Abstract
Key message
Natural regeneration patterns of conifer species were studied. Seedling regeneration follows patterns responding to stand structure and site condition factors along shade and drought tolerance gradients. Our findings can assist in adaptive forest management for maintaining sustainable regeneration and plant biodiversity.
Context
Seedling regeneration can vary with stand factors of overstory trees and understory non-tree vegetation and site conditions.
Aims
Natural seedling regeneration patterns of coniferous species were investigated using Forest Inventory and Analysis (FIA) data of 10 common species across the Inland Northwest, USA.
Methods
Zero-inflated negative binomial models were developed to understand the responses of natural regeneration to stand factors and site conditions.
Results
Seedling occurrence varies along shade and drought tolerance gradients responding to stand structure and site conditions. Two moderate shade-tolerant species of different drought tolerance contributed as a transition. Strong response patterns were revealed for seedling density, in which seedling density was improved with the presence of conspecific trees while limited by competition, especially from the understory vegetation layer.
Conclusion
Overstory structure and understory vegetation could improve or hinder natural regeneration of coniferous tree species given different shade tolerance and site conditions. Our findings can be effectively implemented in adaptive forest management for maintaining sustainable regeneration of specific conifers in broad temperate mixed forests.
Similar content being viewed by others
1 Introduction
Forest regeneration is fundamental to forest succession and is integral to maintaining ecosystems and ecosystem processes. Natural regeneration responds and adapts to various stand factors including overstory and understory composition and structure and site conditions (Grubb 1977; Hart and Chen 2006; Walters et al. 2016). The understory vegetation layer has the greatest plant biodiversity in forest ecosystems (Gilliam 2007). The ecological significance of understory vegetation in forest ecosystems has been studied and highlighted by previous studies and reviews (e.g., Gilliam 2007). The canopy and understory layers may facilitate seedlings by improving microclimatic conditions and soil properties (Callaway 2007; Bonanomi et al. 2011). At the other hand, the presence of trees, shrubs, or herbaceous vegetation can compete with the seedlings for essential resources and thus affects how well seedlings regenerate (Lewis and Tanner 2000; Al-Namazi et al. 2017). Often studies emphasize the significance of overstory trees or isolate the effects of different vegetation functional groups (e.g., herbaceous vegetation or shrubs) when examining effects of stand factors on seedling growth or survival (Gilliam 2007; Vandenberghe et al 2006; Jensen and Löf 2017). In order to better understand natural regeneration and the response to growth conditions, it is imperative to study the impacts of multi-layer stand factors and examine whether both overstory and understory could significantly affect neighboring natural regeneration of tree species in the forests.
Single tree species have been studied in different forest types or different regions, among which findings generally showed complex effects of stand factors on regeneration of various species. The complexity was exemplified by showing that one type of factor could either facilitate or hinder seedling growth. For instance, a conspecific adult canopy tree can facilitate seedlings by serving as a seed source (Comita et al. 2007). Contrarily, overstory trees can obstruct regeneration by reducing available light and deteriorating soil conditions required for seedling germination and growth of coniferous species, (for example, longleaf pine [Pinus palustris Mill.], Battaglia et al. 2002). Understory vegetation can have divergent influences on seedling growth and survival in different forest types. Seedling growth of woody species could be impeded or even prevented by competition for resources from herbaceous species and shrub species (Tilman 1994; Gordon and Rice 2000; Forbes et al. 2016; Thrippleton et al. 2016). Positive effects of understory vegetation (e.g., protection from herbivory browsing) have also been observed widely at early stages of seedling development (Jensen et al. 2012; Gavinet et al. 2016). Studies in Asian and European temperate forests and temperate forests of the southern hemisphere found that seedling regeneration also vary with site conditions, such as elevation, slope, and aspect (Fei et al. 2013; Bergin and Kimberley 2014; Thrippleton et al. 2016).
Regeneration of tree species respond to a variety of biotic and abiotic factors, and all effective factors play a role by changing available resources and conditions, which can be indispensable to seedling occurrence, growth, and survival (Thrippleton et al. 2016). Under changing environment, it is of rising interest that studying the responses of multiple species in temperate forests to environment and conditions and how tree species and their offspring adapt to the environment. For instance, coniferous species and their seedlings in temperate regions of the Eastern USA tend to show similar shift along latitude under climate change (Woodall et al. 2009; Zhu et al. 2012), which may result from similar response patterns to climate and environment among these species. While on a smaller scale, it needs to be addressed whether species show certain patterns responding to stand structure conditions, particularly those species with similar traits under environmental stress (e.g., shade and drought tolerance).
The Inland Northwest, USA, encompasses a large area of temperate forests with diverse coniferous species and understory vegetation. Many of these species are commonly associated, which are both ecologically significant in forest ecosystems and economically productive with premium wood and other forest products. Understanding the relationship between seedling occurrence and density of these species and multi-layer stand structure factors would assist in understanding how specific tree species respond and adapt to micro-environment in wide ranging temperate forests under climate change. Further, understanding the relationships between seedlings and stand structure and composition could have potential implications on adaptive forest management, improving regeneration of specific species and possibly maintaining forest biodiversity (Beckage and Clark 2003).
