Abstract
The positive response of grafting by tolerant rootstocks or scion-stock interactions on yield and fruit traits of tomatoes under saline conditions is attributed to several physiological and biochemical changes. In this study, we investigated some tolerance mechanisms by which grafting on wild rootstocks in tomatoes can prevent or minimize the effects of salt stress in plants under hydroponics conditions. Two tomato cultivars H2274 and Galaxy were grafted onto three S. pimpinellifolium, three S. habrochaites, S. lycopersicum L. × S. pimpinellifolium and S. lycopersicum L. × S. Habrochaites hybrid tomato genotypes. Plants were grown in hydroponic culture at two electrical conductivity (EC) levels (control at 1.5 dSm-1 and salt at 8.0 dSm-1). Salt stress led to a significant reduction in biomass growths of both grafted and nongrafted tomatoes. However, the plants that are least affected by salt stress are those grafted on wild tomato rootstocks. Leaf nutrient contents were significantly affected by rootstocks under both control and salt stress conditions. In this study, under saline conditions, plants grafted on wild rootstocks had higher N, P, K, Ca, Mg, S, Mn, Fe, Zn and B contents in leaf tissues and lower Na and Cl contents than ungrafted plants. Biochemical and physiological results revealed that S. pimpinellifolium and S. habrochaites have inherited salt tolerance from their genetic background. These wild tomato genotypes can be used as rootstocks in tomato breeding programs to develop salt-tolerant tomatoes or in grafting techniques under saline irrigation conditions.
Similar content being viewed by others
1 Introduction
In agricultural production worldwide, biotic and abiotic stress factors consistently lead to crop losses and reduced product quality. Nevertheless, abiotic stresses are considered to be the primary factor for yield loss in plants, up to about 70% (Bauchet et al. 2012; Ansari et al. 2019; Behera et al. 2022). Salinity is one of the most important abiotic stress factors that prevent the expected yield in crop production worldwide (Munns and Tester 2008). Salinity in soil or water is a serious threat to plant growth that prevents plants from achieving their genetic potential (Yamaguchi and Blumwald 2005; Zhu et al. 2008) Salinity annually damages about 20% of the world’s crops grown under irrigation (Arora 2019). Salinity has been reported to disrupt the physiological and biochemical processes of plants, causing changes in morphological characteristics that eventually lead to yield loss (Ali et al. 2021; Hasanuzzaman and Fujita 2022). Salt stress causes a reduction in shoot and root biomass, root length, root volüme, stem diameter and leaf area in vegetables (Ulas et al. 2020; Göçer et al. 2021; Aydın and Yetişir 2022). Physiologically and biochemically, it decreases chlorophyll, carotenoid and polysaccharide levels, stomatal conductivity, photosynthetic activities and increases the accumulation of reactive oxygen species in plants (Brugnoli and Lauteri 1991; Rubaye et al. 2020; Aydın and Yetişir 2022; Kesawat et al. 2023). In addition to the difficulty of transporting water under salt stress, the plant has to deal with salt ions (Na+ and Cl−) that are toxic at high concentrations (Summart et al. 2012; Hou et al. 2022).
Tomato (Solanum lycopersicon Mill.), a member of Solanaceae is one of the most-produced vegetables around the world and also is one of the most economically important vegetables, grown in many countries in the open and greenhouses using soil and soilless techniques. Salinity is a significant threat to tomato cultivation. It causes considerable reductions in tomato growth and yield (Kiferle et al. 2022; Raziq et al. 2022). Most tomato cultivars are considered moderately susceptible to salt stress, which affects seed germination and the vegetative and reproductive stages of growth (Ali et al. 2021). Since improving saline soils is very costly and difficult, salt-resistant or tolerant varieties/rootstocks need to be developed for these types of soils. One of the approaches to improve the performance of commercial cultivars susceptible to salinity in plants is the technique of grafting the target plant with other salt tolerant plants (Keatinge et al. 2014). The grafting of fruiting vegetables is now a common technique for developing salt-tolerant plants, in which both the rootstock and the scion influence the salt tolerance of the grafted plants (Etehadnia et al. 2008; Colla et al. 2013). Previous studies have reported that grafting tomatoes increases plant vigor, provides earlier maturity, and has a positive effect on resistance to stress factors, depending on which rootstock genotypes are used (Martorana et al. 2007; Di Gioia et al. 2013; İşeri et al. 2015).
In contrast to cultivated tomatoes (S. lycopersicum L.), which are moderately sensitive to salt stress, some wild species are reported to be resistant to salt (Foolad 2004; Rao et al. 2013). Tomato can be grafted onto intra/interspecies hybrids of other species in the Solanaceae family. Wild tomato species are a rich source of the genes and traits needed to increase resistance to various biotic and abiotic stresses. Major biotic and abiotic resistance genes for different stress factors such as viruses, fungi, bacteria, nematodes and salt and drought tolerant are mainly derived from wild tomato species and introgressed into cultivated tomatoes (Foolad 2004; Ji et al. 2009; Robbins et al. 2009; Hutton et al. 2012; Ebert and Schafleitner 2015; Szymański et al. 2020; Conti et al. 2023). Genetic variability for salt tolerance traits is limited in domesticated tomatoes, whereas wild Solanum species, such as S. pimpinellifolium, S. habrochaites, S. peruvianum, S. chilense and S. pennellii, have been reported to be a source of salt tolerance (Frary et al. 2010; Gharbi et al. 2017; Kashyap et al. 2020; Ali et al. 2021). However, these wild species could be exploited as salt-resistant rootstocks for grafting susceptible but high yielding commercial tomato cultivars (Voutsela et al. 2012). Generally excludes the salt ions, most wild types accumulate higher concentrations of Na+ and Cl− in the leaves (Albaladejo et al. 2017). Using salt-tolerant species as rootstocks can protect sensitive species from the deleterious effects of salinity. Thus, the objective of the present study was to investigate whether grafting with different wild tomato rootstocks could improve the salt tolerance of tomato scions and to determine the physiological and nutritional responses induced by the rootstocks under different EC levels.
2 Materials and methods
2.1 Plant material, treatments, and experimental design
A hydroponic experiment was conducting during 2022–2023 growing season in a Kırsehir Ahi Evran University, Agricultural Research and Application Greenhouse, Kırsehir, Turkey. Two tomato cultivars Galaxy (Sakata Seed Southern Africa (Pty) Ltd.) and H2274 were used as scion and eight wild tomatoes were used as rootstock (Table 1). The hybrid tomato genotypes were developed in our tomato rootstock development project. The seeds were sown in multipots in a mixture of peat (pH: 6.0–6.5) and perlite in a 2:1 ratio and then the appropriate seedlings were selected for the grafting process using the procedure of “tube grafting” described by Lee and Oda (2010), while non-grafted tomato used as control plants. The seedlings were transplanted to 136 L plastic pots after cleaning from the growth substrate by washing them with tap water. The cultivation solution was constantly aerated with a pump. The experiment was conducted with two different EC levels (1.5 dSm-1 and 8 dSm-1). The nutrient solution contained 1125 µM Ca(N03)2, 375 µM (NH4)2SO4 750 µM K2SO4, 650 µM MgSO4, 500 µM KH2PO4, 10 µM H3BO3, 0.5 µM MnSO4, 0.5 µM ZnSO4, 0.4 µM CuSO4, 0.4 µM MoNa2O4 and 80 µM Fe EDDHA (Hoagland and Arnon 1950). The experiment was designed according to the randomized plot design with 3 replications and 3 plants in each replication. The study was continued for 30 days under controlled greenhouse conditions (22–24 °C day /16–18 °C night and 60% relative humidity).
