Abstract
Key Message
The best yields in the extraction of hydroxycinnamic acids, flavanols and total phenolics from black cherry flowers were obtained with 40% ethanol and the poorest with water. A 30% higher content of total phenolics was obtained from leaves by using methanol rather than water.
Context
Black cherry (Prunus serotina Erhr.) is widespread in Europe. Because it is an invasive species, most research has been aimed at trying to find a method to remove this shrub from forests. The incentive for the development of the research was that we were trying to find a possible folk use of black cherry leaves and flowers.
Aims
The aim of this work was to study the detailed phenolic profile of extracts of P. serotina leaves and flowers.
Methods
Three types of extracts of P. serotina were made. The extracts were analysed for differences in phenolics content based on different extraction methods. HPLC-MSn was used to identify the phenolic compounds, and HPLC–DAD was used for their quantification.
Results
The results show that different extraction methods result in differences in the amount of extractables. Flavanols are the main group of identified compounds in both leaves and flowers. A larger extraction efficiency leads to a higher amount of phenolics in the flowers compared to leaves (49.8 vs. 36.5 g/kg dry weight). No difference was detected in the amount of phenolics between water extracts of leaves and flowers. Each extract can be considered rich in phenolics.
Conclusion
This work shows that leaves and flowers of P. serotina are a rich source of phenolic compounds. We provide interesting results that might be of interest in the industrial valuation of several compounds.
Similar content being viewed by others
1 Introduction
Black cherry (Prunus serotina Ehrh.) originates from North America and is also called American black cherry. It is a native tree from Mexico-Guatemala to south-east Canada, where it can grow up to 30 m, and is one of the most valued woods for furniture (Petitpierre et al. 2009). In Poland, it mostly grows as a shrub or in the form of small trees. P. serotina blooms intensively in late spring (end of May). Flowers have a characteristic almond-like odour. Every part of the plant has this smell, so this characteristic is used for distinguishing this species from other similar species (Nestby 2020).
Black cherry is an invasive species in Europe. Problems caused by black cherry have been studied in many European countries, e.g. Italy, Hungary, Belgium, Germany, the Netherlands, France and Norway (Deckers et al. 2005; Csiszár et al. 2013, Gentili et al. 2019, Godefroid et al. 2005, Poyet et al. 2014; Nestby 2020; Desiea et al. 2020). Researchers have pointed out that a high seed production is the reason for the invasive success of P. serotina, and this plus the early age of flowering has made black cherry a great pest in wood stands. Trees can produce fruits very early, when the plants are 3–4 years old. Additionally, it can also spread vegetatively (Motta et al. 2009; Kawaletz et al. 2014). The idea behind the introduction of black cherry into European forests was to find a plant for the understory of forests growing on poor soil (Grajewski et al. 2010). However, P. serotina grows on both types of soil (rich and poor), and it can grow more intensively when the soil is well fertilized. In such cases, black cherry may reach a dominant position in a forest (Motta et al. 2009; Grajewski et al. 2010; Kawaletz et al. 2014; Halarewicz and Pruchniewicz, 2015). Black cherry competes with native species of the understorey of Polish forests, supplanting, e.g. native Prunus padus L. Because it supplants native species, dominates the understory and has high fertility, the concern of Polish National Forests is mainly to eliminate P. serotina from woods. According to data from 2014, Prunus serotina covers almost 100,000 hectares, which represents 1.4% of Polish National Forests (Bijak et al. 2014). Removing P. serotina from European forests is a highly cost-intensive measure, and, until now, no effective method has been developed, so it is nearly impossible to completely eliminate it (Halarewicz 2011a, b; Annighöfer et al. 2012). Its wide occurrence, cost-intensive removal and high yield provide a perfect opportunity to find a use for this plant. Pacyniak and Surmiński (1976) conducted the first research into the utilization of P. serotina from Polish forests, the work being focused on the wood. The conclusion was that black cherry wood is similar to the wood of both birch and beech, because of which it can perhaps be used in the papermaking industry. They also stated that further research is needed to find the best way of utilizing black cherry.
The shape, very often uneven, and the small diameter of the P. serotina trunk reduce the possibility of using this tree in the wood-based industry. The biggest problem with this plant is its large flower and fruit production. These parts plus leaves are a source of compounds in many plants that can be used in value-added products. There is currently a tendency in many areas of life to return to natural products that are both functional and friendly to humans and the environment (so-called bio-based products). There is growing awareness of the importance of products containing natural bioactive compounds, which are ingredients of food, cosmetics, medicines etc. In Poland, the use of fruits and herbs for home production of liqueurs, syrups, and herbal teas is also very popular (Świderski and Waszkiewicz-Robak 2005; Filipiak-Florkiewicz et al. 2015; Żyngiel and Platta 2015). P. serotina may be a source of this group of compounds.
The aim of this work was to identify the phenolic compounds in leaves and flowers of P. serotina. Identification of polyphenolic compounds of P. serotina is a first stage in finding uses for this widespread, invasive species. Furthermore, differences in chemical composition between black cherry and other forest understory species may suggest P. serotina as a species of first choice in term of phenolic extractives. There have been only a few studies on phenolics from European populations of black cherry (Olszewska 2005a, b, 2007; Techlichowska et al. 2020). It follows from interviews with foresters that, for now, black cherry utilization is limited to firewood or for preparing infusions or liqueurs. Different methods of extraction were therefore investigated, firstly, preparations according to traditional recipes, tea from flowers or liqueur using ethanol (home prepared fruit brandy). The first extract uses hot water, because water is the most available and least harmful solvent. Ethanolic extract is prepared from flowers, while ethanolic leaf extract is not prepared because of the taste, and we did not find any evidence of liqueur made from leaves. The third solvent studied was methanol, since it is most often recommended in the literature for the most efficient or maximum extraction of polyphenolic substances. We hypothesized that extraction with ethanol and hot water would result in a lower concentration of some phenolic compounds in the extracts, compared to the standard methanol procedure. The amount of phenolics extracted in the different extraction procedures was closely followed, and the compounds with lower or higher extraction potential were identified.