In this study, natural regeneration patterns of coniferous species in temperate forests across the Inland Northwest, USA, in response to stand structure and site conditions were investigated. Studying these coniferous species, the hypotheses include that natural regeneration is affected by both stand structure factors including overstory and understory layers and site conditions and the responses of natural regeneration of multiple species to stand factors follow certain patterns given their tolerance to environmental stress. With the assistance of modeling approaches, the objectives are to examine (1) the responses of natural regeneration to multi-layer stand factors and site conditions for multiple coniferous species, (2) natural regeneration patterns of coniferous species in response to stand factors and site conditions along shade and drought tolerance gradients in temperate forests, and (3) the significance of understory non-tree vegetation to natural regeneration of coniferous species in temperate forests.
2 Materials and methods
2.1 Study area
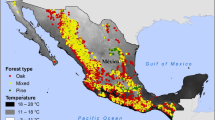
The Inland Northwest is considered the large catchment area of the Interior Columbia River Basin in the coterminous USA (Hessburg and Agee 2003). This area is characterized by varying climate and environmental conditions among five ecoregions (Fig. 1, Bailey 1995). Figure 1 displays the geographic range within ecoregions and elevation of the study area. The geographic location of sampling size is not shown because it is not publically accessible. Multiple valuable coniferous species predominate in mixed forests at different elevations of this area, including grand fir (Abies grandis (Douglas ex D. Don) Lindl.), Douglas-fir (Pseudotsuga menziesii (Mirb.) Franco), lodgepole pine (Pinus contorta Douglas ex Loudon), western redcedar (Thuja plicata Donn ex D. Don), western hemlock (Tsuga heterophylla (Raf.) Sarg.), and ponderosa pine (Pinus ponderosa Lawson & C. Lawson).
Map of study area in the Inland Northwest, USA. Since the locations of specific sampling plots are confidential, precise coordinates of sampling plots are not shown in this figure. Ecoregions codes are presented: Cascade mixed forest—Coniferous forest—Alpine meadow province (M242), Middle Rocky Mountain steppe—Coniferous forest—Alpine meadow province (M332), Northern Rocky Mountain forest—Steppe—Coniferous forest—Alpine meadow province (M333), Great plains—palouse dry steppe province (331), Intermountain semidesert province (342). The elevation is shown by digital elevation model (DEM) of 800 m resolution. In the map of USA (enclaves not shown), area within the red rectangle shows the reference location of study area in Northwest USA
2.2 Forest inventory data and data preparation
The Forest Inventory and Analysis (FIA, 2017) Program of the US Forest Service data from the four Northwestern US states (Idaho, Montana, Oregon, and Washington) were acquired. Plots with natural regeneration east of the cascade crest, western Montana, and northern and central Idaho were selected for analysis. As a result of species composition, natural dynamics (such as competition and succession) and natural disturbances (such as dry sites) in these mixed forests in the Inland Northwest, stands are covered by different degrees of canopies, including open stands, probably left timber harvesting. The FIA sampling design uses fixed-radius plots and subplots. In each FIA plot, four 0.02-ha subplots are located at the center, and 36.6 m from the center at azimuths of 360°, 120°, and 240°. The diameter at the breast height, the height, live crown ratio, and species are measured and recorded for all trees 12.7 cm and greater in DBH in the subplots. Non-tree vegetation species are identified and the vertical distribution layer and horizontal cover to the nearest 1% are measured in each subplot. Vegetation layer is classified into four levels by their heights: 0–5, 5.1–15.1, 15.2–40.6, and > 40.6 cm. A 0.001-ha microplot within each subplot is set to sample small trees, where DBH is measured for trees in 2.5–12.7 cm DBH (saplings), and a count of trees smaller than 2.5 cm (seedlings) by species. The data of this study was from the FIA census data collected between 2001 and 2015. FIA plots are remeasured every 5 to 10 years since the first census.
Data of nine conifer species and one deciduous conifer species (Larix occidentalis Nutt) (Table 1) with inventory and re-measurement were utilized to determine seedling regeneration patterns. Species were categorized into five types considering shade tolerance: very tolerant species (VT), tolerant species (T), moderately tolerant species (MT), intolerant species (I), and very intolerant species (VI) according to Niinemets and Valladares (2006). Drought tolerance of each species were also acquired by the same method. Because sampling data was collected in subplots, all calculations were conducted on subplot level. Since these species are generally widely distributed in the Inland Northwest, particularly some are symbiotic in the mixed forests, nestedness is inevitably observed in a spatial context for different reasons, such as dispersal limitation and hierarchical niche requirements (Honnay et al. 1999). Sampled at nearby locations, variables may show spatial autocorrelation within certain distance if they are distance-related or not independent (Keitt et al. 2002). In this study, samples of a species were mostly collected from subplots of different plots, which are distant. For samples collected from multiple subplots within a single plot, Moran’s I correlograms were utilized to detect spatial autocorrelation of variables between sampling subplots within a plot for each species (Fortin and Dale 2005) and the results showed that the level of spatial autocorrelation could decrease to the absence (close to a value of 0) given larger sample size than 1, which is the case in this study.
For each species, seedling density (SeedD) was scaled to stem per hectare value. Basal area of conspecific (the same species) and heterospecific species (other species) (BAs) and total basal area (BA) were calculated; tree and species quantity were summed for all tree species to calculate the Shannon’s diversity index on the stands (SHAN, Shannon 1948). Based on individual tree height, maximum and mean canopy height were calculated, along with mean live crown ratio. Tree biomass and sapling biomass were estimated by the sum of stump, bole, and top biomass of trees greater than 2.5 cm using FIA estimates (Heath et al. 2008).