2.2 Plant growth measurements
After four weeks of growing, plants were harvested and separated into shoots and roots. Main stem length (cm) was measured using a meter rule. To determine shoot and root dry weight (g), plant materials were dried in a forced-air oven for 48 h at 70 °C. The root length, of the plants was determined by using the special software program WinRHIZO (Win/Mac RHIZO Pro V. 2002c Regent Instruments Inc. Canada). Total leaf area (cm2 plant–1) and healthy and damaged leaf area (%) of a plant were measured by WinDIAS Leaf Image Analysis System (WinDIAS 3 Rapid System, Delta-T Devices, Cambridge, U. K.).
2.3 Determination of chlorophyll İndex
The Minolta SPAD-502 chlorophyll meter was used to take SPAD readings. During the growth period, fully expanded leaves of whole plants for each treatment were twice measured for SPAD.
2.4 Determination of ion leakage
Ion leakage (IL) of three replication was measured by using the method described by Flint et al. (2011). Samples were cut into equal size pieces (0.5 g per replication) and placed in a test tube containing 10 mL of distilled water, and at 45 °C for 30 min in a water bath. The initial conductance of the solution was measured using a conductivity meter (model-146, Systronics India Limited, Mumbai, India). The tubes were then kept in a boiling water bath for 10 min then cooled to room temperature, and their conductivity was measured once again. IL (%) was calculated by following formula. IL= (initial EC/final EC) × 100.
2.5 Nutrient analysis
After harvest, fresh plant material was divided into two parts. One part was frozen in liquid nitrogen and stored at 80 °C for later use. The remaining fresh plant material was dried at 70 °C for 24 h. For determination of N, P, K, Ca, Mg, S, Mn, Fe, Zn, B, Cu, Na and Cl concentrations, 100 mg dried plant material was extracted by one hour boiling in 5 ml MilliQ. The solution was filtered through 0.2 mm filters (Whatman, England) and N, P, K, Ca, Mg, S, Mn, Fe, Zn, B, Cu, Na and Cl contents in the filtrate were analyzed using high-performance liquid chromatography (HPLC, Shimadzu Japan). The HPLC system was equipped with a ø 4.6 mm 6125 mm Shodex IC YS-50 column (Showa Denko). As an eluent, 4.0 mM methane sulfonic acid was used in HPLC graded H2O (J.T. Baker, The Netherlands) with a flow rate of 1 mL min-1. Final ion concentrations in the filtrate were calculated according to a calibration curve.
2.6 Statistical analysis
The data were analyzed with the SAS Statistical Software (SAS 9.0, SAS Institute Inc., Cary, NC, USA). A two-factorial analysis of variance was performed to study the effects of salinity (NaCl), rootstock, scion, salt and interactions on the variables. Levels of significance are represented by * p < 0.05, ** p < 0.01, *** p < 0.001, and ns means not significant. Differences between means were analyzed using the Duncan Multiple Test (p < 0.05). Classification of genotypes was achieved by principal component analysis (PCA) using XLSTAT software (XLSTAT, New York, USA).
3 Results and discussion
The results of the main stem length, shoot dry weight and root dry weight at the of graft combinations in different NaCl levels (1.5 and 8 dSm-1) were shown in Table 2. Main stem length was significantly (p < 0.001) affected by scion, rootstock, NaCl levels, scion × rootstock, scion × NaCl levels and rootstock × NaCl levels interactions while main stem length was not affected by rootstock × scion × NaCl levels interactions. Under control conditions, the longest main stem length was recorded in SP5/Galaxy, SP4/Galaxy and SH1/Galaxy graft combinations and SH3/H2274, SH5/H2274, SP4/H2274, SP3/H2274 and ungrafted H2274 graft combinations had the shortest main stem length. In saline conditions the highest main stem length is measured in SP4/Galaxy (39.33 cm plant–1) and SP5/Galaxy (39.33 cm plant–1) graft combinations. The lowest main stem length in saline conditions was measured in the graft combinations SH3/H2274 (17.67 cm plant–1) and SH5/H2274 (18.67 cm plant–1). All graft combinations in Group I (all graft combinations grafted with the H2274 scion), except the L×SH3/H2274 and SH1/H2274 graft combinations, showed better tolerance than the ungrafted H2274. In the comparison of the control conditions and the salt application plants, the shoot lengths in Group II. (all graft combinations grafted with the Galaxy scion) decreased less than those of the ungrafted (Galaxy) plants. Shoot dry weight was significantly (p < 0.001) affected by rootstock, scion NaCl levels, rootstock × scion, rootstock × NaCl, scion × NaCl and rootstock × scion × NaCl levels interactions. Under control conditions, SP4/Galaxy (19.96 g plant–1) and SP5/Galaxy (19.94 g plant–1) graft combinations produced significantly higher shoot dry weight, whereas non grafted H2274 (6.30 g plant–1) significantly produced the minimum shoot dry weight. Under saline conditions, SH1/Galaxy (5.57 g plant–1), SP5/Galaxy (5.33 g plant–1) and SP4/Galaxy (5.28 g plant–1) graft combinations produced the highest shoot dry weight, respectively, while the ungrafted H2274 (2.24 g plant–1) plant produced the lowest shoot dry weight, as in the control conditions (Fig. 1).
Root dry weight was significantly (p < 0.001) affected by rootstock, scion NaCl levels, rootstock × scion, rootstock × NaCl, scion × NaCl and rootstock×scion × NaCl levels interactions (p < 0.01). Under control and salinity conditions, the Galaxy scion had a strong root system compared to the H2274 scion. Under control conditions, the SH1/Galaxy (4.02 g plant–1) graft combination produced the highest root dry weight, while the SP5/Galaxy (2.70 g plant–1) graft combination produced the highest root dry weight under saline conditions. The lowest root dry weight under control (1.39 g plant–1) and saline (1.09 g plant–1) conditions were produced by ungrafted H2274 plants. The mean of main stem length, shoot dry weight and root dry weight parameters of all graft combinations using Galaxy scion in control and saline conditions were higher than the mean of graft combinations using H2274 scion. Salt application reduced main stem length and shoot and root dry weight in both grafted and non-grafted plants. The decline in plant growth may be caused by osmotic stress (indirect negative effect of salinity), which limits water uptake and determines metabolic activities(Singh et al. 2012; Brdar et al. 2015). This problem is followed by ion toxicity (Na+ and Cl− ), which can cause inhibition of enzymes or alteration of hormonal activities, leading to a decrease in vegetative growth(Gomes-Filho et al. 2008; Dehnavi et al. 2020). In our study, the degree to which plants were affected by salt stress varied depending on the rootstock/scion combination. Balliu et al. (2008), reported that plants grafted on ‘Cyndia’ rootstocks had a higher stem growth rate than ungrafted plants. Plant growth (e.g., stem growth rate) of plants grafted on Zhejiang, which is widely used in China for production of grafted tomatoes, was less affected by salt stress than non-grafted plants (He et al. 2009). Since roots are the primary organ exposed to salt stress, salt-induced inhibition of root growth is quite obvious. Salt stress reduced the root growth of tomato plants in all graft combinations, but the rate of root dry mass reduction was less in grafted plants than in ungrafted plants. The performance of grafted plants compared to non-grafted plants under stress conditions generally depends on the characteristics of the root system of the rootstock; a strong root system may be the most important criterion for increasing salt tolerance. In parallel with our results, He et al. (2009), reported that grafted plants under salt stress (100 and 150 mM NaCl) conditions had more root dry mass than ungrafted plants. In all graft combinations, the dry weight of the shoots decreased more than that of the roots under salt stress. This finding agrees with Foolad (1997) and Ali et al. (2021),who mentioned that salinity reduces shoot growth more than root growth. In the study, the tolerance to salinity of SP5 and SP4 (S. pimpinellifolium) wild rootstocks increased. Parallel to our results, Pailles et al. (2020),reported that salt-tolerant and wild tomato species were able to maintain growth (based on dry mass) under salt stress conditions better than the cultivated tomato.