2 Materials and methods
2.1 Chemicals
Phenolic compound standards were purchased from Sigma-Aldrich Chemie GmbH (Steinheim, Germany): chlorogenic acid, procyanidin B1, p-coumaric acid, caffeic acid, ferulic acid, (-)-epicatechin, quercetin-3-rutinoside, myricetin-3-rhamnoside, kaempferol-3-glucoside, quercetin-3-galactoside and quercetin-3-glucoside; ( +)-catechin from Roth (Karlsruhe, Germany); quercetin-3-xyloside, quercetin-3-arabinofuranoside and quercetin-3-arabinopyranoside from Apin-Chemicals (Abingdon, UK) and isorhamnetin-3-glucoside from Extrasynthèse (Genay, France). All standards were at least 95% pure. The chemicals for solvents methanol, formic acid and acetonitrile were obtained from from Sigma-Aldrich Chemie GmbH, all HPLC grade. Double distilled water, used to prepare all aqueous solutions, was produced using the Millipore purification system (Millipore, Bedford, MA, USA).
2.2 Materials
We used leaves and flowers of black cherry for the study. Flowers were collected in spring, on 22nd May 2019 in full development stage from a location in Dabrowa forestry district (location: latitude 53° 29′ 45.74′′ N, longitude 18° 31′ 0.72′′ E, 79 m altitude). Flowers were collected randomly from ten individual bushes in the given location and then freeze-dried. The dried material was ground with the help of liquid nitrogen and then processed into extracts. The flowers collected from a single bush were used for one biological repetition for analysis. We prepared three types of extracts (methanolic, water and ethanolic) for the analysis.
Fully developed leaves were collected from the same population of P. serotina 2 weeks later, on 4th June 2019 at the phenophase end of flowering (location: latitude 53° 29′ 45.74′′ N, longitude 18° 31′ 0.72′′ E, 79 m altitude). Similar to the flowers, leaves were collected from several shrubs. Each bush represented one repetition for chemical analyses. We prepared ten repetitions for each leaf extract (methanol, water). Lyophilized leaf material was used for the extraction.
2.3 Preparation of extracts
We compared the success of phenolics extraction of black cherry leaves and flowers simulating different traditional extraction procedures. Black cherry leaves were extracted with methanol, which is a standard extraction of phenolics, and with hot water simulating infusion. In extraction of phenolics from flowers, we additionally used also ethanol simulating homemade liqueur. Detailed procedures are described below.
Extracts for optimal/maximum extraction efficiency for phenolics (Magwaza et al. 2016; Mikulic-Petkovsek et al. 2020) from leaves and flowers were prepared as follows: 1 g of dried ground material was placed in a 12-mL screw-top tube, to which 5 mL of extraction solution (60% MeOH) was then added. The mass of material and the volume of MeOH were previously experimentally checked, and all samples were tested by HPLC. Maximum or optimal means of extraction of phenolics were chosen based on the highest peaks on the chromatogram. Extraction of the samples was conducted in an ice-cooled ultrasonic bath for 45 min. The extracts were then centrifuged for 9 min, 9000 rpm at 4 °C, and then filtered through a polyamide 20 µm filter (Macherey–Nagel) into vials. Vials were stored in a freezer at − 20 °C until further examination.
Water extracts of flowers and leaves, an infusion, were prepared in the same way for both plant materials. The procedure was the same as is usually recommended for herbal tea. One gram of ground leaves or flowers was placed in a beaker; 10 mL of hot (below boiling point) distilled water was poured into the beaker. Extraction lasted 10 min under a lid. Extraction was performed at room temperature. The extracts were then transferred into tubes and centrifuged for 9 min, 9000 rpm at 4 °C, and then filtered through a cellulose filter (Macherey–Nagel) into vials and stored in a freezer until further examination.
Additionally, ethanolic extract was prepared for flowers. We chose 40% ethanol as a commercially available solvent, and because fruit brandy (which has approximately 38–42% of ethanol) is used for various home prepared liqueurs. Four mL of 40% EtOH and 0.6 g of ground dry flowers was used. Extraction took 5 days in a closed glass flask at room temperature, as is recommended in the traditional recipe. After 5 days, extracts were centrifuged for 9 min, 9000 rpm at 4 °C, and then filtered through a polyamide filter into vials and stored in a freezer until further examination by HPLC.
2.4 Analysis of phenolic compounds by HPLC–DAD joined with MSn
Phenolic compounds were detected using the Thermo Scientific Dionex HPLC system with diode array detector (Thermo Scientific, San Jose, CA, USA) using Chromeleon workstation software. Phenolic components were analysed at two wavelengths, 280 nm and 350 nm. Analyses were performed with a Gemini C18 (150 × 4.6 mm 3 µm; Phenomenex, Torrance, USA) column heated to 25 °C. We used two mobile phases. Mobile phase A was a mixture of 3% acetonitrile/0.1% formic acid/96.9% bi-distilled water and mobile phase B 3% water/0.1% formic acid/96.9% acetonitrile. Elution of mobile phases was performed according to a linear gradient from 5 to 20% B in the first 15 min, followed by a linear gradient from 20 to 30% B for 5 min, then an isocratic mixture for 5 min, followed by a linear gradient from 30 to 90% B for 5 min, and then an isocratic mixture for 15 min before returning to the initial conditions (Mikulic-Petkovsek et al., 2016). The injection volume of the extract was set at 20 µL and the flow rate of the mobile phase at 0.6 mL/min.
All phenolic compounds were identified with a mass spectrometer (LTQ XL Linear Ion Trap Mass Spectrometer, Thermo Fisher Scientific, USA) with electrospray ionization in negative ion operating mode. The scanning range was from m/z 110 to 1600 — data-dependent full scan. The injection volume of the extract was set at 10 µL and the flow rate at 0.6 mL/min. The capillary temperature was 260 °C; the sheath gas and auxiliary gas were 55 and 13 units, respectively; the source voltage was 5 kV, and normalized collision energy was between 15 and 30%. Spectral data were elaborated using Excalibur software (Thermo Scientific). The contents of identified phenolics were expressed in mg per kg of dried weight (DW) of analysed material.
