Abstract
Purpose of Review
To review when to consider a diagnosis of cutaneous T-cell lymphoma (CTCL) in the inpatient setting and the diagnostic approach, as well as complications of disease and treatment that could lead to hospitalization.
Recent Findings
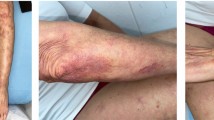
Cutaneous T-cell lymphomas (CTCLs) are a diverse group of non-Hodgkin lymphomas that are difficult to diagnosis as the initial presentation can mimic other benign inflammatory or infectious conditions of the skin. While the clinical course is typically indolent, with most cases normally diagnosed and managed in the outpatient settings, some CTCL-associated clinical scenarios, including infections, treatment-related toxicity/side effects, hemophagocytic lymphohistiocytosis (HLH), or spread to extra-cutaneous organs, warrant management in the inpatient setting.
Summary
Early involvement of dermatology specialists is critical for recognizing and diagnosing CTCL in a timely matter, managing treatments and their toxicities, and for improving both morbidity and mortality in patients.








Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Larocca C, Kupper T. Mycosis fungoides and Sezary syndrome: an update. Hematol Oncol Clin North Am. 2019;33(1):103–20.
• Kirsch IR, Watanabe R, O'Malley JT, et al. TCR sequencing facilitates diagnosis and identifies mature T cells as the cell of origin in CTCL. Sci Transl Med. 2015;7(308):308ra158. The study demonstrated the success of and introduces high-throughput TCR sequencing as an accurate diagnostic tool for CTCL in all stages and provided insights into the CTCL cell of origin and location.
Jackow CM, et al. Association of erythrodermic cutaneous T-cell lymphoma, superantigen-positive Staphylococcus aureus, and oligoclonal T-cell receptor V beta gene expansion. Blood. 1997;89(1):32–40.
van Doorn R, et al. Mycosis fungoides: disease evolution and prognosis of 309 Dutch patients. Arch Dermatol. 2000;136(4):504–10.
• Larocca CA, LeBoeuf NR. Overview of cutaneous T-cell lymphomas. Hematol Oncol Clin North Am. 2019;33(4):669–686. Literature review on mycosis fungoides and sézary syndrome.
Semaan S, et al. A clinician’s guide to cutaneous T-cell lymphoma presenting as recalcitrant eczematous dermatitis in adults. Int J Womens Dermatol. 2021;7(4):422–7.
Martinez-Escala ME, et al. Progression of undiagnosed cutaneous lymphoma after anti-tumor necrosis factor-alpha therapy. J Am Acad Dermatol. 2018;78(6):1068–76.
Marks J. Erythroderma and its management. Clin Exp Dermatol. 1982;7(4):415–22.
Andersen RM, Thyssen JP, Maibach HI. The role of wet wrap therapy in skin disorders - a literature review. Acta Derm Venereol. 2015;95(8):933–9.
Pielop JA, Jones D, Duvic M. Transient CD30+ nodal transformation of cutaneous T-cell lymphoma associated with cyclosporine treatment. Int J Dermatol. 2001;40(8):505–11.
Botella-Estrada R, et al. Erythroderma. A clinicopathological study of 56 cases. Arch Dermatol. 1994;130(12):1503–7.
Olsen EA, et al. Sezary syndrome: immunopathogenesis, literature review of therapeutic options, and recommendations for therapy by the United States Cutaneous Lymphoma Consortium (USCLC). J Am Acad Dermatol. 2011;64(2):352–404.
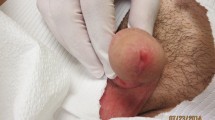
Axelrod PI, Lorber B, Vonderheid EC. Infections complicating mycosis fungoides and Sezary syndrome. JAMA. 1992;267(10):1354–8.
Tsambiras PE, et al. Infectious complications of cutaneous t-cell lymphoma. Cancer Control. 2001;8(2):185–8.
Tokura Y, et al. Cutaneous colonization with staphylococci influences the disease activity of Sezary syndrome: a potential role for bacterial superantigens. Br J Dermatol. 1995;133(1):6–12.
Tokura Y, et al. Stimulation of cutaneous T-cell lymphoma cells with superantigenic staphylococcal toxins. J Invest Dermatol. 1992;98(1):33–7.
Knaak C, et al. Treatment and mortality of hemophagocytic lymphohistiocytosis in adult critically Ill patients: a systematic review with pooled analysis. Crit Care Med. 2020;48(11):e1137–46.
Janka GE, Lehmberg K. Hemophagocytic lymphohistiocytosis: pathogenesis and treatment. Hematology Am Soc Hematol Educ Program. 2013;2013:605–11.
Mehta RS, Smith RE. Hemophagocytic lymphohistiocytosis (HLH): a review of literature. Med Oncol. 2013;30(4):740.
Filipovich A, McClain K, Grom A. Histiocytic disorders: recent insights into pathophysiology and practical guidelines. Biol Blood Marrow Transplant. 2010;16(1 Suppl):S82–9.
Fajgenbaum DC, June CH. Cytokine storm. N Engl J Med. 2020;383(23):2255–73.
Lee DE, et al. Hemophagocytic lymphohistiocytosis in cutaneous T-cell lymphoma. JAMA Dermatol. 2018;154(7):828–31.
Fardet L, et al. Development and validation of the HScore, a score for the diagnosis of reactive hemophagocytic syndrome. Arthritis Rheumatol. 2014;66(9):2613–20.
Henter JI, et al. HLH-2004: Diagnostic and therapeutic guidelines for hemophagocytic lymphohistiocytosis. Pediatr Blood Cancer. 2007;48(2):124–31.
Jordan MB, et al. How I treat hemophagocytic lymphohistiocytosis. Blood. 2011;118(15):4041–52.