Plant growth was negatively affected by increasing the salinity level of the nutrient solution. The leaf area and root length of the grafted and non-grafted tomato plants decreased in response to the solution NaCl level. The result of leaf area and root length at the end of the growing period of graft combinations and tomato cultivars in different NaCl levels (1.5 and 8 dSm-1) was shown in Table 3. Leaf area was significantly affected by rootstock, scion NaCl levels, rootstock × scion, rootstock × NaCl, scion × NaCl levels and rootstock × scion × NaCl levels interactions in both control and saline conditions (p < 0.001). In control conditions, all grafted plants using the H2274 scion produced higher leaf area than ungrafted H2274 plants. Under control conditions, the highest leaf area was determined in the SP5/Galaxy 3123.00 cm2 plant–1 and SP4/Galaxy (3111.00 cm2 plant–1) graft combinations, respectively and the lowest leaf area was determined in the ungrafted H2274 plants (648.10 cm2 plant–1). With the salt application, there was a decrease in the leaf area varying between 41% (ungrafted H2274) and 77% (SH3/Galaxy) in all graft combinations. Under salt stress, all graft combinations produced more leaf area than the ungrafted H2274 plants except for the SH5/H2274 graft combination. Under saline conditions, the highest leaf area was determined in the SP5/Galaxy (983.70 cm2 plant–1), SH1/Galaxy (931.60 cm2 plant–1) and SP4/Galaxy (911.10 cm2 plant–1) graft combinations, respectively. Root length was significantly (p < 0.001) affected by scion, rootstock, NaCl levels, scion × rootstock, scion × NaCl levels (p < 0.01) and rootstock × NaCl levels interactions while main stem length was not affected by rootstock × scion × NaCl levels interactions. The root length was ranked between 12.29 m plant–1 (ungrafted H2271) and 39.32 m plant–1 (SH1/Galaxy) under control conditions. Under saline conditions, SP5/Galaxy graft combination produced significantly longer roots (24.41 m plant–1) and the shorter roots produced SH3/H2274 (7.05 m plant-1), SH5/H2274 (7.54 m plant-1) and ungrafted H2274 (9.92 m plant-1) plants, respectively. The mean of main of root length parameter of all graft combinations using Galaxy scion in control and saline conditions was higher than the mean of graft combinations using H2274 scion. The performance of grafted plants compared to ungrafted plants under stress conditions generally depends on the characteristics of the root system of the rootstock. A vigorous root system (root length and volume) could be the most important criterion for increasing salt tolerance (Ulas et al. 2020; Göçer et al. 2021; Aydın and Yetişir 2022). In this study, the salt-tolerant graft combinations (SP4/Galaxy, SP5/Galaxy and SH1/Galaxy) were specified with increased leaf area and root length under saline conditions in comparison to nongrafted control plants (H2274 and Galaxy). In parallel with our study, Ulas (2021), reported that grafted plants under saline conditions had greater leaf area and root length than ungrafted plants.
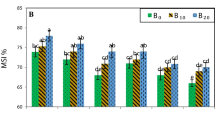
Saline conditions, healthy and damaged leaf area (%) were determined with the WinDIAS 3 Rapid System (Fig. 2). The healthy leaf area varied between 67.12% and 98.45% under saline conditions, while the damaged leaf area varied between 1.55% and 32.88%. Under saline conditions, SP4/Galaxy (98.45%), SP5/Galaxy(97.53%) and SH3/H2274 (97.02%) graft combinations produced the highest healthy leaf area, respectively, while the ungrafted H2274 (67.12%) plant produced the lowest healthy leaf area (Fig. 3). As a result of the salt application, significant differences were found between graft combinations in both total leaf area and area of damaged leaves. Specifically, salt damage can reduce leaf expansion. Plants that reduce leaf area adapt to salt stress by reducing transpiration and nutrient requirements (Bernstein et al. 1993; Greenway and Munns 2003). The present result for leaf area is in agreement with the results of Balibrea et al. (2000), who reported that leaf area of the salt-sensitive genotype of tomato was significantly affected by salinity. Rao et al. (2013) (S. pimpinellifolium), Albacete et al. (2009) (S. cheesmaniae) and Ali et al. (2021) (S. habrochaites and S. pennellii) reported that it reduced the negative effects of salt stress in tomato cultivation in areas with soil salinity problems from wild rootstocks.
Leaf chlorophyll content, which was significantly affected by rootstock, NaCl levels and rootstock × NaCl levels interactions in both control and saline conditions (p < 0.001). Salt application caused decreases in leaf chlorophyll content in all applications and the highest decrease was observed in L×SH3/Galaxy graft combination plants at 50% (Table 4). With the salt application, there was a decrease in the leaf chlorophyll content varying between 5% (ungrafted H2274) and 50% (L×SH3/Galaxy) in all graft combinations. Under saline conditions, the highest leaf chlorophyll content was determined in the ungrafted Galaxy (54.10) and SH3/H2274 (52.37) graft combinations. There was no significant variation in ion leakage (root and leaf) under control conditions among graft conditions. Root ion leakage, which was significantly affected by scion (p < 0.01), NaCl levels (p < 0.001), rootstock × scion (p < 0.05) and rootstock × NaCl levels (p < 0.05) interactions in both control and saline conditions. The root ion leakage ranged from 7 to 31% under salt stress. Saline conditions the highest root ion leakage was determined in the ungrafted plants H2274 (96.75%) and the lowest root ion leakage was determined in the SP5/H2274 (88.11%) graft combination. Leaf ion leakage was significantly (p < 0.001) affected by rootstock, scion NaCl levels, rootstock × NaCl, scion × NaCl levels and rootstock × scion × NaCl levels interactions. The leaf leakage ranged from 22 to 129% under salt stress. The highest increase in leaf ion leakage was determined in the L×SP5/H2274 (129%) and ungrafted H2274 (114%) plants. In saline conditions, the highest leaf ion leakage was determined in H2774 (68.95%) plants, as was root ion leakage. The results of this study show that the value of SPAD decreases under saline conditions in all graft combinations. The decrease in SPAD value in response to abiotic stress leads to a decrease in net photosynthesis and thus energy, which is very important for metabolism and growth (Sousaraei et al. 2021). The current result on leaf chlorophyll content is consistent with the result by Ali et al. (2021),working on wild tomato rootstock who reported that leaf chlorophyll content was significantly affected by the salinity in salt sensitive genotype. Similar to our leaf chlorophyll content results, in the study conducted on 8 salt-resistant rootstocks, the leaf chlorophyll content of plants grafted on salt-resistant rootstocks were higher than non-grafted plants (Abdeldym et al. 2020). It is a well-known phenomenon that salt stress leads to an increase in IL in plants. Reduced translocation of NaCl to the shoot system is achieved either by exclusion or restricted absorption by the roots (Moya et al. 1999). In our study, ion leakage from roots of all graft combinations was higher than leaf ion leakage from leaves under salt stress. However, the increase in ion leakage in leaf tissue is higher than in root tissue with salt applications. Aydın and Yetişir (2022), obtained results similar conclusions to our study in their study on grafted cucumbers.