2.5 Statistical analysis
Statistical data analysis was performed using Statgraphics Plus 4.0 (Manugistics, Rockville, MD, USA). Using a statistical program, we compared the differences in the content of individual phenolic compounds between different extraction techniques. The results are presented in Tables 1 and 2 as mean ± standard deviation of ten repetitions. To test the variance of the measured data, the one-way Kruskal–Wallis test was performed. Using this test, all characteristics were found to follow a normal distribution (p 0.05). Significant differences between different black cherry extracts were calculated by one-way analysis of variance (ANOVA). Estimation of differences in the content levels of individual components was carried out with Fisher’s least significant difference test (p < 0.05).
3 Results
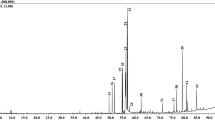
Chromatograms of the HPLC analysis of leaf methanolic extract are presented in Fig. 1, chromatograms of flower methanolic extract are presented in Fig. 2. All identified phenolic compounds of the leaf and flower methanolic extracts are presented in Table 1 and Table 2, respectively.
Chromatograms of Prunus serotina leaves recorded at A 280 nm and B 350 nm. Peak numbers are described in Table 1
Chromatograms of Prunus serotina flowers recorded at A 280 nm and B 350 nm. Peak numbers are described in Table 1
The analysed phenolics are shown in Table 3 and Table 4, in which they are divided into 5 groups: hydroxycinnamic acids, flavones, flavanols, flavanones and flavonols. Table 3 shows the results for phenolics analysed in leaves, whereas Table 4 shows the results for flowers. The contents of the determined compounds are expressed in mg/kg of dried weight (DW).
In total, fifty phenolic components were identified in extracts of black cherry leaves. In terms of the number of identified phenolics, flavonols were the major group (twenty-one compounds). The content of total analysed flavonols was significantly higher with methanol extraction (13.48 g/kg in methanolic extract, 6.5 g/kg in water extract); flavonols’ share in methanolic extract represented 36.9% of total analysed phenolics and 23% of total analysed phenolics in water extract (Table 1). Eight different glycosides of quercetin, followed by five kaempferol glycosides, seven isorhamnetin glycosides and syringetin hexoside were identified. Quercetin-3-arabinofuranoside (avicularin) was the major compound among all flavonols, accounting for 38% of flavonols in the extract in which 60% MeOH was used and 28% in water extract. The second prevalent flavonol was quercetin-3-glucoside, 2.6 g/kg in methanolic extract and 1.4 g/kg in water extract. These two flavonols exceeded 1 g/kg in the water infusion. Two more compounds exceeded this amount with the methanolic extract: quercetin-3-xyloside (1473 mg/kg in methanolic extract; 722 mg/kg in water extract) and quercitin-3-rutinoside (1046 mg/kg in methanolic extract; 857 mg/kg in water extract). The amount of quercetin-3-rutinoside was lower in the infusion (857 mg/kg), but the difference was not significant.
The second largest group of compounds in black cherry leaves was hydroxycinnamic acid derivatives (HCA). Fourteen derivatives of HCA were identified in the infusion and thirteen in the standard extract. HCA represented 24.1% of total analysed phenolics (TAP) in the methanolic extract and 37.9% of TAP in the infusion (Table 1). In both extracts, the compound with the highest share was chlorogenic acid (5-caffeoylquinic acid 1). This was confirmed with the standard of trans-5-caffeoylquinic acid. It represented 66% of HCA and 16% of TAP (5.77 g/kg) in the methanolic extract and, in the infusion, 70% of HCA and 27% of TAP (7.55 g/kg). The compound with the second largest quantity was 5-caffeoylquinic acid 2. This compound is probably cis-5-caffeoylquinic acid because it has the same fragmentation pattern as chlorogenic acid. Its content in the methanol extract was 936 mg/kg, while in the infusion it was 1268 mg/kg. The two isomers represented 76% HCA in methanolic extract and 82% HCA in water extract. Differences in quantities of these two isomers between extracts were not significant. There were three isomers of caffeic acid hexoside in water extract and two of the same isomers in methanolic extract. These isomers represented approx. 1% of HCA in methanolic extract and 3% of HCA in water extract. Three isomers of p-coumaric acid hexoside were also identified. The isomer with the largest quantity was p-coumaric acid hexoside 3 (223 mg/kg in ME and 293 mg/kg in water extract).
The third group in, in terms of number of identified compounds, was flavanols. In water and methanolic extracts, flavanols represented 38% of TAP. In methanolic extracts, the amount of flavanols was 14 g/kg, and in infusion it was 10.84 g/kg of flavanols. The LSD test showed that the difference in the content of flavanols was not significant between the two extracts. We identified twelve different flavanols in the infusion and ten in the methanolic extract extract. The identified compounds were catechin, epicatechin, three isomers of procyanidin dimer and seven isomers of procyanidin trimer. Isomers of procyanidin dimer represented 25% of flavanols in methanolic extract and 51% of FLA in water extract. Procyanidin trimer isomers represented 56% of flavanols in methanolic extract, while in the water extract they represented 38% of FLA. There was almost three times more catechin in methanolic extract than in the infusion (17 flavanols and 6% flavanols, respectively). Epicatechin was also identified in the extracts, its amount being approx. 3% of flavanols in methanolic extract and 5% of flavanols in water extract.
Groups of flavanones and flavones in leaves consist of three compounds. The compounds of the flavanones group were eriodictyol and eriodictyol hexoside. The share of this compound in TAP was fairly small, in methanolic extract only 0.5% TAP, whereas in the infusion, it was 0.7% of TAP. Flavones were represented by one compound: apigenin hydroxyhexoside; in both extracts it was about 0.2% of TAP.
Black cherry flowers were extracted with three different solvents: methanol as the most widely used for phenolic extraction, 40% ethanol used for an alcoholic drink or liqueur and hot water — as an infusion or tea extract. The contents of phenolic compounds in these three extracts are shown in Table 4. In total, forty-two different phenolic compounds were identified, thirty-six of them being in common with the compounds identified in leaf extracts. We got the exactly the same compounds with the different extraction solutions, but there were big differences in their concentrations. Comparable with the leaves, phenolics were divided into six groups. Ten compounds of hydroxycinnamic acid derivatives (HCA) were identified in flowers, one flavanone, six compounds of flavanols, twenty-four flavonols and one flavone.
