Abstract
Background
We aimed to determine the frequency of mutations in 3 common genes causing neonatal diabetes mellitus (NDM) in Isfahan.
Methods
This study was performed on 15 patients with permanent NDM (PNDM). KCNJ11, INS, and ABCC8 gene mutations were examined in all patients. Other related gene mutations were also examined if the first three mentioned mutations were negative. The patients were classified into two groups based on the time of NDM diagnosis (before 6 and 6–12 months of age).
Results
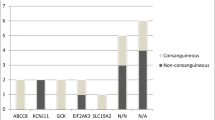
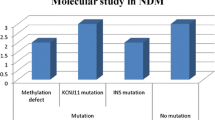
Patients with PNDM who were diagnosed before 6 months of age had more concomitant anomalies than those diagnosed between 6 and 19 months of age (p < 0.05). The genetic study did not show any mutation in ABCC8, KCNJ11, and INS genes. Evaluation of other gene mutations indicated that 3 patients (two boys and one girl) had a homozygous mutation in EIF2AK3, one patient (boy) had a homozygous mutation in PTF1A, and also one patient (boy) had a homozygous mutation in GLIS3 gene which are all considered as rare genes which could cause NDM. All of the patients with mutations were diagnosed before 6 months of age. Four out of the five patients were male and had parental consanguinity (2nd and 3rd degree).
Conclusion
We found that mutations in KCNJ11, INS, and ABCC8 are not the common cause of PNDM in our population, and mutations in EIF2AK3, PTF1A, and GLIS3 genes are more frequent. The PNDM cases and their related reported mutation were related to the syndromic form of NDM, and all of them were diagnosed before 6 months of age.

Similar content being viewed by others
References
Dahl A, Kumar S. Recent advances in neonatal diabetes. Diabetes Metab Syndr Obes Targets Ther. 2020;13:355.
Naylor RN, Greeley SAW, Bell GI, Philipson LH. Genetics and pathophysiology of neonatal diabetes mellitus. J Diabetes Investig. 2011;2(3):158–69.
Grulich-Henn J, Wagner V, Thon A, Schober E, Marg W, Kapellen TM, et al. Entities and frequency of neonatal diabetes: data from the diabetes documentation and quality management system (DPV). Diabetic Med. 2010;27(6):709–12.
Wirth M, Jellimann JM, Jellimann S, Hascoët JM. Neonatal diabetes mellitus: improved screening and early management of an underestimated disease. Clin Case Rep. 2018;6(1):18.
Babenko AP, Polak M, Cavé H, Busiah K, Czernichow P, Scharfmann R, et al. Activating mutations in the ABCC8 gene in neonatal diabetes mellitus. N Engl J Med. 2006;355(5):456–66.
Polak M, Cavé H. Neonatal diabetes mellitus: a disease linked to multiple mechanisms. Orphanet J Rare Dis. 2007;2(1):12.
Gahagan S, Kliegman R, Stanton B, Geme JS, Schor N, Behrman R. Nelson textbook of pediatrics. Philadelphia: Saunders; 2016.
Mackay DJ, Temple IK, editors. Transient neonatal diabetes mellitus type 1. American Journal of Medical Genetics Part C: Seminars in Medical Genetics; 2010: Wiley Online Library.
Zung A, Glaser B, Nimri R, Zadik Z. Glibenclamide treatment in permanent neonatal diabetes mellitus due to an activating mutation in Kir6. 2. J Clin Endocrinol Metab. 2004;89(11):5504–7.
Al-Agha AE, Ahmad IAA-H, Basnawi FAA, AL-Nasser EYF. A recessive mutation in the insulin gene in neonatal diabetes. Turk J Endocrinol Metab. 2015;19(1).
Craig M. International Society for Pediatric Adolescent Diabetes. ISPAD Clinical Practice Consensus Guidelines 2006-2007. Definition, epidemiology and classification. Pediatr Diabetes. 2006;7:343–51.
Laurenzano SE, McFall C, Nguyen L, Savla D, Coufal NG, Wright MS, Tokita M, Dimmock D, Kingsmore SF, Newfield RS. Neonatal diabetes mellitus due to a novel variant in the INS gene. Mol Case Stud. 2019;5(4):a004085.
Barbetti F, D'Annunzio G. Genetic causes and treatment of neonatal diabetes and early childhood diabetes. Best Pract Res Clin Endocrinol Metab. 2018;32(4):575–91.
Nagashima K, Tanaka D, Inagaki N. Epidemiology, clinical characteristics, and genetic etiology of neonatal diabetes in Japan. Pediatr Int. 2017;59(2):129–33.
Russo L, Iafusco D, Brescianini S, Nocerino V, Bizzarri C, Toni S, et al. The ISPED Early Diabetes Study Group. Permanent diabetes during the first year of life: multiple gene screening in 54 patients. Diabetologia. 2011;54:1693–701.
Habeb AM, Flanagan SE, Deeb A, et al. Permanent neonatal diabetes: different aetiology in Arabs compared to Europeans. Arch Dis Child. 2012;97:721–3.
Abujbara MA, Liswi MI, El-Khateeb MS, Flanagan SE, Ellard S, Ajlouni KM. Permanent neonatal diabetes mellitus in Jordan. J Pediatr Endocrinol Metab. 2014;27:879–83.
Nouri N, Nouri N, Tirgar S, et al. Consanguineous marriages in the genetic counseling centers of Isfahan and the ethical issues of clinical consultations. J Med Ethics History Med. 2017;10:12.