The leaf nitrogen (N), phosphorus (P) and potassium (K) contents of tomato plants grown under different salt concentrations are given in Table 5. Leaf N content was significantly affected by rootstocks, salt treatment, rootstock × scion, rootstock × salt and scion × salt interaction in saline conditions. There was a significant decrease (1-78%) in the N content of the leaves under salt stress. Under salt stress, the highest leaf N content was detected in plants grafted SH3/H2274 (1.49%), SP4/H227(1.47%), L×SH3/H2274 (1.38%) and SH5/Galaxy (1.28%) while the lowest leaf N content was determined in ungrafted Galaxy (0.42%) plants. Leaf P content was also significantly influenced by the rootstock, scion, NaCl, rootstock × scion, rootstock × NaCl and rootstock × scion × NaCl interactions in both control and saline conditions (p < 0.001). In saline conditions, the highest leaf P content was determined in SP5/H2274, SH3/H2274 and L×SH3/H2274 graft combinations, while the lowest was determined in L×SH3/Galaxy, L×SP5/Galaxy and Galaxy (ungrafted) plants. Leaf potassium (K) content was also significantly influenced by all applications and the interaction of the applications in both control and saline conditions. In graft combinations under salt stress, the highest leaf K content was determined in SH1/H2274 (1.16%), SH3/Galaxy (0.84%) and SP5/H2274 (0.83%) plants, while the lowest was determined in SH5/Galaxy and ungrafted Galaxy plants with 0.33%. Significant negative correlations were found between leaf N, P and K content and Na, Cl, Cu, root ion leakage, leaf ion leakage and damaged leaf area. (Fig. 4). The accumulation of Na in the biomass may also be an indicator of salt tolerance. However, when Na accumulates in the cytosol of cells, it is toxic and leads to ionic imbalance (Hanin et al. 2016). In addition, Na reduces the availability of K binding sites for important metabolic processes in the cytoplasm. For the plant to protect itself from salt stress, it must either limit Na influx from the roots or control Na concentration and distribution after entry (Tester and Davenport 2003; Wei et al. 2017). A higher K, P and N content of plants seems to be related to the improvement in salt tolerance in grafted plants (Huang et al. 2009; Aydın and Yetişir 2022). In addition, high concentrations of Cl cause nitrogen or phosphorus deficiency (Wu and Li 2019). Accessions of the wild tomato species S. pimpinellifolium were screened for salt tolerance and provided tolerance to an EC level of 40 dSm-1 by Rao et al. (2013) who suggested that it could be a potential source of salt tolerance for the breeding of S. pimpinellifolium and results of the path analysis along with heritability and genetic advance showed that shoot dry weight and K/Na ratio are the two most critical component traits for survival, while fruit number is critical for yield per plant.
Leaf calcium (Ca), magnesium (Mg) and sulphur (S) contents of plants grown in control and saline conditions are given in Table 6. In control and saline conditions, leaf Ca content was significantly affected by rootstock, scion, NaCl, rootstock × scion, rootstock × NaCl and scion × NaCl interactions (p < 0.001). Salt application caused a decrease in Ca content in all graft combinations except SP4/H2274, SH5/Galaxy, SP5/Galaxy, L×SH3/H2274 and SP5/H2274 graft combinations. Under saline conditions, the highest leaf Ca content was determined in SP4/H2274 and SH3/H2274 grafted plants, while the lowest was determined in ungrafted H2274 plants. Under control and saline conditions, leaf Mg content was significantly affected by all treatments and interactions except rootstock × scion interaction. Salt application caused a decrease in leaf Mg content in all graft combinations except SP5/Galaxy (43%) graft combination, while the highest decrease was determined in L×SH3/Galaxy (90%) and ungrafted Galaxy (86%) plants. Leaf S content was significantly affected by all applications and interactions (p < 0.001). In plants grown under salt stress, leaf S content decreased in all graft combinations except SP4/H2274. Under saline conditions, the highest leaf S content was determined in the SP4/H2274 (0.15%) graft combination, which did not decrease (125%) with salt application, while the lowest was determined in the ungrafted H2274 (0.02%) plants, which showed the highest decrease (-96%). Leaf calcium (Ca), magnesium (Mg) and sulphur (S) contents were significantly positively correlated with N, P, K, Zn, Ca, Fe, SPAD and healthy leaf area (Fig. 4). The root system of grafted plants is stronger and more efficient in water and nutrient uptake. In addition, grafted tomato plants improve salt tolerance by reducing ionic stress, increasing K, Ca and Mg transfer to shoots and leaves (Singh et al. 2012; Koleška et al. 2018). Al-Harbi et al. (2017), reported that under saline conditions, grafted plants accumulated more Ca and K in the leaves and had lower Na and Cl levels. Salinity with a predominance of Na+ salts not only reduces the availability of Ca2+, but also reduces Ca2+ transport and mobility to the growing parts of the plant, which affects the quality of both vegetative and reproductive organs (Navarro et al. 2000; Aydın and Yetişir 2022). In our study, leaf S content decreased under salt stress. Similarly, Mor and Manchanda (2009), reported that S content decreased in tomato shoots under salt stress. P, Mg, Fe, Mn, Zn and Cu contents of plants generally decreased under salt stress (Kipçak et al. 2019).