Stand density index (SDI), an expression of relative stand density, was calculated by the summation method for uneven-aged stands (Shaw 2000). SDImax is the highest possible SDI for a given species. SDImax of these species were computed using FIA data of relatively pure stands with the highest possible composition by Shaw (2016, personal communication). Relative stand density index (RSDI) of a species is described and calculated as the occupation of SDI to SDImax. In a mixed stand, RSDI value was weighted based on the SDImax value and the composition proportion of each species. Classification of RSDI indicates significant thresholds in stand density (Long and Shaw 2005), from open-grown to competition, full occupancy, self-thinning, and the maximum. Relative density (RD), another indicator of stand development, was derived by combining basal area and quadratic mean diameter, Dq (Curtis 1982).
Understory vegetation species richness was calculated to indicate the possible intensity of competition. Understory non-tree vegetation were classified into three groups: SH (shrubs, subshrubs, and woody vine), FB (forbs: herbaceous, broad-leaved plants; also includes non-woody-vines, ferns), and GR (graminoids: grasses and grass-like plants). Height of each vegetation group was represented by the maximum height of the vegetation and the cover percent was the sum of horizontal cover of all vegetation in a group.
Slope, aspect, and elevation (physiographic attributes) were acquired as site condition factors. Two combined variables were added to determine the interactions among these factors and possible changes of effects: (1) slope percentage: tangent of slope times cosine of aspect (SCA); (2) the natural logarithmic function of elevation interacted with cosine of aspect (LCA) (Stage and Salas 2007; Xiang et al. 2016).
2.3 Seedling regeneration models
2.3.1 Candidate variables selection
Candidate variables included stand structure factors: canopy trees and saplings (all trees DBH > 2.5 cm) and understory non-tree vegetation and site conditions. All candidate variables are described briefly in Table 2. Data usability was tested with multivariate analysis of variance test (MANOVA) and then multicollinearity was diagnosed through a variance-inflation factor (VIF) test to avoid potential redundancy and determine the most explanatory variables. Variables with collinearity (VIF > 5) were discarded.
2.3.2 Seedling regeneration models
There could be a large amount of zero values for regeneration due to the absence of specific species in sampling sites. Traditional linear or non-linear modeling approaches may fail or result in bias when simulating and estimating the population with many zero counts (Monserud et al. 2005). Previous studies introduced a two-stage conditional logistic regression: simulating positive counts with Poisson or negative binomial functions, seedling density in this study; simultaneously using zero-inflated model to address the possible zero observations by binomial with a logit link, the occurrence of natural regeneration in this study (Bravo et al. 2008; Zuur et al. 2009). Compared to Poisson models, zero-inflated negative binomial models (ZINB) perform more satisfactorily because they are more flexible for disperse and asymmetric distributed data and with less restrictions on the variance of positive counts (Zeileis et al. 2008; Zhang et al. 2012).
Model fitting and validation were conducted with the pscl package in R (Jackman et al. 2015; R Core Team 2017). Vuong’s non-nested hypothesis test was applied to compare non-nested models for count data (Vuong 1989). Comparing a generalized linear model (model 1) and zero-inflated negative binomial model (model 2) for regeneration of each species, the negative values of Z test statistic test (Jackman et al. 2015) provided the evidence of the superiority of zero-inflated negative binomial models that they could perform more predicted probabilities for natural regeneration in this study.
Zero-inflated negative binomial models were selected for modeling natural regeneration in all sampling stands, specifically the occurrence of seedlings by zero-inflation model and the density of seedlings by negative binomial functions. Maximum likelihood was applied to estimate parameters. Akaike’s information criterion (AIC) was used for model comparison with the same dataset. Models with the lowest AIC value were retained. Relied on the number of model parameters, AIC statistics ensured the best trade-off between the number of parameters and fitting goodness for selected models. Chi-square (Χ2) independence statistics and log-likelihood ratio test were calculated to evaluate the predictive performance of models, and p values of chi-square statistics smaller than 0.05 (p < 0.05) represented good fit of the models. Besides these relative statistics, goodness of fit was exhibited by QQ plots of residuals (residuals between predicted and observed values plotted against the count).
2.4 Data availability
The data utilized in this paper is acquired from a part of inventory data of FIA conducted by USDA Forest Service, USA. Inventory data is publically accessible on the official website (https://www.fia.fs.fed.us/). Any researcher who is interested in the FIA data, like the authors of this manuscript, could download the data from the database provided on the same website (https://apps.fs.usda.gov/fia/datamart/datamart.html). Data can be acquired by state or entirely. User guide is also provided. The authors do not have the right to deposit this data on a public repository and re-share it. The authors hope this policy can be understandable.
3 Results
3.1 Model evaluation and validation
A relatively high amount of sampling stands had no occurrence of regeneration of a specific species in this study. Natural regeneration of the species included an excessive number of zeros and displayed a large variety of dispersion around the mean and skewness (Fig. 2). Based on MANOVA test and multicollinearity diagnosis, SHAN was not included in any model due to insignificance or high multicollinearity. Stand relative density, basal area, and biomass variables were discarded by model fitting because of insignificance (p > 0.05). QQ plots of model residuals (Fig. 3) were approximately linear indicating the residuals are normally distributed. The models performed well but slightly overestimated seedling density when counts were high, which could result from inadequate high count samples. The statistics of best modeling results for seedling occurrence (zero-inflation model) and seedling density (count model) of these species are displayed in Appendices Tables 5, 6, 7, 8 and 9. Specific relationships are demonstrated for seedling occurrence and density in the following sections.