The group of flavanols in flowers was responsible for 58% of total analysed phenolics (TAP) in methanolic extract and ethanolic extract and 45% of TAP in water extract. Four derivatives of procyanidin represented more than 50% of TAP in both alcohol extracts, and over 40% of TAP in the water infusion. The major compound in each extract was procyanidin trimer 1, in methanolic extract representing 25% of TAP, in ethanolic extract 30% of TAP and in water extract 13% of TAP.
In flowers, the largest group of compounds were flavonols; twenty-four different phenolics were determined. The group consisted of nine glycosides of quercetin, seven glycosides of kaempferol, seven glycosides of isorhamnetin and syringetin hexoside. Flavonols represented 32% of TAP in methanolic extract, 31% of TAP in ethanol extract and 34% of TAP in the water infusion. Glycosides of quercetin represented approx. 70% of flavonols in each of the three extracts. Among them, three glycosides were most relevant: quercetin 3-rutinoside (rutin) with a significantly higher quantity in ethanol (3.04 g/kg followed by 2.50 g/kg in methanolic extract and 2.12 g/kg in water extract) and quercetin 3-glucoside, of which the quantity was again significantly higher in ethanol extract (4.03 g/kg followed by 3.29 g/kg in methanolic extract and 3.16 g/kg in water extract, Table 2). The last of those three glycosides was quercetin 3-arabinofuranoside (avicularin). The quantity of avicularin was significantly higher (3.87 g/kg in methanolic extract, 3.76 g/kg in ethanol compared to 1.12 g/kg in water infusion). The two compounds, kaempferol pentoside 2 and kaempferol hexoside, represented the highest share of kaempferol glycosides. Their content in alcohol extracts was significantly higher than in infusion.
Hydroxycinnamic acids (HCAs) were the third most relevant group in black cherry flowers. The major elements of this group consisted of three derivatives of p-coumaric acid hexoside. There was the highest quantity of them in water extract (2.99 g/kg) followed by ethanolic (2.68 g/kg) and methanolic extract (2.18 g/kg). The second relevant compound was 5-feruloylquinic acid, its amount being significantly higher in ethanolic extract. Two isomers of 5-p-coumaroylquinic acid were also better extracted with ethanol, over 800 mg/kg compared to approx. 600 mg/kg in methanol extract and around 550 mg/kg in water extract. HCA represented about 9% of TAP in methanolic extract, 10% of TAP in ethanolic and 18% of TAP in infusion.
The two remaining groups in flowers were flavanones and flavones, each of them being represented by one compound. In flavanones this compound was eriodictyol hexoside. Eriodictyol hexoside in methanolic extract represented 0.4% of TAP, in ethanolic 0.7% of TAP and 2.8% of TAP in infusion. Flavones were represented by apigenin hydroxyhexoside, which constituted 0.2% of TAP in each extract. Its amount was in the range from 68.6 to 133 mg/kg.
The level of total analysed phenolics showed no significant differences among extraction methods in leaf samples, and differences between each group were also not significant. An exception was the group of flavonols; the content of which was twofold lower in leaf water infusion. Total analysed phenolics were at a similar level in both leaf and flower infusions (28.3 g/kg in leaf infusion and 29.7 g/kg in flower infusion). The impact of the extraction method on extractives is more clearly seen in flower extracts. Five days of maceration in 40% ethanol gave the best results of TAP content, as well as HCA and flavones extraction. Flavanones extraction was better in water infusion, giving four times higher contents than methanolic extract and almost two times more than for ethanolic extract (Table 4). Flavanols from the flowers were significantly better extracted in ethanol 33.39 g/kg, followed by methanol 29.1 g/kg and water 13.28 g/kg. Hydroxycinnamic acids had a higher content in leaf than in flower extracts (8.78 and 10.72 g/kg leaves compared with 6.0, 5.21 and 4.34 g/kg flowers).
The content of total analysed phenolics in black cherry flowers and leaves are shown in Fig. 3. The quantity of phenolics in both leaf extracts and in flower water infusion was very similar, the difference not being significant. Methanolic extraction showed that flowers are richer in phenolics than leaves. Extraction using 40% ethanol was the most efficient and allowed the extraction of nearly 60 g of phenolics per kilogram of dried flowers.
Content of total analysed phenolics in black cherry leaves and flowers (g/kg DW) using different extraction methods. Methanol, standard extraction for phenolics (60% MeOH); water, extraction with hot water (infusion); ethanol (liqueur, 40% EtOH). Different letters (a–c) indicate significant differences in phenolic contents between different treatments (p < 0.05) by Duncan’s multiple range test
4 Discussion
To the best of our knowledge, this paper is the first to report a detailed analysis of phenolic components from black cherry leaves and flowers. It was observed that people have started to use wild plants, including black cherry, in various forms: liqueurs, infusions, jams and syrups. The use of black cherry in Poland was first reported in 2007 (Łuczaj 2011). There is still a lack of information on the influence of process methods on phenolics extraction from different plant parts of P. serotina. In previous studies (Olszewska 2005a, b, 2007; Techlichowska et al. 2020), phenolics compounds in Prunus serotina have also been reported, but, in our study, we performed a detailed analysis and accurate identification of a broad spectrum of phenolics in three different extracts.
Polyphenolics are an important group of compounds in human diet, the average daily consumption varying from 25 mg to 1 g/person/day. Polyphenolics are important in human diet for the prevention of lifestyle diseases (Przeor et al. 2019). The main groups of polyphenols important in human diet are flavonoids, phenolic acids, lignans, and stilbenes (Spencer et al. 2008). The health-promoting properties of plants and plant products are strongly connected with their bioactive constituents, such as the above-mentioned polyphenolics, essential oils and resins (Kobus-Cisowska et al. 2019). The main sources of polyphenolics in the European diet are beverages: tea and coffee (Clifford 1999; Scalbert and Williamson 2000; Zamora-Ros et al. 2015). The leaves, flowers and bark of black cherry contain bioactive substances (Olszewska 2005a, b, 2007; Techlichowska et al. 2020). Extracts of these plant parts are used in traditional Mexican medicine. Tea made from leaves of Prunus serotina, in Mexico called Capulin, is used as a natural remedy for stomach upsets, hypertension, diarrhoea, paludism, bronchitis, mouth infections and coughs (Ibarra-Alvarado et al. 2009). Leaves and flowers of black cherry are used in the treatment of intestinal, cardiovascular and pulmonary diseases (Camacho-Corona et al. 2015).
