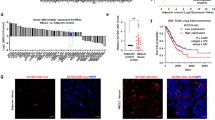
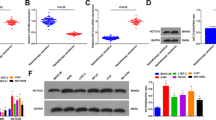
Abstract
Crosstalk between cancer cells and macrophages plays a crucial role in the development of cancer. In this study, our data showed that M1 macrophages attenuate, while M2 macrophages and tumor-associated macrophages enhance the EGFR-TKIs resistance in non-small cell lung cancer (NSCLC) cell line H1975. Next, long non-coding RNA SOX2 overlapping transcript (SOX2-OT) is highly expressed in NSCLC cells-derived exosomes. NSCLC cells-derived exosomes promote macrophages M2 polarization and inhibit M1 polarization through transferring SOX2-OT to macrophages. Subsequently, our results indicated that NSCLC cells-induced M2-polarized macrophages enhance the EGFR-TKIs resistance in H1975 cells. Furthermore, our data revealed that NSCLC cells-derived exosomes inhibit the expression of miR-627-3p, while promote Smads expression in THP-1 cells. SOX2-OT acts as miR-627-3p sponge to facilitate Smad2, Smad3 and Smad4 expression. Finally, our results indicated that NSCLC cells promote macrophages M2 polarization and suppress M1 polarization through targeting miR-627-3p/Smads signaling pathway by transferring exosomes to THP-1 cells. In conclusion, our data revealed that NSCLC cells promote macrophages M2 polarization through transferring exosomal SOX2-OT, thus to enhance its own EGFR-TKIs resistance. Mechanismly, NSCLC cells-derived exosomal SOX2-OT promotes macrophages M2 polarization via promoting Smads by sponging miR-627-3p. Our data provide a novel therapeutic target for EGFR-TKIs resistance in NSCLC.






Similar content being viewed by others
References
Goebel C, Louden CL, McKenna R Jr, Onugha O, Wachtel A, Long T. Diagnosis of non-small cell lung cancer for early stage asymptomatic patients. Cancer Genomics Proteomics. 2019;16(4):229–44. https://doi.org/10.21873/cgp.20128.
Saito S, Espinoza-Mercado F, Liu H, Sata N, Cui X, Soukiasian HJ. Current status of research and treatment for non-small cell lung cancer in never-smoking females. Cancer Biol Ther. 2017;18(6):359–68. https://doi.org/10.1080/15384047.2017.1323580.
Remark R, Becker C, Gomez JE, Damotte D, Dieu-Nosjean MC, Sautès-Fridman C, Fridman WH, Powell CA, Altorki NK, Merad M, Gnjatic S. The non-small cell lung cancer immune contexture. A major determinant of tumor characteristics and patient outcome. Am J Respir Crit Care Med. 2015;191(4):377–90. https://doi.org/10.1164/rccm.201409-1671PP.
Jonna S, Subramaniam DS. Molecular diagnostics and targeted therapies in non-small cell lung cancer (NSCLC): an update. Discov Med. 2019;27(148):167–70.
Leighl NB, Karaseva N, Nakagawa K, Cho BC, Gray JE, Hovey T, Walding A, Rydén A, Novello S. Patient-reported outcomes from FLAURA: Osimertinib versus erlotinib or gefitinib in patients with EGFR-mutated advanced non-small-cell lung cancer. Eur J Cancer (Oxford, England: 1990). 2020;125:49–57. https://doi.org/10.1016/j.ejca.2019.11.006.
Isidro RA, Appleyard CB. Colonic macrophage polarization in homeostasis, inflammation, and cancer. Am J Physiol Gastrointest Liver Physiol. 2016;311(1):G59-73. https://doi.org/10.1152/ajpgi.00123.2016.
Genin M, Clement F, Fattaccioli A, Raes M, Michiels C. M1 and M2 macrophages derived from THP-1 cells differentially modulate the response of cancer cells to etoposide. BMC Cancer. 2015;15:577. https://doi.org/10.1186/s12885-015-1546-9.
Ruffell B, Coussens LM. Macrophages and therapeutic resistance in cancer. Cancer Cell. 2015;27(4):462–72. https://doi.org/10.1016/j.ccell.2015.02.015.
Li H, Huang N, Zhu W, Wu J, Yang X, Teng W, Tian J, Fang Z, Luo Y, Chen M, Li Y. Modulation the crosstalk between tumor-associated macrophages and non-small cell lung cancer to inhibit tumor migration and invasion by ginsenoside Rh2. BMC Cancer. 2018;18(1):579. https://doi.org/10.1186/s12885-018-4299-4.