3.2 Seedling occurrence, stand structure, and site conditions
Seedling occurrence of coniferous species were significantly related to stand structure factors (Table 3). Generally, significant canopy factors played consistent effects on seedling occurrence of species with different shade tolerance, particularly maximum canopy height (MaxH) was a negative factor (p < 0.001) while mean crown ratio (MeanCR) was positive (p < 0.001).
The effects of understory non-tree vegetation and slope on seedling occurrence were group specific along shade and drought tolerance gradients (p < 0.05). In specific, seedling occurrence possibility of three very shade-tolerant species (Picea engelmannii, T. plicata, T. heterophylla) was significantly negatively related to forb cover (SumCP_FB) (p < 0.001) and T. heterophylla was positively related to maximum grass height (MaxL_GR, 15.2 cm, p < 0.001) as well. Seedling occurrence of T. plicata and T. heterophylla was restricted along rising slope (p < 0.05).
As a shade-tolerant species, seedling emergence of A. grandis was significantly negatively related to understory vegetation height (MaxL, about 40 cm), especially MaxL_GR (p < 0.001), and slope (p < 0.05).
Seedling occurrence of two moderately shade-tolerant species with different drought tolerance showed converse responses to influential variables, negatively to vegetation cover (SumCP) and slope for P. menziesii (p < 0.01) while positively to grass cover (SumCP_GR) and slope for P. monticola (p < 0.001).
As a typical shade-intolerant but drought-tolerant species, P. ponderosa seedling occurrence was positively related to SumCP_FB and slope (p < 0.001).
Two very shade-intolerant species (P. albicaulis and P. contorta) showed significantly positive responses of seedling occurrence to understory vegetation (vegetation richness (VegR) and MaxL for both species (p < 0.001) and SumCP_GR for P. contorta in addition, p < 0.01). Seedling occurrence of L. occidentalis (very shade intolerant and drought intolerant) was significantly negatively correlated to VegR and SumCP_GR (p < 0.001). Seedling occurrence of P. contorta was significantly negatively related to slope (p < 0.001) while that of the other two species (P. albicaulis and L. occidentalis) was positively correlated to slope (p < 0.01).
3.3 Seedling density, stand structure, and site conditions
Seedling density of these species showed consistent response to significant factors with one exception (Table 4). Seedling density of T. plicata was positively related to MeanCR (p < 0.05) and insignificantly related to understory vegetation. Seedling density of other species was negatively affected by MeanCR (p < 0.001). MaxH and stem density of conspecific species (StemD_CSP) played significantly positive effects on seedling density of different coniferous species regardless of their shade tolerance (p < 0.01). Seedling density of the species was negatively correlated to significant understory vegetation variables and slope (p < 0.05).
4 Discussions
4.1 Significant factors of seedling regeneration
Seedling regeneration of these coniferous species show patterns responding to maximum canopy height, mean live crown ratio, stem density of conspecific trees, understory non-tree vegetation, and slope but does not differentiate significantly with stand basal area, biomass, stand quadratic diameter, or relative density, indicating regeneration of coniferous species is affected by structure factors of multiple levels in the mixed temperate forests in this study.
4.2 Conifer seedling occurrence pattern along shade and drought tolerance gradients
Seedling occurrence of these coniferous species shows patterns with complexity responding to significant stand factors and site conditions, in which the complexity is represented by varying effects of a single effective factor on seedling occurrence of different species. First, germination could be prolonged even inhibited due to deficient sun light reaching the ground intercepted by canopy. Trees tend to increase height growth for acquiring more light rather than reproducing seeds for regeneration under dense canopy, which is also reported in other temperate areas, such as British Columbia area of North America (Li and Burton 1994). Positive relationship between high live crown ratio and seedling establishment may result from extending the possibility of seed production (Bilan 1960; Kneeshaw and Burton 1997). Seedling occurrence of conifer species may improve its probability with more available seeds. Second, the responses of seedling occurrence to understory vegetation vary with species along shade and drought tolerance gradients. For the species with positive shade tolerance, competing understory vegetation may inhibit seed germination and seedling establishment because horizontal spread and height growth of vegetation can occupy potential growth space and consume soil moisture. Consequently, insufficient space and poor soil conditions may delay even cease the germination of conifer seeds and reduce seedling occurrence possibility, which is consistent to the observations of different species in other temperate forests worldwide (e.g., Noble and Alexander 1977; Krasowski and Owens 1991; Royo and Carson 2006; Kern et al. 2013). Partial shade and favorable soil temperature is also necessary for seedling occurrence of T. heterophylla (Edwards 1976), which is indicated by its positive relationship with tall grass of understory layer in this study.
Moderately shade-tolerant species, which are differentiated by different degrees of drought tolerance showed contrary responses of seedling occurrence to understory vegetation. P. monticola limitedly occurs in moist and cool sites because it is intolerant to drought (Feurtado et al. 2004), and grass cover is found to keep favorable soil moisture and temperature for successful seedling emergence in this study. P. menziesii spans broader geographic range while proper temperature is also required for regeneration in cool areas (Lavender 1984). In the current study, vegetation cover, which probably leads to low soil temperature is not favored for P. menziesii occurrence.
P. ponderosa is able to regenerate in dry areas; however, drought is still a major obstacle to the regeneration (Larson and Schubert 1969; Brown and Wu 2005). In this circumstance, understory vegetation cover may provide partial moist conditions for establishment of this species. P. albicaulis can associate with diverse undergrowth species because this species is a keystone species in the upper subalpine ecosystems (Mahalovich et al. 2006), which is also implied by the strongest positive connection of seedling occurrence and vegetation factors among all species in this study. Moist soil with low temperature provide favored soil conditions for emergence of P. contorta (Stuart et al. 1989). Compared to more exposed areas, vegetation could keep soil moister and temperature lower in the study area. Being intolerant to environmental stress such as shade and drought, L. occidentalis occurrence is sensitive to micro-environment change (Oswald and Neuenschwander 1993), such as moisture insufficiency driven by competition from vegetation layer shown in this study.