Leaf manganese (Mn), iron (Fe) and zinc (Zn) contents of tomato plants grown under control and saline conditions are given in Table 7. In all graft combinations, salt application significantly decreased leaf Mn (19–90%) and Fe (4–61%) contents. Under saline conditions, the highest leaf Mn content was determined in SH1/Galaxy (21.38 mg kg-1) graft combination, while the lowest was determined in L×SH3/Galaxy (4.49 mg kg-1) and ungrafted Galaxy (4.60 mg kg-1) plants. In salt application plants, the highest leaf Fe content was determined in SH3/H2274 (48.92 mg kg-1), SP5/Galaxy (38.20 mg kg-1) and SP3/Galaxy (37.89 mg kg-1) grafted plants, while the lowest was determined in ungrafted H2274 (13.23 mg kg-1) plants. Leaf Zn content was significantly affected by all applications and interactions (p < 0.001). Under saline conditions, leaf Zn content decreased in all graft combinations except SP4/H2274 graft combination, while the highest decrease was obtained in ungrafted H2274 plants with 96%. The solubility and mobility of Cu, Fe and Zn ions are further reduced in saline soils. With increasing salinity, leaf Cu, Zn and Fe levels decrease proportionally. Similar to our results, NaCl-induced Mn deficiency has been previously observed in tomato (Balliu et al. 2015).
Leaf boron (B), copper (Cu), sodium (Na) and chlorine (Cl) contents of tomato plants grown under control and saline conditions are given in Table 8. Salt application caused an increase in leaf boron content in some graft combinations, while it caused a decrease in some graft combinations. The highest increase was determined in SP4/H2274 (204%) graft combination, while the highest decrease was determined in SP4/Galaxy (74%) graft combination. While the leaf Cu content of plants under salt stress decreased between 77 and 98%, the highest leaf Cu content was determined in ungrafted Galaxy (0.33 mg kg-1) and H2274 (0.32 mg kg-1) plants under saline conditions. Leaf Na and Cl contents of tomato plants grown under control and saline conditions were significantly affected by all applications and interactions (p < 0.001). Salt stress application increased leaf Na content between 375 and 8937% and Cl content between 389 and 5875% in all graft combinations. Under saline conditions, the lowest Na content was determined in SP4/Galaxy (8.34 mg kg-1) and SP3/Galaxy (9.16 mg kg-1) graft combinations, while the highest was determined in ungrafted H2274 (21.25 mg kg-1) and Galaxy (21.24 mg kg-1) plants. In NaCl application, the highest leaf Cl content was determined in H2274 (119.80 mg kg-1) and Galaxy (101.87 mg kg-1) ungrafted plants, while the lowest was determined in SH1/Galaxy (12.51 mg kg-1) graft combination. Leaf Na and Cl content were positively correlated with leaf Cu content, root ion leakage, leaf ion leakage and damaged leaf area, while SPAD, healthy leaf area, leaf area, root length, main stem length, shoot dry weight and root dry weight were negatively correlated (Fig. 4). In general, the main factor inhibiting growth of salt-stressed plants is elevated levels of Na and Cl with roots remaining the primary sites for stress perception and the subsequent responses at the cell, organ or whole plant levels (Rajaei et al. 2009). He et al. (2009) found that Na+ levels in leaves and roots increased significantly with increasing NaCl concentration. In parallel with our results, Al-Harbi et al. (2017), reported that leaf Na and Cl accumulation of grafted tomato plants were lower than ungrafted tomato plants under salt stress conditions. The detrimental effects of Cl are caused by interference with the uptake or metabolism of other essential ions such as N, P and K (Al-Harbi et al. 2017; Zhao et al. 2020). Parallel to our results, tomato cultivars Fanny and Goldmar grafted onto rootstock AR-9704 showed differential accumulation of Na and Cl, with Cl and Na concentrations being significantly higher in non-grafted than in grafted plants (Fernández-García et al. 2004). Di Gioia et al. (2013), reported that the leaf K/Na, Ca/Na and Mg/Na ratios of grafted tomato plants grown under salt stress were higher than those without grafting.
3.1 Principal component analysis
PCA was used for classifying graft combinations based on plant growth parameters and leaf ion content under salt conditions. According to the PCA analysis, the two principal components formed 99% of the total variation (99.89% by PC1 and 0.10% by PC2). When PCA charts were examined, it could be seen that graft combinations were separated into five different groups based on measured features (Fig. 4). The first group is the group containing L×SH3/Galaxy graft combination in the region I of the graph and is indicated by the black circle. The L×SH3/Galaxy graft combination in this circle has the highest leaf Cu content, while the healthy leaf area is the lowest. The ungrafted H2274 graft combination in the yellow circle contains the most Na and Cl in the leaf tissues, while it is negatively correlated with biomass parameters. The graft combinations in the green circle are the graft combinations that contain the most nutrients in the leaves except Na, Cl and Cu. The graft combinations in the red circle show positive correlation with biomass parameters and negative correlation with leaf Na, Cl, and leaf and root ion leakage parameters. Principal component analysis (PCA) was used in different crops such as Brassica napus L. (Shuvo 2021), Zea mays L. (Andrade et al. 2020), Glycine max (Azam et al. 2020), Triticum aestivum (Uzair et al. 2022) and Oryza sativa (Das et al. 2019) to identify salt tolerant plants.
PCA formed by physiological and biochemical contents of plants under salt stress. PCA:principal component analysis, RL:root length, RDW:root dry weight,SDW:shoot dry weight, MSL:main stem length, LA:leaf area, HL: healthy leaf, D:damaged leaf, Root IL: root ion leakage, Leaf IL:leaf ion leakage, N:nitrogen, P:phosphorus, K:potassium, Ca:calcium, Mg:magnesium, S:sulphur, Mn:manganese, Fe:iron, Zn:zinc, B:boron, Cu:copper, Na:sodium and Cl:chlorine
4 Conclusion
Salinity, one of the most common abiotic stressors, often has detrimental effects on crop production capacity by reducing yield and quality, especially in arid and semi-arid regions of the world. Salt stress causes a decrease in plant height, shoot and root biomass root length, plant height, leaf area, and the overall development process in tomato plants. To overcome this problem grafting with salt-tolerant rootstocks can be an effective management strategy to improve the salt tolerance of crop plants. The selection of salt-tolerant rootstocks is an important strategy to improve salt tolerance. Testing and screening of existing commercial and wild relatives under salt stress conditions is a prerequisite for grafting. Due to incompatibility in crossing and linkage drag, it is cumbersome to transfer salt tolerant genes from wild to cultivated tomato cultivars. However, these wild species could be exploited as salt-resistant rootstocks for grafting susceptible but high yielding commercial tomato cultivars. Plants grafted on wild tomato rootstocks (S. habrochaites and S. pimpinellifolium) used in our study showed more tolerance to salt stress than ungrafted plants. Plants grafted on wild rootstocks were less affected in terms of biomass development by having less Na and Cl in the leaf than ungrafted plants. Biotic and abiotic stress factors vary in different locations or countries, so the combination of scion and rootstock should be carefully selected to obtain maximum benefit from grafting.