A water infusion of black cherry leaf is used in traditional folk medicine (Rivero-Cruz 2014). The leaf infusion can be compared to a tea. The tea is usually made of three grams of leaves, which are infused in 200 mL of boiling water. Total flavonols in tea infusions have been studied, their content varying from 36.5 to 75.8 mg/L for regional teas and from 45.0 to 88.3 mg/L in blended black teas (McDowell et al. 1990). Our results showed that an infusion (tea) from black cherry leaves contained around 97.5 mg flavonols/L and tea from flowers contained 151.8 mg flavonols/L (data are not shown). Infusions of black cherry leaves or flowers thus appear to be a better source of flavonol glycosides than most common teas.
Previous studies on phenolics from Prunus serotina leaves showed seven flavonol glycosides in the study of Olszewska (2005a, b), six flavonoids and chlorogenic acid in the study of Olszewska (2007) and fourteen phenolic compounds in the study of Techlichowska et al. (2020). Among these fourteen compounds were eleven phenolic acids and three flavonoids. In our work, fifty phenolics were identified, of which fourteen were HCAs and thirty-six flavonoids. Comparing our results with previous papers, some of the quercetin derivatives were also identified in Olszewska (2007). These are quercetin-3-xyloside, quercetin-3-arabinofuranoside and quercetin-3-rutinoside, which was detected in Olszewska (2005a, b, 2007) but which was not detected by Techlichowska et al. (2020). All previously mentioned derivatives of quercetin were also identified in our extracts, and we identified many new compounds, e.g. various phenolic acid derivatives, procyanidins, isorhamnetin and kaempferol glycosides, which had not previously been determined in black cherry leaves and flowers.
Total analysed phenolics (TAP) for leaf extracts of black cherry ranged from 28 g/kg DW (water infusion) to 37 g/kg DW (methanolic extract). In the work of Jesus et al. (2019), sweet cherry (Prunus avium) infusion had almost 47 g/kg DW of TAP, and ethanolic extract had almost 70 g/kg DW of TAP, but the final concentration of extract also depends on the mass of material and volume of solution. In the leaves of P. avium, 14 compounds were identified in the work of Jesus et al. (2019), as opposed to 50 compounds identified in our study. In both studies, the major component of the group of HCAs was 5-caffeoylquinic acid (chlorogenic acid). Among flavonoids, the main component in sweet cherry leaves was rutin, while in black cherry leaves, the main flavonoid was quercetin-3-arabinofuranoside. Similarly, as in both black and sweet cherry, the main phenolic component in sour cherry leaf was chlorogenic acid (Oszmiański and Wojdyło 2014). A total of 36 phenolic compounds were identified in P. cerasus leaves, including 22 flavonoids and 14 HCAs. The richest flavonoids in black cherry were procyanidins and (-)-epicatechin, which are probably the reason for the astringent taste of the prepared product. Peleg et al. (1999) also reported that the stage of flavanol polymerization increased astringency. The second reason for the astringent taste of black cherry leaves is probably cyanogenic glycosides (CGG). Prunus serotina leaves contain cyanogenic glycoside prunasin (Zhou et al., 2002). When leaves are damaged, CGG release hydrogen cyanide (HCN), which is toxic for organisms (Hu and Poulton, 1999). The main role of cyanogenesis is thus plant protection against pathogens and herbivores. Fitzgerald (2008) reported that black cherry leaves have a mean value of HCN of 1068 ppm of cyanide.
Black cherry leaves from our research had about half the content of total analysed phenolics (36.51 mg/g DW) in comparison with sour cherry leaves (87.37 mg/g DW) (Oszmiański and Wojdyło 2014). Alcoholic extracts of black cherry flowers were richer in phenolic compounds than their leaves. Water infusions of leaves and flowers had similar phenolic content. Two extraction methods for leaves and three methods for flowers were used in this study. The influence of solvent on extracted phenolics is significant. While the share of flavanols among total analysed phenolics was almost the same, 38.4 and 38.3%, in both leaf extracts (methanolic and water), hydroxycinnamic acids had the highest share of TAP in water leaf extract, 37.9% of TAP compared to 24.1% of TAP in methanolic leaf extract. In flower extracts, HCAs also had the highest share of TAP in water extracts compared to 60% methanol and 40% ethanol (17.5% of TAP in water followed by 8.7% in methanolic extract and 10.4% in ethanolic extract). Better extraction of HCAs in water has also been previously described in other studies (Reis et al. 2012; Mikulic-Petkovsek et al. 2015). The proportion of flavonoids was higher in both organic solvent extracts compared to the water extract. In the leaf extract, the major difference in flavonol glycosides was 36.9% of TAP in methanolic extract and 23% of TAP in water extract. In flower extracts, the biggest difference was in the flavanol group. The share of flavanols in both organic solutions was about 58% of TAP, while in the water extract, this share was 45% of TAP. It has also been previously reported that flavonoids are better extracted in organic solvents (especially methanol) (Siddhuraju and Becker 2003; Chaves et al. 2020).
One of the most widely used Polish wild shrubs is elderberry (Sambucus nigra). Fruits and flowers of this species are used to make traditional syrups, liquors, teas and jams (Senica et al. 2016). The results show that the flowers of P. serotina are richer in phenolics compounds than the flowers of S. nigra. A study by Todorovic et al. (2017) showed that elderflower water extracts contained 4145 mg of HCAs/kg, but our results for black cherry flower water extracts were 5211 mg of HCA/kg. The amount of flavonols and flavanones was also higher in the infusion of P. serotina flowers than in the infusion of S. nigra flowers (10.12 vs. 8.22 g/kg of flavonols, 0.84 vs. 0.39 g/kg of flavanones) (Todorovic et al. 2017). The water infusion of S. nigra flowers contained approx. 6703 mg of quercetin glycosides/kg DW, and it was found to be a rich source of these compounds. Similarly, the water extracts of P. serotina flower had similar levels of quercetin glycosides (7106 mg/kg), in contrast to the leaf water extracts which had slightly lower contents of them (5358 mg derivatives of quercetin/kg). The results also showed that there was no significant difference in the contents of TAP between flower and leaf extracts (29.72 for flowers, 28.32 g/kg for leaves). The main group of phenolics in elderberry leaves and flowers were flavonols (Todorovic et al. 2017; Senica et al. 2017). The results for black cherry leaves showed that flavonols together with flavanols were the main groups of phenolics, while the main group in flowers was flavanols. Flavanols content in flower methanolic extract was twofold higher than that of flavonols.