Site condition factors, such as slope and aspect can significantly affect seedling occurrence of tree species through their effects on obtainable light intensity, soil moisture, and nutrients (Ferguson et al. 1986; Mühlenberg et al. 2012). Areas with greater slopes are normally characterized with shallow and dry soil and possibly high soil temperature due to exposure (Kang et al. 2000). These conditions probably limit seedling occurrence of shade-tolerant species found in this study. On contrary, shade-intolerant species improve their seedling occurrence along rising slopes with more available sun light, except for P. albicaulis because this species is commonly limited on subalpine elevations (Mahalovich et al. 2006), where steep slopes may fail to keep favorable soil nutrients.
4.3 Conifer seedling density pattern along shade and drought tolerance gradients
Adaptation of tree seedlings to stand structure and site conditions is inevitable in multi-layer mixed forests. Higher seedling densities of these coniferous species tend to occur under canopy with more conspecific trees, less understory vegetation, and lower slopes in temperate forests in the Inland Northwest, USA. Coniferous species are strongly adaptive to overstory height growth (Lewis et al. 2000) so that tree height does not perform as a limiting factor of seedling density. The presence of conspecific trees is beneficial to seedlings probably resulting from less interspecific competition, which is also reported in temperate forests of Eastern Asia (e.g. Fei et al. 2013).
Competition from overstory and understory may be one of the most influential factors limiting seedling density of coniferous species in this study, especially for species of less shade and drought tolerance. Contrary effect of live crown ratio on T. plicata compared to other conifers, which may result from its strong resistance to different light intensity. Generally, seedlings prefer sunlight to partial shade when grow larger even some species are shade tolerant (Adams and Mahoney 1991; Carter and Klinka 1992), which implies large crown of overstory becomes an obstacle to available light for seedlings in this study.
As seedlings grow, the competition for essential resources from surrounding understory vegetation is becoming more predominant when seedling roots can be still shallow so that the effects of understory layer on the regeneration is considered to last for a long period (Balandier et al. 2006). Seedling density of coniferous species is potentially restricted due to insufficient resources caused by competition from various neighboring vegetation (Zobel and Antos 1991; Balandier et al. 2006; Vyse et al. 2006).
Unstable solar energy and temperature flux occur frequently on slopes (Ferguson et al. 1986), which could negatively affect plant growth. It is implied by that higher seedling densities at lower slope areas for these coniferous species in this study.
4.4 Significance of understory vegetation to natural regeneration
Understory vegetation is considered affected by tree population in their one-way interaction by some previous studies (e.g., Li et al. 2012). However, understory vegetation is increasingly demonstrated to have a wide range of effects on regeneration through competing for light and other resources in the mixed forests of different regions (Balandier et al. 2006; Royo and Carson 2006; Thrippleton et al. 2016). In this current study, understory vegetation is indicated to show significant effects on both seedling occurrence and density of multiple coniferous species in the forests. In the study area, timber harvesting is widely conducted in managed stands (Jurgensen et al. 1997). Under open gaps, seedlings and understory vegetation could obtain more resources and growth space, which may lead to that potential effect (competition or facilitation) of understory vegetation can be altered (Knapp et al. 2016). Thus, it is important to identify the significance of understory vegetation to natural regeneration and potential tree population dynamics on both covered and open stands in the mixed forests.
4.5 Potential implications on adaptive forest management
Temperate forests compose significant parts in forest ecosystems, which also contribute predominantly to global timber production (Sohngen et al. 1999). Understanding natural regeneration patterns of coniferous species in temperate forests responding to stand composition, structure, and micro-environment is highly meaningful and desirable in adaptive forest management. The major objective of forest management in temperate forests is to improve and maintain natural regeneration and growth of conifer species by controlling the spread of competing plants. Competition control and thinning are probably effective to release more growth space, reduce competition intensity, and maintain desired forest structure (Mallik 2003). In this study, to improve seedling occurrence of a species, maintenance or removal of neighboring understory vegetation could be applied based on the relationships between seedling occurrence and different types of vegetation for specific species. Thinning on heterospecific tree species and control on specific competing understory vegetation could be employed to maintain and increase seedling density of coniferous species. For instance, understory grasses and forbs could be removed to reduce the competition for coniferous species, especially shade-intolerant and very shade-intolerant species. Revealing the effects of overstory trees and understory vegetation on seedling occurrence and density enables proper management for successful and sustainable regeneration and treatment for maintaining forest biodiversity in temperate forests. Under changing environment, artificial regeneration may be supplementary management practice in areas of insufficient natural regeneration.
5 Conclusions
Natural regeneration of common coniferous species in temperate mixed forests in the Inland Northwest, USA, was studied through the relationships with stand structure factors and site conditions. Overstory, understory vegetation, and slope were found driving factors for seedling occurrence and density of these coniferous species. Canopy factors played consistent effects on seedling occurrence while the effects of understory vegetation and slope varied along shade tolerance and drought gradients. Seedling density was controlled by competition from both canopy and understory vegetation. Our findings demonstrated that the significance of understory vegetation to natural regeneration of tree species in temperate forests through affecting occurrence probability and competing for essential resources. Thinning on heterospecific tree species for canopy density regulation and reducing or removing competing vegetation of seedlings such as cutting could be applied to maintain and improve seedling density of the coniferous species studied in this area. The modeling methods may be applied to study natural regeneration of coniferous species and the relationship with stand composition, structure, and site conditions in broad temperate forests.