References
Abdeldym EA, El-Mogy MM, Abdellateaf HRL, Atia MAM (2020) Genetic Characterization, Agro-Morphological and Physiological Evaluation of Grafted Tomato under Salinity Stress Conditions. Agronomy 2020, Vol 10, Page 1948 10:1948. https://doi.org/10.3390/AGRONOMY10121948
Al-Harbi A, Hejazi A, Al-Omran A (2017) Responses of grafted tomato (Solanum Lycopersiocon L.) to abiotic stresses in Saudi Arabia. Saudi J Biol Sci 24:1274–1280. https://doi.org/10.1016/J.SJBS.2016.01.005
Albacete A, MartÍnez-AndÚjar C, Ghanem ME et al (2009) Rootstock-mediated changes in xylem ionic and hormonal status are correlated with delayed leaf senescence, and increased leaf area and crop productivity in salinized tomato. Plant Cell Environ 32:928–938. https://doi.org/10.1111/J.1365-3040.2009.01973
Albaladejo I, Meco V, Plasencia F et al (2017) Unravelling the strategies used by the wild tomato species Solanum pennellii to confront salt stress: from leaf anatomical adaptations to molecular responses. Environ Exp Bot 135:1–12. https://doi.org/10.1016/J.ENVEXPBOT.2016.12.003
Ali AAM, Romdhane W, Ben, Tarroum M et al (2021) Analysis of Salinity Tolerance in Tomato Introgression Lines Based on Morpho-Physiological and Molecular Traits. Plants (Basel) 10:. https://doi.org/10.3390/PLANTS10122594
Andrade GC, Medeiros Coelho CM, Uarrota VG (2020) Modelling the vigour of maize seeds submitted to artificial accelerated ageing based on ATR-FTIR data and chemometric tools (PCA, HCA and PLS-DA). https://doi.org/10.1016/J.HELIYON.2020.E03477. Heliyon 6:
Ansari WA, Atri N, Ahmad J et al (2019) Drought mediated physiological and molecular changes in muskmelon (Cucumis melo L). PLoS ONE 14:e0222647. https://doi.org/10.1371/JOURNAL.PONE.0222647
Arora NK (2019) Impact of climate change on agriculture production and its sustainable solutions. Environmental Sustainability 2019 2:2 2:95–96. https://doi.org/10.1007/S42398-019-00078-W
Aydın A, Yetişir H (2022) Rootstock Effect of Auto- and Allotetraploid Citron (Citrullus lanatus var. citroides) on hydroponically grown Cucumber under Salt stress. https://doi.org/10.1007/S10343-022-00782-4/TABLES/9. Gesunde Pflanzen 1–14
Azam M, Zhang S, Abdelghany AM et al (2020) Seed isoflavone profiling of 1168 soybean accessions from major growing ecoregions in China. Food Res Int 130:108957. https://doi.org/10.1016/J.FOODRES.2019.108957
Balibrea ME, Dell’Amico J, Bolarín MC, Pérez-Alfocea F (2000) Carbon partitioning and sucrose metabolism in tomato plants growing under salinity. Physiol Plant 110:503–511. https://doi.org/10.1111/J.1399-3054.2000.1100412.X
Balliu A, Vuksani G, Nasto T et al (2008) Grafting effects on tomato growth rate, yield and fruit quality under saline irrigation water. Acta Hortic 801 PART 2:1161–1166. https://doi.org/10.17660/ACTAHORTIC.2008.801.141
Balliu A, Sallaku G, Rewald B (2015) AMF Inoculation Enhances Growth and Improves the Nutrient Uptake Rates of Transplanted, Salt-Stressed Tomato Seedlings. Sustainability 2015, Vol 7, Pages 15967–15981 7:15967–15981. https://doi.org/10.3390/SU71215799
Bauchet G, Causse M, Bauchet G, Causse M (2012) Genetic diversity in Tomato (Solanum lycopersicum) and its wild relatives. Genetic Divers Plants. https://doi.org/10.5772/33073
Behera TK, Krishna R, Ansari WA et al (2022) Approaches involved in the Vegetable crops salt stress tolerance improvement: Present Status and Way ahead. Front Plant Sci 12:787292. https://doi.org/10.3389/FPLS.2021.787292/BIBTEX
Bernstein N, Silk WK, Läuchli A (1993) Growth and development of sorghum leaves under conditions of NaCl stress - spatial and temporal aspects of leaf growth inhibition. Planta 191:433–439. https://doi.org/10.1007/BF00195744/METRICS
Brdar M, · J, Zdravković J (2015) Germination of tomatoes under PEG-induced drought stress. Ratarstvo i Povrtarstvo 52:108–113. https://doi.org/10.5937/RATPOV52-8324
Brugnoli E, Lauteri M (1991) Effects of Salinity on Stomatal Conductance, photosynthetic capacity, and Carbon Isotope discrimination of Salt-Tolerant (Gossypium hirsutum L.) and salt-sensitive (Phaseolus vulgaris L.) C3 non-halophytes. Plant Physiol 95:628. https://doi.org/10.1104/PP.95.2.628
Colla G, Rouphael Y, Jawad R et al (2013) The effectiveness of grafting to improve NaCl and CaCl2 tolerance in cucumber. Sci Hortic 164:380–391. https://doi.org/10.1016/J.SCIENTA.2013.09.023
Conti V, Parrotta L, Romi M et al (2023) Tomato Biodiversity and Drought Tolerance: a Multilevel Review. Int J Mol Sci 2023 24:10044. https://doi.org/10.3390/IJMS241210044
Das B, Manohara KK, Mahajan GR, Sahoo RN (2019) Spectroscopy based novel spectral indices, PCA- and PLSR-coupled machine learning models for salinity stress phenotyping of rice. Spectrochim Acta Mol Biomol Spectrosc 229:117983–117983. https://doi.org/10.1016/J.SAA.2019.117983
Dehnavi AR, Zahedi M, Ludwiczak A et al (2020) Effect of Salinity on Seed Germination and Seedling Development of Sorghum (Sorghum bicolor (L.) Moench) Genotypes. Agronomy 2020, Vol 10, Page 859 10:859. https://doi.org/10.3390/AGRONOMY10060859
Di Gioia F, Signore A, Serio F, Santamaria P (2013a) Grafting improves Tomato Salinity Tolerance through Sodium partitioning within the shoot. HortScience 48:855–862. https://doi.org/10.21273/HORTSCI.48.7.855
Di Gioia F, Signore A, Serio F, Santamaria P (2013b) Grafting improves Tomato Salinity Tolerance through Sodium partitioning within the shoot. HortScience 48:855–862. https://doi.org/10.21273/HORTSCI.48.7.855
Ebert AW, Schafleitner R (2015) Utilization of wild relatives in the breeding of Tomato and other Major vegetables. Crop Wild Relatives Clim Change 141–172. https://doi.org/10.1002/9781118854396.CH9
Etehadnia M, Waterer DR, Tanino KK (2008) The method of ABA application affects salt stress responses in resistant and sensitive potato lines. J Plant Growth Regul 27:331–341. https://doi.org/10.1007/S00344-008-9060-9
Fernández-García N, Martínez V, Carvajal M (2004) Effect of salinity on growth, mineral composition, and water relations of grafted tomato plants. J Plant Nutr Soil Sci 167:616–622. https://doi.org/10.1002/JPLN.200420416
Flint HL, Boyce BR, Beattie DJ, Index of injury—a useful expression of freezing injury to plant tissues as determined by the electrolytic method (2011). 47:229–230. https://doi.org/10.4141/CJPS67-043
Foolad MR (1997) Genetic basis of physiological traits related to salt tolerance in tomato, Lycopersicon esculentum Mill. Plant Breeding 116:53–58. https://doi.org/10.1111/J.1439-0523.1997.TB00974.X
Foolad MR (2004) Recent advances in genetics of salt tolerance in tomato. Plant Cell Tissue Organ Cult 76:101–119. https://doi.org/10.1023/B:TICU.0000007308.47608.88/METRICS
Frary A, Göl D, Keleş D et al (2010) Salt tolerance in Solanum pennellii: antioxidant response and related QTL. BMC Plant Biol 10:1–16. https://doi.org/10.1186/1471-2229-10-58/FIGURES/5