Bergsten E, et al. Confirmed efficacy of etoposide and dexamethasone in HLH treatment: long-term results of the cooperative HLH-2004 study. Blood. 2017;130(25):2728–38.
Marsh RA, et al. Salvage therapy of refractory hemophagocytic lymphohistiocytosis with alemtuzumab. Pediatr Blood Cancer. 2013;60(1):101–9.
Maschalidi S, et al. Therapeutic effect of JAK1/2 blockade on the manifestations of hemophagocytic lymphohistiocytosis in mice. Blood. 2016;128(1):60–71.
Henter JI, et al. Hypercytokinemia in familial hemophagocytic lymphohistiocytosis. Blood. 1991;78(11):2918–22.
Yu JT, et al. Reduced intensity conditioning allogeneic hematopoietic stem cell transplant could be beneficial to angioimmunoblastic T-cell lymphoma patients with hemophagocytic lymphohistiocytosis. Ann Hematol. 2012;91(5):805–7.
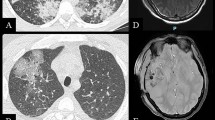
Baser S, et al. Pulmonary manifestations in patients with cutaneous T-cell lymphomas. Cancer. 2007;109(8):1550–5.
Stein M, et al. Central neurologic involvement in mycosis fungoides: ten cases, actuarial risk assessment, and predictive factors. Cancer J. 2006;12(1):55–62.
Zhao G, et al. Central nervous system involvement in cutaneous T-cell lymphoma: 2 illustrative cases and a review of current literature. Clin Lymphoma Myeloma Leuk. 2014;14(1):e25-30.
Vu BA, Duvic M. Central nervous system involvement in patients with mycosis fungoides and cutaneous large-cell transformation. J Am Acad Dermatol. 2008;59(2 Suppl 1):S16-22.
Duarte RF, et al. Long-term outcome of allogeneic hematopoietic cell transplantation for patients with mycosis fungoides and Sezary syndrome: a European society for blood and marrow transplantation lymphoma working party extended analysis. J Clin Oncol. 2014;32(29):3347–8.
Foss FM. DAB(389)IL-2 (ONTAK): a novel fusion toxin therapy for lymphoma. Clin Lymphoma 2000;1(2):110–6, discussion 117.
Ohmachi K, et al. E7777 in Japanese patients with relapsed/refractory peripheral and cutaneous T-cell lymphoma: a phase I study. Cancer Sci. 2018;109(3):794–802.
Kawai H, et al. Phase II study of E7777 in Japanese patients with relapsed/refractory peripheral and cutaneous T-cell lymphoma. Cancer Sci. 2021;112(6):2426–35.
Duvic M, et al. Phase 1/2 study of mogamulizumab, a defucosylated anti-CCR4 antibody, in previously treated patients with cutaneous T-cell lymphoma. Blood. 2015;125(12):1883–9.
Kim YH, et al. Mogamulizumab versus vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): an international, open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2018;19(9):1192–204.
Larocca C, Kupper TS, LeBoeuf NR. Mogamulizumab forecast: clearer patients, with a slight chance of immune mayhem. Clin Cancer Res. 2019;25(24):7272–4.
Chen L, et al. Mogamulizumab-associated cutaneous granulomatous drug eruption mimicking mycosis fungoides but possibly indicating durable clinical response. JAMA Dermatol. 2019;155(8):968–71.
Kwan JM, et al. Mogamulizumab-associated acute myocarditis in a patient with T-cell lymphoma. JACC Case Rep. 2021;3(7):1018–23.
Larocca CA, TM, LeBoeuf N, Virgin C, Goyal A, Amato A, De Girolami U, McGrath CKE, Mullally W, Sparks J, Fisher DC, Kupper TS. Successful treatment of mogamulizumab-induced myositis. USCLC annual meeting. 2021.
Roccuzzo G, et al. Immune check point inhibitors in primary cutaneous T-cell lymphomas: biologic rationale, clinical results and future perspectives. Front Oncol. 2021;11: 733770.
Lesokhin AM, et al. Nivolumab in patients with relapsed or refractory hematologic malignancy: preliminary results of a phase Ib study. J Clin Oncol. 2016;34(23):2698–704.
Khodadoust MS, et al. Pembrolizumab in relapsed and refractory mycosis fungoides and Sezary syndrome: a multicenter phase II study. J Clin Oncol. 2020;38(1):20–8.
•• Weng WK, Arai S, Rezvani A, et al. Nonmyeloablative allogeneic transplantation achieves clinical and molecular remission in cutaneous T-cell lymphoma. Blood Adv. 2020;4(18):4474–4482. Pivotal clinical trial that showed allogeneic stem cell transplant to be an effective treatment in cutaneous T-cell lymphoma.
Divito SJ, et al. Peripheral host T cells survive hematopoietic stem cell transplantation and promote graft-versus-host disease. J Clin Invest. 2020;130(9):4624–36.
Foo SH, et al. Unmasking mycosis fungoides/Sezary syndrome from preceding or co-existing benign inflammatory dermatoses requiring systemic therapies: patients frequently present with advanced disease and have an aggressive clinical course. Br J Dermatol. 2016;174(4):901–4.
Weenig RH, et al. Fatal cytotoxic cutaneous lymphoma presenting as ulcerative psoriasis. Arch Dermatol. 2009;145(7):801–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors do not have existing conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Hospital-Based Dermatology
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jakuboski, S., Doyle, T., Tawa, M. et al. Inpatient Considerations in the Diagnosis and Management of the Cutaneous T-Cell Lymphoma Patient. Curr Derm Rep 11, 233–243 (2022). https://doi.org/10.1007/s13671-022-00367-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13671-022-00367-4