References
Adams DL, Mahoney RL (1991) Effects of shade and competing vegetation on growth of western redcedar regeneration. West J Appl For 6:21–22
Al-Namazi AA, El-Bana MI, Bonser SP (2017) Competition and facilitation structure plant communities under nurse tree canopies in extremely stressful environments. Ecol Evol 7:2747–2755. https://doi.org/10.1002/ece3.2690
Bailey RG (1995) Description of ecoregions of the United States, 2nd ed. Misc. Publ. 1391. USDA Forest Service, Washington, DC, p 108
Balandier P, Collet C, Miller JH, Reynolds PE, Zedaker SM (2006) Designing forest vegetation management strategies based on the mechanisms and dynamics of crop tree competition by neighbouring vegetation. Forestry 79:3–27. https://doi.org/10.1093/forestry/cpi056
Battaglia MA, Mou P, Palik B, Mitchell RJ (2002) The effect of spatially variable overstory on the understory light environment of an open-canopied longleaf pine forest. Can J For Res 32:1984–1991. https://doi.org/10.1139/X02-087
Beckage B, Clark JS (2003) Seedling survival and growth of three forest tree species: the role of spatial heterogeneity. Ecol 84(7):1849–1861. https://doi.org/10.1890/0012-9658
Bergin DO, Kimberley MO (2014) Factors influencing natural regeneration of totara (Podocarpus totara D.Don) on grazed hill country grassland in Northland, New Zealand. NZ J For Sci 44:13. https://doi.org/10.1186/s40490-014-0013-8
Bilan MV (1960) Stimulation of cone and seed production in pole-size loblolly pine. For Sci 6:207–220
Bonanomi G, Incerti G, Mazzoleni S (2011) Assessing occurrence, specificity, and mechanisms of plant facilitation in terrestrial ecosystems. Plant Ecol 212:1777–1790. https://doi.org/10.1007/s11258-011-9948-5
Bravo F, Pando V, Ordóñez C, Lizarralde I (2008) Modelling ingrowth in mediterranean pine forests: a case study from scots pine (Pinus sylvestris L.) and mediterranean maritime pine (Pinus pinaster Ait.) stands in Spain. For Syst 17:250–260. https://doi.org/10.5424/srf/2008173-01039
Brown PM, Wu R (2005) Climate and disturbance of episodic tree recruitment in a southwestern ponderosa pine landscape. Ecology 86:3030–3038. https://doi.org/10.1890/05-0034
Callaway RM (2007) Positive interactions and interdependence in plant communities. Houten, Netherlands: Springer Netherlands
Carter RE, Klinka K (1992) Variation in shade tolerance of Douglas fir, western hemlock, and western red cedar in coastal British Columbia. For Ecol Manag 55:87–105. https://doi.org/10.1016/0378-1127(92)90094-P
Comita LS, Aguilar S, Pérez R, Lao S, Hubbell SP (2007) Patterns of woody plant species abundance and diversity in the seedling layer of a tropical forest. J Veg Sci 18:163–174. https://doi.org/10.1658/1100-9233
Curtis RO (1982) A simple index of stand density for Douglas-fir. For Sci 28:92–94
Edwards DGW (1976) Seed physiology and germination in western hemlock. In: Atkinson WA, Zasoski RJ (eds) Proceedings, Western Hemlock Management Conference. University of Washington, Seattle, Washington, pp 87–102
Ferguson DE, Stage AR, Boyd RJ (1986) Predicting regeneration in the grand fir-cedar-hemlock ecosystem of the Northern Rocky Mountains. For Sci 32:1–42
Feurtado JA, Ambrose SJ, Cutler AJ, Ross ARS, Abrams SR (2004) Dormancy termination of western white pine (Pinus monticola Dougl. Ex D. Don) seeds is associated with changes in abscisic acid metabolism. Planta 218:630–639. https://doi.org/10.1007/s00425-003-1139-8
Fei Y, Wang DX, Shi XX, Yi XF, Huang QP, Hui YN (2013) Effects of environmental factors on tree seedling regeneration in a pine-oak mixed forest in the Qinling Mountains, China. J Mount Sci 10:845–853. https://doi.org/10.1007/s11629-013-2548-1
FIA (2017) Forest Inventory and Analysis Database. U.S. Department of Agriculture, Forest Service, Northern Research Station, St. Paul
Forbes A, Norton DA, Carswell FE (2016) Tree fern competition reduces indigenous forest tree seedling growth within exotic Pinus radiata plantations. For Ecol Manag 359:1–10. https://doi.org/10.1016/j.foreco.2015.09.036
Fortin MJ, Dale MRT (2005) Spatial analysis-a guide for ecologists. Cambridge University Press
Gavinet J, Prévosto B, Fernandez C (2016) Do shrubs facilitate oak seedling establishment in Mediterranean pine forest understory? For Ecol Manag 381:289–296. https://doi.org/10.1016/j.foreco.2016.09.045
Gilliam FS (2007) The ecological significance of the herbaceous layer in temperate forest ecosystems. Bioscience 57:845–858. https://doi.org/10.1641/B571007
Gordon DR, Rice KJ (2000) Competitive suppression of Quercus douglasii (Fagaceae) seedling emergence and growth. Am J Bot 87:986–994
Grubb PJ (1977) The maintenance of species-richness in plant communities: the importance of the regeneration niche. Bio Rev 52:107–145
Hart SA, Chen HYH (2006) Understory vegetation dynamics of North American boreal forests. Crit Rev Plant Sci 25:381–397. https://doi.org/10.1080/07352680600819286