Gharbi E, Martínez JP, Benahmed H et al (2017) Phytohormone profiling in relation to osmotic adjustment in NaCl-treated plants of the halophyte tomato wild relative species Solanum chilense comparatively to the cultivated glycophyte Solanum lycopersicum. Plant Sci 258:77–89. https://doi.org/10.1016/J.PLANTSCI.2017.02.006
Göçer H, ∃ HY, Ulaş A et al (2021) Plant Growth, Ion Accumulation and essential oil content of Salvia officinalis Mill. And S. Tomentosa L. grown under different salt stress. KSU Tarim VE Doga Dergisi-KSU Journal of Agriculture. Nat 24:505–514. https://doi.org/10.18016/KSUTARIMDOGA.VI.730477
Gomes-Filho E, Lima CRFM, Costa JH et al (2008) Cowpea ribonuclease: Properties and effect of NaCl-salinity on its activation during seed germination and seedling establishment. Plant Cell Rep 27:147–157. https://doi.org/10.1007/S00299-007-0433-5/FIGURES/8
Greenway H, Munns R (2003) Mechanisms of Salt Tolerance in Nonhalophytes. 31:149–190. https://doi.org/10.1146/ANNUREV.PP.31.060180.001053
Hanin M, Ebel C, Ngom M et al (2016) New insights on Plant Salt Tolerance mechanisms and their potential use for breeding. Front Plant Sci 7. https://doi.org/10.3389/FPLS.2016.01787
Hasanuzzaman M, Fujita M (2022) Plant responses and tolerance to salt stress: physiological and molecular interventions. Int J Mol Sci 23:4810. https://doi.org/10.3390/IJMS23094810
He Y, Zhu Z, Yang J et al (2009a) Grafting increases the salt tolerance of tomato by improvement of photosynthesis and enhancement of antioxidant enzymes activity. Environ Exp Bot 66:270–278. https://doi.org/10.1016/J.ENVEXPBOT.2009.02.007
He Y, Zhu Z, Yang J et al (2009b) Grafting increases the salt tolerance of tomato by improvement of photosynthesis and enhancement of antioxidant enzymes activity. Environ Exp Bot 66:270–278. https://doi.org/10.1016/J.ENVEXPBOT.2009.02.007
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Circular Calif Agricultural Exp Stn 347
Hou C, Li X, Tian D et al (2022) Evaluation of the Effects of Water and Salinity Stress on the Growth and Biochemistry of Alfalfa (Medicago sativa L.) at the Branching Stage. Sustainability 2022, Vol 14, Page 10262 14:10262. https://doi.org/10.3390/SU141610262
Huang Y, Tang R, Cao Q, Bie Z (2009) Improving the fruit yield and quality of cucumber by grafting onto the salt tolerant rootstock under NaCl stress. Sci Hortic 122:26–31. https://doi.org/10.1016/J.SCIENTA.2009.04.004
Hutton SF, Scott JW, Schuster DJ (2012) Recessive resistance to Tomato yellow leaf curl virus from the Tomato Cultivar Tyking is located in the same region as Ty-5 on chromosome 4. HortScience 47:324–327. https://doi.org/10.21273/HORTSCI.47.3.324
İşeri ÖD, Körpe DA, Sahin FI, Haberal M (2015) High salt induced oxidative damage and antioxidant response in tomato grafted on tobacco. Chil J Agric Res 75:192–201. https://doi.org/10.4067/S0718-58392015000200008
Ji Y, Scott JW, Schuster DJ, Maxwell DP (2009) Molecular mapping of Ty-4, a New Tomato Yellow Leaf Curl Virus Resistance Locus on chromosome 3 of Tomato. J Am Soc Hortic Sci 134:281–288. https://doi.org/10.21273/JASHS.134.2.281
Kashyap SP, Prasanna HC, Kumari N et al (2020) Understanding salt tolerance mechanism using transcriptome profiling and de novo assembly of wild tomato Solanum chilense. Scientific Reports 2020 10:1 10:1–20. https://doi.org/10.1038/s41598-020-72474-w
Keatinge JDH, Lin LJ, Ebert AW et al (2014) Overcoming biotic and abiotic stresses in the Solanaceae through grafting: current status and future perspectives. http://dx.doi.org/101080/014487652014964317 30:272–287. https://doi.org/10.1080/01448765.2014.964317
Kesawat MS, Satheesh N, Kherawat BS et al (2023) Regulation of reactive oxygen species during salt stress in plants and their crosstalk with other Signaling molecules—current perspectives and future directions. https://doi.org/10.3390/PLANTS12040864. Plants 12:
Kiferle C, Gonzali S, Beltrami S et al (2022) Improvement in fruit yield and tolerance to salinity of tomato plants fertigated with micronutrient amounts of iodine. Scientific Reports 2022 12:1 12:1–14. https://doi.org/10.1038/s41598-022-18301-w
Kipçak S, Ekıncıalp A, Erdınç Ç et al (2019) Tuz Stresinin Farklı Fasulye Genotiplerinde Bazı Besin Elementi İçeriği Ile Toplam Antioksidan ve Toplam Fenol İçeriğine Etkisi. Yüzüncü Yıl Üniversitesi. Tarım Bilimleri Dergisi 29:136–144. https://doi.org/10.29133/YYUTBD.504748
Koleška I, Hasanagić D, Todorović V et al (2018) Grafting influence on the weight and quality of tomato fruit under salt stress. Ann Appl Biol 172:187–196. https://doi.org/10.1111/AAB.12411
Lee J-M, Oda M (2010) Grafting of Herbaceous Vegetable and Ornamental crops. Hortic Rev (Am Soc Hortic Sci 61–124. https://doi.org/10.1002/9780470650851.CH2
Martorana M, Giuffrida F, Leonardi C, Kaya S (2007) Influence of rootstock on tomato response to salinity. Acta Hortic 747:555–561. https://doi.org/10.17660/ACTAHORTIC.2007.747.72
Mor RP, Manchanda HR (2009) Influence of phosphorus on the tolerance of table pea to chloride and sulfate salinity in a sandy soil. 6:41–52. https://doi.org/10.1080/15324989209381295. http://dx.doi.org/101080/15324989209381295
Moya JL, Primo-Millo E, Talon M (1999) Morphological factors determining salt tolerance in citrus seedlings: the shoot to root ratio modulates passive root uptake of chloride ions and their accumulation in leaves. Plant Cell Environ 22:1425–1433. https://doi.org/10.1046/J.1365-3040.1999.00495.X
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681. https://doi.org/10.1146/ANNUREV.ARPLANT.59.032607.092911
Navarro JM, Martínez V, Carvajal M (2000) Ammonium, bicarbonate and calcium effects on tomato plants grown under saline conditions. Plant Sci 157:89–96. https://doi.org/10.1016/S0168-9452(00)00272-7
Pailles Y, Awlia M, Julkowska M et al (2020) Diverse traits contribute to Salinity Tolerance of Wild Tomato seedlings from the Galapagos Islands. Plant Physiol 182:534–546. https://doi.org/10.1104/PP.19.00700
Rajaei SM, Niknam V, Seyedi SM et al (2009) Contractile roots are the most sensitive organ in Crocus sativus to salt stress. http://bp.ueb.cas.cz/doi/101007/s10535-009-0095-y.html 53:523–529. https://doi.org/10.1007/S10535-009-0095-Y
Rao ES, Kadirvel P, Symonds RC, Ebert AW (2013a) Relationship between survival and yield related traits in Solanum pimpinellifolium under salt stress. Euphytica 190:215–228. https://doi.org/10.1007/S10681-012-0801-2/TABLES/6
Rao ES, Kadirvel P, Symonds RC, Ebert AW (2013b) Relationship between survival and yield related traits in Solanum pimpinellifolium under salt stress. Euphytica 190:215–228. https://doi.org/10.1007/S10681-012-0801-2/TABLES/6
Raziq A, Wang Y, Mohi Ud Din A et al (2022) A comprehensive evaluation of Salt Tolerance in Tomato (Var. Ailsa Craig): responses of physiological and transcriptional changes in RBOH’s and ABA biosynthesis and signalling genes. Int J Mol Sci 23. https://doi.org/10.3390/IJMS23031603/S1
Robbins MD, Darrigues A, Sim SC et al (2009) Characterization of hypersensitive resistance to bacterial spot race T3 (Xanthomonas perforans) from Tomato Accession PI 128216. 1037–1044. https://doi.org/101094/PHYTO-99-9-103799.