Methanolic extract of fresh elderberry flowers has been reported to be rich in phenolics (Mikulic-Petkovsek et al. 2015). Comparing the concentrations of some phenolic groups, black cherry leaves contain higher levels of HCA, and especially higher levels of flavanols (14 and 29.1 g/kg) and flavonol glycosides (13.4 and 15.9 g/kg DW) have been determined in both plant parts, leaves and flowers. Total phenolic content was higher in P. serotina leaves (36.5 g/kg DW) and flowers (49.8 g/kg) than in S. nigra (10.2 g/kg FW) (Mikulic-Petkovsek et al. 2015). Comparing the contents of total phenolics in leaves and flowers with some Prunus species that are grown in intensive cultivation, black cherry contains higher or similar contents than the mentioned species. In previous studies, total phenolics in extracts from Prunus avium leaves were 46.3 g/kg DW and from flowers 27.3 g/kg (Jesus et al. 2019). It has been found that Prunus armeniaca flowers reach only 4.04 to 7.68 g/kg FW (Gottingerova et al. 2020).
5 Conclusion
In this study we identified a larger number of phenolic compounds than in previous works. Infusions of hand-picked black cherry leaves and flowers are a good source of phenolics, especially flavonols, which have a higher content than black tea. The identified phenolic compounds, fifty in leaves and forty-two in flowers, and also their high content, allow the assertion that infusions and liqueurs made with the investigated parts of black cherry are rich in phenolics. Methanolic extract of leaves contained 36.51 g of total analysed phenolics (TAP)/kg DW, and methanolic extract of flowers had 49.82 g of TAP/kg DW. Different solvents used for the extraction of phenolic compounds allow extracts with different quantities of phenolic groups to be obtained. Ethanolic flower extract had the highest content of hydroxycinnamic acids, flavanols and TAP, and with water there was the lowest content of flavonols and TAP in the flower extract. The highest efficiency of extraction of flavonols and TAP from the leaves was obtained with methanol, which gave about two times higher content of flavonols and about 30% higher content of total analysed phenolics than with water. It is recommended that the extraction method is adjusted according to the expected amount of phenolics in the extract. Leaves and flowers of Prunus serotina are similar or richer in phenolic compounds than the leaves of widely used Prunus species such as Prunus avium and Prunus armeniaca. Flowers of industrially utilized sweet cherry and apricot are not considered to be a phenolic source since they are needed to produce fruits. On the other hand, collecting the flowers of black cherry can make this plant more valuable and will reduce plant expansion in the forest. Leaves and flowers of the widespread black cherry, which is a problem in many European countries, should be considered by the pharmaceutical, cosmetics and food industries and may become a food additive, enriching products with phenolic compounds. However, further studies on the toxicology of products made from Prunus serotina, as well as studies of antioxidant activity and possible activity against microorganisms, will be required in the future.
This work was performed as part of Research Programme P4-0013–0481 Horticulture, of the Slovenian Research Agency (ARRS). Additionally, the results were obtained within the Erasmus + scholarship programme of the corresponding author.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Annighöfer P, Mölder I, Zerbe S, Kawaletz H, Terwei A, Ammer C (2012) Biomass functions for the two alien tree species Prunus serotina Ehrh. and Robinia pseudoacacia L. in floodplain forests of Northern Italy. Eur J for Res 131:1619–1635. https://doi.org/10.1007/s10342-012-0629-2
Bijak S, Czajkowski M, Ludwisiak Ł (2014) Occurrence of black cherry (Prunus serotina Ehrh.) in the State Forests in Poland. For Res Pap 75:359–365
Camacho-Corona MR, Garcia A, Mata-Cardenas BD, Garza-Gonzalez E, Ibarra-Alvarado C, Rojas-Molina A, Rojas-Molina I, Bah M, Sanchez MAZ, Gutierrez SP (2015) Screening for antibacterial and antiprotozoal activities of crude extracts derived from Mexican medicinal plants. Afr J Tradit Complement Altern Med 12:104–112. https://doi.org/10.4314/ajtcam.v12i3.13
Chaves JO, de Souza MC, da Silva LC, Lachos-Perez D, Torres-Mayanga PC, Machado APF, Forster-Carneiro T, Vazquez-Espinosa M, Gonzalez-de-Peredo AV, Barbero GF, Rostagno MA (2020) Extraction of flavonoids from natural sources using modern techniques. Front Chem 8. https://doi.org/10.3389/fchem.2020.507887
Csiszár Á, Korda M, Schmidt D, Šporčić D, Süle P, Teleki B, Tiborcz V, Zagyvai G, Bartha D (2013) Allelopathic potential of some invasive plant species occurring in Hungary. Allelopathy J 31:309–318
Clifford MN (1999) Chlorogenic acids and other cinnamates – nature, occurrence and dietary burden. J Sci Food Agric 79:362–372
Deckers B, Verheyen K, Hermy M, Muys B (2005) Effects of landscape structure on the invasive spread of black cherry Prunus serotina in an agricultural landscape in Flanders, Belgium. Ecography 28:99–109. https://doi.org/10.1111/j.0906-7590.2005.04054.x
Desiea E, Vancampenhoutb K, van den Berg L, Nyssena B, Weijterse M, den Ouden J, Muysa B (2020) Litter share and clay content determine soil restoration effects of rich litter tree species in forests on acidified sandy soils. For Ecol Manag 474. https://doi.org/10.1016/j.foreco.2020.118377