Heath LS, Hansen MH, Smith JE, Miles PD (2008) Investigation in calculating tree biomass and carbon in the FIADB using a biomass expansion factor approach. Forest Inventory and Analysis (FIA) Symposium; October 21–23. UT, Park City, p 2008
Hessburg PF, Agee JK (2003) An environmental narrative of Inland Northwest United States forests, 1800-2000. For Ecol Manag 178:23–59. https://doi.org/10.1016/S0378-1127(03)00052-5
Honnay O, Hermy M, Coppin P (1999) Nested plant communities in deciduous forest fragments: species relaxation or nested habitats? Oikos 84:119–129. https://doi.org/10.2307/3546872
Jackman S, Tahk A, Zeileis A, Maimore C, Fearon J (2015) Package ‘pscl’: Political Science Computational Laboratory, Stanford University. URL http://pscl.stanford.edu/
Jensen AM, Götmark F, Löf M (2012) Shrubs protect oak seedlings against ungulate browsing in temperate broadleaved forests of conservation interest: a field experiment. For Ecol Manag 266:187–193. https://doi.org/10.1016/j.foreco.2011.11.022
Jensen AM, Löf M (2017) Effects of interspecific competition from surrounding vegetation on mortality, growth and stem development in young oaks (Quercus robur). For Ecol Manag 392:176–183. https://doi.org/10.1016/j.foreco.2017.03.009
Kang S, Kim S, Oh S, Lee D (2000) Predicting spatial and temporal patterns of soil temperature based on topography, surface cover and air temperature. For Ecol Manage 136:173–184. https://doi.org/10.1016/S0378-1127(99)00290-X
Jurgensen MF, Harvey AE, Graham RT, Page-Dumroese DS, Tonn JR, Larsen MJ, Jain TB (1997) Impacts of timber harvesting on soil organic matter, nitrogen, productivity, and health of Inland Northwest forests. For Sci 43(2):234–251
Keitt TH, Bjørnstad ON, Dixon PM, Citron-Pousty S (2002) Accounting for spatial pattern when modelling organism-environment interactions. Ecography 25:616–625. https://doi.org/10.1034/j.1600-0587.2002.250509.x
Kern CC, D’Amato AW, Strong TF (2013) Diversifying the composition and structure of managed, late-successional forests with harvest gaps: what is the optimal gap size? For Ecol Manag 304:110–120. https://doi.org/10.1016/j.foreco.2013.04.029
Knapp BO, Wang GG, Walker JL, Hu HF (2016) Using silvicultural practices to regulate competition, resource availability, and growing conditions for Pinus palustris seedlings underplanted in Pinus taeda forests. Can J For Res 46:902–913. https://doi.org/10.1139/cjfr-2016-0066
Kneeshaw DD, Burton PJ (1997) Canopy and age structures of some old sub-boreal Picea stands in British Columbia. J Veg Sci 8:615–626. https://doi.org/10.2307/3237365
Krasowski MJ, Owens JN (1991) Growth and morphology of western redcedar seedlings as affected by photoperiod and moisture stress. Can J For Res 21:340–352. https://doi.org/10.1139/x91-042
Larson MM, Schubert GH (1969) Effect of osmotic water stress on germination and initial development of ponderosa pine seedlings. For Sci 15:30–36
Lavender DP (1984) Plant physiology and nursery environments: Interactions affecting seedling growth. In: Duryea ML, Landis TD (eds) Forest nursery manual: production of bare-rooted seedlings. Martinus Nijhoff/Dr. W. Junk Publishers, Boston, pp 133–141
Lewis JD, McKane RB, Tingey DT, Beedlow PA (2000) Vertical gradients in photosynthetic light response within an old-growth Douglas-fir and western hemlock canopy. Tree Physiol 20:447–456. https://doi.org/10.1093/treephys/20.7.447
Lewis SL, Tanner EV (2000) Effects of above- and belowground competition on growth and survival of rain forest tree seedlings. Ecology 81:2525–2538. https://doi.org/10.1890/0012-9658
Li MH, Du Z, Pan HL, Yan CF, Xiao WF, Lei JP (2012) Effects of neighboring woody plants on target trees with emphasis on effects of understorey shrubs on overstorey physiology in forest communities: a mini-review. Commun Ecol 13:117–128. https://doi.org/10.1556/ComEc.13.2012.1.14
Li XJ, Burton PJ (1994) Interactive effects of light and stratification on the germination of some British Columbia conifers. Can J Bot 72:1635–1646. https://doi.org/10.1139/b94-201
Long JN, Shaw JD (2005) A density management diagram for even-aged ponderosa pine stands. West J Appl For 20:205–215
Mahalovich MF, Burr KE, Foushee DL (2006) Whitebark pine germination, rust resistance, and cold hardiness among seed sources in the Inland Northwest: planting strategies for restoration. USDA Forest Service Proceedings RMRS-P-43
Mallik A (2003) Conifer regeneration problems in boreal and temperate forests with ericaceous understory: role of disturbance, seedbed limitation, and keytsone species change. Crit Rev Plant Sci 22(3–4):341–366. https://doi.org/10.1080/713610860
Monserud RA, Ledermann T, Sterba H (2005) Are self-thinning constraints needed in a tree-specific mortality model? For Sci 50:848–858
Mühlenberg M, Appelfelder J, Hoffmann H, Ayush E, Wilson KJ (2012) Structure of the montane taiga forests of West Khentii, Northern Mongolia. J For Sci 58:45–56