Rubaye OM, Al, Yetisir H, Ulas F, Ulas A (2020) Growth of pepper inbred lines as affected by rootstocks with vigorous root system under salt stress conditions. Acta Hortic 1273:479–485. https://doi.org/10.17660/ACTAHORTIC.2020.1273.60
Shuvo II (2021) A holistic decision-making approach for identifying influential parameters affecting sustainable production process of Canola bast fibres and predicting end-use textile choice using principal component analysis (PCA). Heliy 7:e06235. https://doi.org/10.1016/J.HELIYON.2021.E06235
Singh J, Sastry EVD, Singh V (2012) Effect of salinity on tomato (Lycopersicon esculentum Mill.) During seed germination stage. Physiol Mol Biology Plants 18:45–50. https://doi.org/10.1007/S12298-011-0097-Z/TABLES/2
Sousaraei N, Mashayekhi K, Mousavizadeh SJ et al (2021) Screening of tomato landraces for drought tolerance based on growth and chlorophyll fluorescence analyses. Hortic Environ Biotechnol 62:521–535. https://doi.org/10.1007/S13580-020-00328-5/FIGURES/4
Summart J, Thanonkeo P, Panichajakul S et al (2012) Effect of salt stress on growth, inorganic ion and proline accumulation in Thai aromatic rice, Khao Dawk Mali 105, callus culture. Afr J Biotechnol 9:145–152. https://doi.org/10.4314/ajb.v9i2
Szymański J, Bocobza S, Panda S et al (2020) Analysis of wild tomato introgression lines elucidates the genetic basis of transcriptome and metabolome variation underlying fruit traits and pathogen response. Nat Genet 2020 52:10. https://doi.org/10.1038/s41588-020-0690-6
Tester M, Davenport R (2003) Na + tolerance and na + transport in higher plants. Ann Bot 91:503. https://doi.org/10.1093/AOB/MCG058
ULAS F (2021) Effects of grafting on growth, root morphology and leaf physiology of pepino (Solanum muricatum Ait.) As affected by salt stress under hydroponic conditions. Int J Agric Environ Food Sci 5:203–212. https://doi.org/10.31015/JAEFS.2021.2.10
Ulas A, Aydin A, Ulas F et al (2020) Cucurbita rootstocks improve salt tolerance of melon scions by inducing physiological, biochemical and nutritional responses. Horticulturae 6:1–13. https://doi.org/10.3390/HORTICULTURAE6040066
Uzair M, Patil SB, Zhang H et al (2022) Screening direct seeding-related traits by using an Improved Mesocotyl Elongation Assay and Association between Seedling and Maturity traits in Rice. Agronomy 12:975. https://doi.org/10.3390/AGRONOMY12040975/S1
Voutsela S, Khah E, Petropoulos SA (2012) The Effect of Grafting on Plant Growth and yield of tomato plants cultivated outdoors and indoors under water stress. 7:5553–5557. https://doi.org/10.5897/AJAR11.2448
Wei D, Zhang W, Wang C et al (2017) Genetic engineering of the biosynthesis of glycinebetaine leads to alleviate salt-induced potassium efflux and enhances salt tolerance in tomato plants. Plant Sci 257:74–83. https://doi.org/10.1016/J.PLANTSCI.2017.01.012
Wu H, Li Z (2019) The importance of Cl– exclusion and vacuolar Cl– Sequestration: revisiting the role of Cl– transport in Plant Salt Tolerance. Front Plant Sci 10:482468. https://doi.org/10.3389/FPLS.2019.01418/BIBTEX
Yamaguchi T, Blumwald E (2005) Developing salt-tolerant crop plants: challenges and opportunities. Trends Plant Sci 10:615–620. https://doi.org/10.1016/J.TPLANTS.2005.10.002
Zhao C, Zhang H, Song C et al (2020) Mechanisms of plant responses and adaptation to Soil Salinity. Innov (Cambridge (Mass)) 1. https://doi.org/10.1016/J.XINN.2020.100017
Zhu J, Bie Z, Li Y (2008) Physiological and growth responses of two different salt-sensitive cucumber cultivars to NaCl stress. Soil Sci Plant Nutr 54:400–407. https://doi.org/10.1111/J.1747-0765.2008.00245.X
Acknowledgements
We thank all staff members of the To the R&D greenhouse of Kırşehir Ahi Evran University, for the technical supports and supplying all facilities during the experiments.
Funding
This research was not funded.
Open access funding provided by the Scientific and Technological Research Council of Türkiye (TÜBİTAK).
Author information
Authors and Affiliations
Contributions
The contributions of author is defined as follows: study concept and design: A. Aydın; data collection: A. Aydın; analysis and interpretation of results: A. Aydın; preparing a draft text: A. Aydın. Author reviewed the results and approved the final version of the article.
Corresponding author
Ethics declarations
Competing interests
The author declares that he has no known competing financial interests or personal relationships that could influence the work in this article.
Corresponding author CV
I work as a lecturer at Kırşehir Ahi Evran University, under the coordination of pilot projects.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aydin, A. Effects of grafting with wild tomato (Solanum pimpinellifolium and Solanum habrochaites) rootstocks on growth and leaf mineral accumulation in salt stress. Hortic. Environ. Biotechnol. (2024). https://doi.org/10.1007/s13580-024-00607-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13580-024-00607-5