Li Y, Cao F, Li M, Li P, Yu Y, Xiang L, Xu T, Lei J, Tai YY, Zhu J, Yang B, Jiang Y, Zhang X, Duo L, Chen P, Yu X. Hydroxychloroquine induced lung cancer suppression by enhancing chemo-sensitization and promoting the transition of M2-TAMs to M1-like macrophages. J Exp Clin Cancer Res. 2018;37(1):259. https://doi.org/10.1186/s13046-018-0938-5.
Zheng H, Zhan Y, Liu S, Lu J, Luo J, Feng J, Fan S. The roles of tumor-derived exosomes in non-small cell lung cancer and their clinical implications. J Exp Clin Cancer Res. 2018;37(1):226. https://doi.org/10.1186/s13046-018-0901-5.
Pang X, Wang SS, Zhang M, Jiang J, Fan HY, Wu JS, Wang HF, Liang XH. OSCC cell-secreted exosomal CMTM6 induced M2-like macrophages polarization via ERK1/2 signaling pathway. Cancer Immunol Immunother. 2020. https://doi.org/10.1007/s00262-020-02741-2.
Teng Y, Kang H, Chu Y. Identification of an exosomal long noncoding RNA SOX2-OT in plasma as a promising biomarker for lung squamous cell carcinoma. Genet Test Mol Biomarkers. 2019;23(4):235–40. https://doi.org/10.1089/gtmb.2018.0103.
Yang N, Wang Y, Hui L, Li X, Jiang X. Silencing SOX2 expression by RNA interference inhibits proliferation, invasion and metastasis, and induces apoptosis through MAP4K4/JNK signaling pathway in human laryngeal cancer TU212 cells. J Histochem Cytochem. 2015;63(9):721–33. https://doi.org/10.1369/0022155415590829.
Zeng Z, Li Y, Pan Y, Lan X, Song F, Sun J, Zhou K, Liu X, Ren X, Wang F, Hu J, Zhu X, Yang W, Liao W, Li G, Ding Y, Liang L. Cancer-derived exosomal miR-25-3p promotes pre-metastatic niche formation by inducing vascular permeability and angiogenesis. Nat Commun. 2018;9(1):5395. https://doi.org/10.1038/s41467-018-07810-w.
Zeng M, Zhu L, Li L, Kang C. miR-378 suppresses the proliferation, migration and invasion of colon cancer cells by inhibiting SDAD1. Cell Mol Biol Lett. 2017;22:12. https://doi.org/10.1186/s11658-017-0041-5.
Poulet C, Njock MS. Exosomal long non-coding RNAs in lung diseases. Int J Mol Sci. 2020. https://doi.org/10.3390/ijms21103580.
Soria JC, Ohe Y, Vansteenkiste J, Reungwetwattana T, Chewaskulyong B, Lee KH, Dechaphunkul A, Imamura F, Nogami N, Kurata T, Okamoto I, Zhou C, Cho BC, Cheng Y, Cho EK, Voon PJ, Planchard D, Su WC, Gray JE, Lee SM, Hodge R, Marotti M, Rukazenkov Y, Ramalingam SS. Osimertinib in untreated EGFR-mutated advanced non-small-cell lung cancer. N Engl J Med. 2018;378(2):113–25. https://doi.org/10.1056/NEJMoa1713137.
Loong HH, Kwan SS, Mok TS, Lau YM. Therapeutic strategies in EGFR mutant non-small cell lung cancer. Curr Treat Options Oncol. 2018;19(11):58. https://doi.org/10.1007/s11864-018-0570-9.
Xu F, Cui WQ, Wei Y, Cui J, Qiu J, Hu LL, Gong WY, Dong JC. Astragaloside IV inhibits lung cancer progression and metastasis by modulating macrophage polarization through AMPK signaling. J Exp Clin Cancer Res. 2018;37(1):207. https://doi.org/10.1186/s13046-018-0878-0.
Zhang X, Zeng Y, Qu Q, Zhu J, Liu Z, Ning W, Zeng H, Zhang N, Du W, Chen C, Huang JA. PD-L1 induced by IFN-γ from tumor-associated macrophages via the JAK/STAT3 and PI3K/AKT signaling pathways promoted progression of lung cancer. Int J Clin Oncol. 2017;22(6):1026–33. https://doi.org/10.1007/s10147-017-1161-7.
Xiao F, Liu N, Ma X, Qin J, Liu Y, Wang X. M2 macrophages reduce the effect of gefitinib by activating AKT/mTOR in gefitinib-resistant cell lines HCC827/GR. Thoracic Cancer. 2020. https://doi.org/10.1111/1759-7714.13670.
Mashouri L, Yousefi H, Aref AR, Ahadi AM, Molaei F, Alahari SK. Exosomes: composition, biogenesis, and mechanisms in cancer metastasis and drug resistance. Mol Cancer. 2019;18(1):75. https://doi.org/10.1186/s12943-019-0991-5.