Habeb AM, Al-Magamsi MS, Eid IM, Ali MI, Hattersley AT, Hussain K, Ellard S. Incidence, genetics, and clinical phenotype of permanent neonatal diabetes mellitus in northwest Saudi Arabia. Pediatr Diabetes. 2012;13(6):499–505.
Al-Khawaga S, Mohammed I, Saraswathi S, Haris B, Hasnah R, Saeed A, Almabrazi H, Syed N, Jithesh P, El Awwa A, Khalifa A. The clinical and genetic characteristics of permanent neonatal diabetes (PNDM) in the state of Qatar. Mol Genet Genom Med. 2019;7(10):e00753.
Sowers CR, Wang R, Bourne RA, McGrath BC, Hu J, Bevilacqua SC, et al. The protein kinase PERK/EIF2AK3 regulates proinsulin processing not via protein synthesis but by controlling endoplasmic reticulum chaperones. J Biol Chem. 2018;293:5134–49.
Rubio-Cabezas O, Patch AM, Minton JA, Flanagan SE, Edghill EL, Hussain K, Balafrej A, Deeb A, Buchanan CR, Jefferson IG, Mutair A. Wolcott-Rallison syndrome is the most common genetic cause of permanent neonatal diabetes in consanguineous families. J Clin Endocrinol Metab. 2009;94(11):4162–70.
Demirbilek H, Arya VB, Ozbek MN, Houghton JA, Baran RT, Akar M, Tekes S, Tuzun H, Mackay DJ, Flanagan SE, Hattersley AT. Clinical characteristics and molecular genetic analysis of 22 patients with neonatal diabetes from the South-Eastern region of Turkey: predominance of non-KATP channel mutations. Eur J Endocrinol. 2015;172(6):697.
Deeb A, Habeb A, Kaplan W, Attia S, Hadi S, Osman A, Al-Jubeh J, Flanagan S, DeFranco E, Ellard S. Genetic characteristics, clinical spectrum, and incidence of neonatal diabetes in the Emirate of AbuDhabi, United Arab Emirates. Am J Med Genet Part A. 2016;170(3):602–9.
Rurale G, Persani L, Marelli F. GLIS3 and thyroid: a pleiotropic candidate gene for congenital hypothyroidism. Front Endocrinol. 2018;9:730.
Asl SN, Vakili R, Vakili S, Soheilipour F, Hashemipour M, Ghahramani S, De Franco E, Yaghootkar H. Wolcott-Rallison syndrome in Iran: a common cause of neonatal diabetes. J Pediatr Endocrinol Metab. 2019;32(6):607–13.
London S, De Franco E, Elias-Assad G, Barhoum MN, Felszer C, Paniakov M, Weiner SA, Tenenbaum-Rakover Y. Case report: neonatal diabetes mellitus caused by a novel GLIS3 mutation in twins. Front Endocrinol. 2021;12:557.
Zajec A, Trebušak Podkrajšek K, Tesovnik T, Šket R, Čugalj Kern B, Jenko Bizjan B, Šmigoc Schweiger D, Battelino T, Kovač J. Pathogenesis of type 1 diabetes: established facts and new insights. Genes. 2022;13(4):706.
DeForest N, Majithia AR. Genetics of type 2 diabetes: Implications from large-scale studies. Curr Diabetes Rep. 2022;19:1–9.
Dimitri P, Habeb A, Garbuz F, Millward A, Wallis S, Moussa K, et al. Expanding the clinical spectrum associated with GLIS3 mutations. J Clin Endocrinol Metab. 2015;100(10):E1362–E9.
Alghamdi KA, Alsaedi AB, Aljasser A, Altawil A, Kamal NM. Extended clinical features associated with novel Glis3 mutation: a case report. BMC Endocr Disord. 2017;17(1):14.
Fukuda A, Kawaguchi Y, Furuyama K, Kodama S, Horiguchi M, Kuhara T, Kawaguchi M, Terao M, Doi R, Wright CV, Hoshino M, Chiba T, Uemoto S. Reduction of Ptf1a gene dosage causes pancreatic hypoplasia and diabetes in mice. Diabetes. 2008;57:2421–31.
Vakili R, Ghahraman M, Ghaemi N, Faraji B, Ahmadi E, Abbaszadeghan MR, et al. Clinical and molecular genetic analysis of Iranian patients with neonatal diabetes demonstrating mutations in KCNJ11 gene. Iran J Neonatol IJN. 2012;3(2):85–90.
Sousa M, Bruges-Armas J. Monogenic diabetes: genetics and relevance on diabetes mellitus personalized medicine. Curr Diabetes Rev. 2020;16(8):807–19.
Author information
Authors and Affiliations
Contributions
All authors contributed to the concept and design of the study and participated actively in all stages of the work including data collection, analysis, and preparing the first draft of the paper, and all of them have read and confirmed the manuscript.
Corresponding authors
Ethics declarations
Data of patients between the years 2006 and 2017 that had been previously registered were collected. The current study was confirmed by the research committee and regional ethics committee of Isfahan University of Medical Sciences with research and ethics codes of 397529 and IR.MUI.MED.REC.1398.168, respectively. The protocol and methods of the current study were described in details for parents, and written informed consent was obtained from the parents of each patient.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hashemipour, M., Mostofizadeh, N., Ghasemi, M. et al. Molecular genetic analysis of the insulin gene variants in Iranian patients with permanent neonatal diabetes. Int J Diabetes Dev Ctries 43, 821–828 (2023). https://doi.org/10.1007/s13410-022-01152-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13410-022-01152-6