Filipiak-Florkiewicz A, Florkiewicz A, Topolska K, Cabala A (2015) Żywność funkcjonalna (prozdrowotna) w opinii klientów specjalistycznych sklepów z żywnością. Bromatol Chem Toksykol 43:66–175
Fitzgerald TD (2008) Larvae of the fall webworm, Hyphantria cunea, inhibit cyanogenesis in Prunus serotina. J Exp Biol 211:671–677. https://doi.org/10.1242/jeb.013664
Grajewski S, Jankowski K, Licznierski M (2010) Czeremcha amerykańska Prunus serotina Ehrh. w polskich lasach na przykładzie drzewostanów nadleśnictwa doświadczalnego Zielonka oraz nadleśnictwa państwowego Szubin. Zarządzanie Ochr Przyr W Lasach 4
Gentili R, Ferrè C, Cardarelli E, Montagnani C, Bogliani G, Citterio S, Comolli R (2019) Comparing negative impacts of Prunus serotina, Quercus rubra and Robinia pseudoacacia on native forest ecosystems. Forests 10:842. https://doi.org/10.3390/f10100842
Godefroid S, Phartyal SS, Weyembergh G, Koedam N (2005) Ecological factors controlling the abundance of non-native invasive black cherry (Prunus serotina) in deciduous forest understory in Belgium. For Ecol Manag 210:91–105. https://doi.org/10.1016/j.foreco.2005.02.024
Gottingerova M, Kumsta M, Necas T (2020) Health-benefitting biologically active substances in edible apricot flowers. Hortscience 55:1372–1377. https://doi.org/10.21273/HORTSCI15038-20
Halarewicz A (2011a) Odnawianie się czeremchy amerykańskiej (Prunus serotina Ehrh.) na siedliskach borowych. Sylwan 155:530–534
Halarewicz A (2011b) Przyczyny i skutki inwazji czeremchy amerykańskiej Prunus serotina w ekosystemach leśnych. Leśne Pr Badaw 72:267–272
Halarewicz A, Pruchniewicz D (2015) Vegetation and environmental changes in a Scots pine forest invaded by Prunus serotina: what is the threat to terricolous bryophytes? Eur J for Res 134:793–801. https://doi.org/10.1007/s10342-015-0890-2
Hu ZH, Poulton JE (1999) Molecular analysis of (R)-(+)-mandelonitrile lyase microheterogeneity in black cherry. Plant Physiol 119:1535–1546. https://doi.org/10.1104/pp.119.4.1535
Ibarra-Alvarado C, Rojas A, Luna F, Rojas JI, Rivero-Cruz B, Rivero-Cruz JF (2009) Vasorelaxant constituents of the leaves of Prunus serotina “capulín.” Rev Latinoam Quim 37:164–173
Jesus F, Goncalves AC, Alves G, Silva LR (2019) Exploring the phenolic profile, antioxidant, antidiabetic and anti-hemolytic potential of Prunus avium vegetal parts. Food Res Int 116:600–610. https://doi.org/10.1016/j.foodres.2018.08.079
Kawaletz H, Mölder I, Annighöfer P, Terwei A, Zerbe S, Ammer C (2014) Back to the roots: how do seedlings of native tree species react to the competition by exotic species? Ann for Sci 71:337–347. https://doi.org/10.1007/s13595-013-0347-z
Kobus-Cisowska J, Szymanowska-Powałowska D, Szczepaniak O, Kmiecik D, Przeor M, Gramza-Michałowska A, Cielecka-Piontek J, Smuga-Kogut M, Szulc P (2019) Composition and in vitro effects of cultivars of Humulus lupulus L. hops on cholinesterase activity and microbial growth. Nutrients 11:1377. https://doi.org/10.3390/nu11061377
Łuczaj Ł (2011) Dziko rosnące rośliny jadalne użytkowane w Polsce od połowy XIX w. do czasów współczesnych. Etnobiologia Pol 1:57–125
Magwaza LS, Opara UL, Cronje PJR, Landahl S, Ortiz JO, Terry LA (2016) Rapid methods for extracting and quantifying phenolic compounds in citrus rinds. Food Sci Nutr 4:4–10. https://doi.org/10.1002/fsn3.210
McDowell I, Bailey RG, Howard G (1990) Flavonol glycosides in black tea. J Sci Food Agric 53:411–414. https://doi.org/10.1002/jsfa.2740530314
Mikulic-Petkovsek M, Samoticha J, Eler K, Stampar F, Veberic R (2015) Traditional elderflower beverages: a rich source of phenolic compounds with high antioxidant activity. J Agric Food Chem 63:1477–1487. https://doi.org/10.1021/jf506005b
Mikulic-Petkovsek M, Stampar F, Veberic R, Sircelj H (2016) Wild Prunus fruit species as a rich source of bioactive compounds. J Food Sci 81:C1928–C1937. https://doi.org/10.1111/1750-3841.13398
Mikulic-Petkovsek M, Koron D, Rusjan D (2020) The impact of food processing on the phenolic content in products made from juneberry (Amelanchier lamarckii) fruits. J Food Sci 85:386–393. https://doi.org/10.1111/1750-3841.15030
Motta R, Nola P, Berretti R (2009) The rise and fall of the black locust (Robinia pseudoacacia L.) in the “Siro Negri” Forest Reserve (Lombardy, Italy): lessons learned and future uncertainties. Ann for Sci 66:410–410. https://doi.org/10.1051/forest/2009012
Nestby RDJ (2020) The status of Prunus padus L. (bird cherry) in forest communities throughout Europe and Asia. Forests 11. https://doi.org/10.3390/f11050497
Olszewska M (2007) Quantitative HPLC analysis of flavonoids and chlorogenic acid in the leaves and inflorescences of Prunus serotina Ehrh. Acta Chromatogr 19:253–269
Olszewska M (2005a) Flavonoids from Prunus serotina Ehrh. Acta Pol Pharm 62:127–133
Olszewska M (2005b) High-performance liquid chromatographic identification of flavonoid monoglycosides from Prunus serotina Ehrh. Acta Pol Pharm 62:435–441
Oszmiański J, Wojdyło A (2014) Influence of cherry leaf-spot on changes in the content of phenolic compounds in sour cherry (Prunus cerasus L.) leaves. Physiol Mol Plant Pathol 86:28–34. https://doi.org/10.1016/j.pmpp.2014.03.002