Whiteside TL. Tumor-derived exosomes and their role in cancer progression. Adv Clin Chem. 2016;74:103–41. https://doi.org/10.1016/bs.acc.2015.12.005.
Li X, Lei Y, Wu M, Li N. Regulation of macrophage activation and polarization by HCC-derived exosomal lncRNA TUC339. Int J Mol Sci. 2018. https://doi.org/10.3390/ijms19102958.
Bhan A, Soleimani M, Mandal SS. Long noncoding RNA and cancer: a new paradigm. Can Res. 2017;77(15):3965–81. https://doi.org/10.1158/0008-5472.can-16-2634.
Lei Y, Guo W, Chen B, Chen L, Gong J, Li W. Tumor-released lncRNA H19 promotes gefitinib resistance via packaging into exosomes in non-small cell lung cancer. Oncol Rep. 2018;40(6):3438–46. https://doi.org/10.3892/or.2018.6762.
Wang Z, Tan M, Chen G, Li Z, Lu X. LncRNA SOX2-OT is a novel prognostic biomarker for osteosarcoma patients and regulates osteosarcoma cells proliferation and motility through modulating SOX2. IUBMB Life. 2017;69(11):867–76. https://doi.org/10.1002/iub.1681.
Herrera-Solorio AM, Peralta-Arrieta I. LncRNA SOX2-OT regulates AKT/ERK and SOX2/GLI-1 expression, hinders therapy, and worsens clinical prognosis in malignant lung diseases. Mol Onocol. 2021;15(4):1110–29. https://doi.org/10.1002/1878-0261.12875.
Yin X, Han S, Song C, Zou H, Wei Z, Xu W, Ran J, Tang C, Wang Y, Cai Y, Hu Q, Han W. Metformin enhances gefitinib efficacy by interfering with interactions between tumor-associated macrophages and head and neck squamous cell carcinoma cells. Cell Oncol (Dordr). 2019;42(4):459–75. https://doi.org/10.1007/s13402-019-00446-y.
Wei C, Yang C, Wang S, Shi D, Zhang C, Lin X, Liu Q, Dou R, Xiong B. Crosstalk between cancer cells and tumor-associated macrophages is required for mesenchymal circulating tumor cell-mediated colorectal cancer metastasis. Mol Cancer. 2019;18(1):64. https://doi.org/10.1186/s12943-019-0976-4.
Piao YJ, Kim HS, Hwang EH, Woo J, Zhang M, Moon WK. Breast cancer cell-derived exosomes and macrophage polarization are associated with lymph node metastasis. Oncotarget. 2018;9(7):7398–410. https://doi.org/10.18632/oncotarget.23238.
Huang Y. The novel regulatory role of lncRNA-miRNA-mRNA axis in cardiovascular diseases. J Cell Mol Med. 2018;22(12):5768–75. https://doi.org/10.1111/jcmm.13866.
Funding
This study was supported by Changsha Natural Science Foundation (No. kq2014273); National Multidisciplinary Cooperative Diagnosis and Treatment Capacity Building Project for Major Diseases (Lung Cancer).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
13577_2021_572_MOESM1_ESM.tif
Supplementary figure 1. Detection of the expression of SOX2-OT. (A) The expression of SOX2-OT in BEAS-2B- and NSCLC cells-derived exosomes was detected using qRT-PCR assay. β-actin served as an internal reference. **P < 0.01 compared with ExoBEAS-2B group. (B) The expression of SOX2-OT in H1975 cells was determined using qRT-PCR to detect the interference efficiency of SOX2-OT shRNA. β-actin served as internal reference. ##P < 0.01 compared with shCtrl group. (C) qRT-PCR assay was performed to determine the expression of SOX2-OT in LPS-stimulated THP-1 cells after exosomes incubation. β-actin served as internal reference. $$P < 0.01 compared with LPS + ExoBEAS-2B group, and &&P < 0.01 contrasted with LPS + ExoH1975 group. The number of repetitions was 3 (TIF 797 KB)
Supplementary figure 2. Detection of the combination of miR-623-3p and SOX2-OT, Smad2, Smad3 as well as Smad4
. (A) RNA pull down was performed to detect the combination of SOX2-OT and miR-627-3p. (B-D) The combination of miR-627-3p and Smad2, Smad3 as well as Smad4 was determined using RNA binding protein immunoprecipitation assay. The number of repetitions was 3 (TIF 864 KB)
Rights and permissions
About this article
Cite this article
Zhou, D., Xia, Z., Xie, M. et al. Exosomal long non-coding RNA SOX2 overlapping transcript enhances the resistance to EGFR-TKIs in non-small cell lung cancer cell line H1975. Human Cell 34, 1478–1489 (2021). https://doi.org/10.1007/s13577-021-00572-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-021-00572-6