater. 22, 1488 (2010)
K. Yoshimatsu, K. Reimhult, A. Krozer, K. Mosbach, K. Sode, L. Ye, Anal. Chim. Acta 584, 112 (2007)
S. Yang, H.R. Liu, Z.C. Zhang, J. Polym. Sci. Pol. Chem. 46, 3900 (2008)
N. Sharmin, R.A. Khan, S. Salmieri, D. Dussault, J. Bouchard, M. Lacroix, J. Agric. Food Chem. 60, 623 (2012)
C. Cacho, E. Turiel, A. Martin-Esteban, C. Perez-Conde, C. Camara, J. Chromatogr. B 802, 347 (2004)
G.Q. Pan, Y. Ma, Y. Zhang, X.Z. Guo, C.X. Li, H.Q. Zhang, Soft Matter 7, 8428 (2011)
G.Q. Pan, Y. Zhang, X.Z. Guo, C.X. Li, H.Q. Zhang, Biosens. Bioelectron. 26, 976 (2010)
M.H. Stenzel, Chem. Eng. Prog. 89, 36 (1993)
X.M. Peng, D.P. Huang, T. Odoom-Wubah, D.F. Fu, J.L. Huang, Q.D. Qin, J. Colloid Interface Sci. 430, 272 (2014)
A. Sari, D. Citak, M. Tuzen, Chem. Eng. J. 162, 521 (2010)
Y.S. Ho, G. McKay, Process Biochem. 34, 451 (1999)
Z.J. Wu, H. Joo, K. Lee, Chem. Eng. J. 112, 227 (2005)
X.Y. Yu, T. Luo, Y.X. Zhang, Y. Jia, B.J. Zhu, X.C. Fu, J.H. Liu, X.J. Huang, A.C.S. Appl, Mater. Interfaces 3, 2585 (2011)
W. Zhang, L. Qin, X.W. He, W.Y. Li, Y.K. Zhang, J. Chromatogr. A 1216, 4560 (2009)
Q. Yu, S.B. Deng, G. Yu, Water Res. 42, 3089 (2008)
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 21176107, 21174057, 21277063, 21446015 and U1407123), the National Basic Research Program of China (973 Program, 2012CB821500), Natural Science Foundation of Jiangsu Province (BK20140534), Innovation Programs Foundation of Jiangsu Province (Nos. SJLX_0481 and CXZZ13_0668), Research Fund for the Doctoral Program of Higher Education of China (20133227110022 and 20133227110010) and Jiangsu Planned Projects for Postdoctoral Research Funds (1102119C).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, J., Dai, J., Ma, W. et al. Surface hydrophilic imprinted particles via a green precipitation polymerization for selective removal of tetracycline from aqueous solution. J IRAN CHEM SOC 13, 489–497 (2016). https://doi.org/10.1007/s13738-015-0758-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-015-0758-x