Pacyniak C, Surmiński J (1976) Drewno czeremchy zwyczajnej i amerykańskiej. Rocz Dendrol 29:147–151
Peleg H, Gacon K, Schlich P, Noble AC (1999) Bitterness and astringency of flavan-3-ol monomers, dimers and trimers. J Sci Food Agric 79:1123–1128. https://doi.org/10.1002/(SICI)1097-0010(199906)79:8%3c1123::AID-JSFA336%3e3.0.CO;2-D
Petitpierre B, Pairon M, Broennimann O, Jacquemart AL, Guisan A, Besnard G (2009) Plastid DNA variation in Prunus serotina var. serotina (Rosaceae), a North American tree invading Europe. Eur J for Res 128:431–436. https://doi.org/10.1007/s10342-009-0287-1
Poyet M, Eslin P, Héraude M, Le Roux V, Prévost G, Gibert P, Chabrerie O (2014) Invasive host for invasive pest: when the Asiatic cherry fly (Drosophila suzukii) meets the American black cherry (Prunus serotina) in Europe. Agric for Entomol 16:251–259. https://doi.org/10.1111/afe.12052
Przeor M, Flaczyk E, Beszterda M, Szymandera-Buszka KE, Piechocka J, Kmiecik D, Szczepaniak O, Kobus-Cisowska J, Jarzębski M, Tylewicz U (2019) Air-drying temperature changes the content of the phenolic acids and flavonols in white mulberry (Morus alba L.) leaves. Ciênc Rural 49
Reis SF, Rai DK, Abu-Ghannam N (2012) Water at room temperature as a solvent for the extraction of apple pomace phenolic compounds. Food Chem 135:1991–1998. https://doi.org/10.1016/j.foodchem.2012.06.068
Rivero-Cruz B (2014) Simultaneous quantification by HPLC of the phenolic compounds for the crude drug of Prunus serotina subsp capuli. Pharm Biol 52:1015–1020. https://doi.org/10.3109/13880209.2013.876054
Scalbert A, Williamson G (2000) Dietary intake and bioavailability of polyphenols. J Nutr 130:2073S-2085S
Senica M, Stampar F, Veberic R, Mikulic-Petkovsek M (2016) Processed elderberry (Sambucus nigra L.) products: A beneficial or harmful food alternative? LWT-Food Science and Technology 72:182–188. https://doi.org/10.1016/j.lwt.2016.04.056
Senica M, Stampar F, Veberic R, Mikulic-Petkovsek M (2017) The higher the better? Differences in phenolics and cyanogenic glycosides in Sambucus nigra leaves, flowers and berries from different altitudes. J Sci Food Agric 97:2623–2632. https://doi.org/10.1002/jsfa.8085
Siddhuraju P, Becker K (2003) Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J Agric Food Chem 51:2144–2155. https://doi.org/10.1021/jf020444
Spencer JP, Abd El Mohsen MM, Minihane AM, Mathers JC (2008) Biomarkers of the intake of dietary polyphenols: strengths, limitations and application in nutrition research. Br J Nutr 99(1):12–22. https://doi.org/10.1017/S0007114507798938
Świderski F, Waszkiewicz-Robak B (2005) SkLadniki bioaktywne w żywności funkcjonalnej. Przemysl Spoż 59:20–22
Techlichowska, Kobus-Cisowska J, Ligaj M, Stuper-Szablewska K, Szymanowska D, Tichoniuk M, Szulc P (2020) Polyphenol content and antioxidant activities of Prunus padus L. and Prunus serotina L. leaves: electrochemical and spectrophotometric approach and their antimicrobial properties. Open Chem 18:1125–1135. https://doi.org/10.1515/chem-2020-0121
Todorovic B, Mikulic-Petkovsek M, Stampar F, Ivancic A (2017) Phenolic compounds in floral infusions of various Sambucus species and their interspecific hybrids. Turk J Agric for 41:154–164. https://doi.org/10.3906/tar-1702-26
Zamora-Ros R, Knaze V, Rothwell J, Hémon B, Moskal A, Overvad K, Tjønneland A, Kyrø C, Fagherazzi G, Boutron-Ruault M-C, Touillaud M, Katzke V, Kühn T, Boeing H, Foerster J, Trichopoulou A, Valanou E, Peppa E, Palli D, Scalbert A (2015) Dietary polyphenol intake in Europe: the European prospective investigation into cancer and nutrition (EPIC) study. Eur J Nutr 55:1359–1375. https://doi.org/10.1007/s00394-015-0950-x
Zhou JM, Hartmann S, Shepherd BK, Poulton JE (2002) Investigation of the microheterogeneity and aglycone specificity-conferring residues of black cherry prunasin hydrolases. Plant Physiol 129:1252–1264. https://doi.org/10.1104/pp.010863
Żyngiel W, Platta A (2015) Oczekiwania konsumentów wobec preparatów kosmetycznych pochodzenia naturalnego wykorzystywamericananych w zabiegach SPA & Wellness. Handel Wewnetrzny 324–333
Acknowledgements
Material for this paper was provided thanks to Polish State Forests from the area of Nadleśnictwo Dąbrowa.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Jean-Michel Leban
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributions of the co-authors
Conceptualization: Jakub Brózdowski. Methodology: Maja Mikulic-Petkovsek. Software: Maja Mikulic-Petkovsek. Validation: Maja Mikulic-Petkovsek. Formal analysis: Maja Mikulic-Petkovsek, Sasa Gacnik. Investigation: Maja Mikulic-Petkovsek, Jakub Brózdowski. Resources: Robert Veberic. Data curation: Jakub Brózdowski. Writing — original draft: Jakub Brózdowski. Writing — review and editing: Maja Mikulic-Petkovszek, Boguslawa Waliszewska, Robert Veberic. Visualisation: Jakub Brózdowski. Supervision: Maja Mikulic-Petkovszek, Boguslawa Waliszewska, Robert Veberic. Project administration: Robert Veberic. Funding acquisition: Metka Hudina.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brozdowski, J., Waliszewska, B., Gacnik, S. et al. Phenolic composition of leaf and flower extracts of black cherry (Prunus serotina Ehrh.). Annals of Forest Science 78, 66 (2021). https://doi.org/10.1007/s13595-021-01089-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13595-021-01089-6