Niinemets Ü, Valladares F (2006) Tolerance to shade, drought, and waterlogging of temperate northern hemisphere trees and shrubs. Ecol Monogr 76:521–547. https://doi.org/10.1890/0012-9615
Noble DL, Alexander RR (1977) Environmental factors affecting natural regeneration Engelmann spruce in the Central Rocky Mountains. For Sci 23:420–429
Oswald BP, Neuenschwander LF (1993) Microsite variability and safe site description for western larch germination and establishment. Bull Torrey Bot Club 120:148–156. https://doi.org/10.2307/2996944
R core team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Royo AA, Carson WP (2006) On the formation of dense understory layers in forests worldwide: consequences and implications for forest dynamics, biodiversity, and succession. Can J For Res 36:1345–1362. https://doi.org/10.1139/x06-025
Shannon CE (1948) A mathematical theory of communication. Bell Syst Tech J 27:379–423 and 623–656
Shaw JD (2000) Application of stand density index to irregularly structure stands. West J Appl For 15:40–42
Sohngen B, Mendelsohn R, Sedjo R (1999) Forest management, conservation, and global timber markets. Am J Agric Econ 81(1):1–13. https://doi.org/10.2307/1244446
Stage AR, Salas C (2007) Interactions of elevation, aspect, and slope in models of forest species composition and productivity. For Sci 53:486–492
Stuart JD, Agee JK, Gara RI (1989) Lodgepole pine regeneration in an old, self-perpetuating forest in a south central Oregon. Can J For Res 19:1096–1104. https://doi.org/10.1139/x89-166
Thrippleton T, Bugmann H, Kramer-Priewasser K, Snell RS (2016) Herbaceous understorey: an overlooked player in forest landscape dynamics? Ecosystems 19:1240–1254. https://doi.org/10.1007/s10021-016-9999-5
Tilman D (1994) Competition and biodiversity in spatially structured habitats. Ecology 75:2–16. https://doi.org/10.2307/1939377
Vandenberghe C, Freléchoux F, Gadallah F, Buttler A (2006) Competitive effects of herbaceous vegetation on tree seedling emergence, growth and survival: does gap size matter? J Veg Sci 17:481–488. https://doi.org/10.1111/j.1654-1103.2006.tb02469.x
Vuong QH (1989) Likelihood ratio tests for model selection and non-nested hypotheses. Econometrica 57:307–333. https://doi.org/10.2307/1912557
Vyse A, Ferguson C, Simard SW, Kano T, Puttonen P (2006) Growth of Douglas-fir, lodgepole pine, and ponderosa pine seedlings under planted in a partially-cut, dry Douglas-fir stand in south-central British Columbia. For Chron 82:723–732. https://doi.org/10.5558/tfc82723-5
Walters MB, Farinosi EJ, Willis JL, Gottschalk KW (2016) Managing for diversity: harvest gap size drives complex light, vegetation, and deer herbivory impacts on tree seedlings. Ecosphere 7:e01397. https://doi.org/10.1002/ecs2.1397
Woodall CW, Oswalt CM, Westfall JA, Perry CH, Nelson MD, Finley AO (2009) An indicator of tree migration in forests of the eastern United States. For Ecol Manag 257:1434–1444. https://doi.org/10.1016/j.foreco.2008.12.013
Xiang W, Lei XD, Zhang XQ (2016) Modelling tree recruitment in relation to climate and competition in semi-natural Larix-Picea-Abies forests in Northeast China. For Ecol Manag 382:100–109. https://doi.org/10.1016/j.foreco.2016.09.050
Zeileis A, Kleiber C, Jackman S (2008) Regression models for count data in R. J Sat Softw 27:1–25. https://doi.org/10.18637/jss.v027.i08
Zhang XQ, Lei YC, Cai D, Liu F (2012) Predicting tree recruitment with negative binomial mixture models. For Ecol Manag 270:209–215. https://doi.org/10.1016/j.foreco.2012.01.028
Zhu K, Woodall CW, Clark JS (2012) Failure to migrate: lack of tree range expansion in response to climate change. Glob Change Bio 18:1042–1052. https://doi.org/10.1111/j.1365-2486.2011.02571.x
Zobel DB, Antos JA (1991) Growth and development of natural seedlings of Abies and Tsuga in old-growth forest. J Ecol 79:985–998. https://doi.org/10.2307/2261093
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York, pp 269–273
Acknowledgements
The authors would like to sincerely thank FIA for sharing sampling data and Dr. John Shaw for the help in maximum stand density index calculation. The efforts of editors and anonymous reviewers are highly appreciated.
Funding
This material is based upon work that is supported by the University of Idaho, College of Natural Resources and the National Institute of Food and Agriculture, US Department of Agriculture, McIntire Stennis project under accession number 1008381.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Andreas Bolte
Contribution of the co-authors
Chenchen Shen designed the experiment, run the data process and analysis, and wrote the paper; Andrew S. Nelson supervised the work, provided important advice, and helped in reviewing and revising the paper.
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Shen, C., Nelson, A.S. Natural conifer regeneration patterns in temperate forests across the Inland Northwest, USA. Annals of Forest Science 75, 54 (2018). https://doi.org/10.1007/s13595-018-0724-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13595-018-0724-8