Abstract
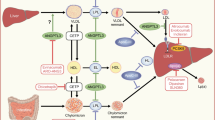
Obesity, insulin resistance (IR), inflammation, and progressive decline in pancreatic β cell function are major features of type 2 diabetes mellitus (T2DM). We aimed to investigate the effect of co-administration of celecoxib (CE) with glimepiride (GL) in the treatment of obese T2DM patients. Body Mass Index (BMI), serum glucose, C-peptide, lipid profile, adiponectin, tumor necrosis factor-α (TNF-α), visfatin, and leptin levels were determined in 40 obese T2DM patients before and after treatment with GL alone or in combination with a selective cyclooxygenase-2 (COX-2) inhibitor CE for 3 months. Homeostasis model assessment of insulin resistance (HOMA2-IR) and atherogenic index (AI) was calculated. Increased levels of serum glucose, C-peptide, total cholesterol (TCH), low-density lipoprotein (LDL-C), triglycerides (TGs), visfatin, TNF-α, leptin, AI, and HOMA2-IR shown in obese diabetic patients were significantly decreased after co-treatment with GL plus CE compared to patients who received GL alone. On the other hand, adiponectin levels showed a significant increase after treatment. The obtained results demonstrate that targeting inflammation using celecoxib with glimepiride improves insulin resistance, glycemia, and inflammatory process in obese type 2 diabetics.
Similar content being viewed by others
Abbreviations
- BG:
-
Blood glucose
- COPD:
-
Chronic obstructive pulmonary disease
- COX-2:
-
Cyclooxygenase-2
- CVD:
-
Cardiovascular disease
- ELISA:
-
Enzyme-linked immunosorbent assay
- GIT:
-
Gastrointestinal tract
- HDL-C:
-
High-density lipoprotein
- HOMA2-IR:
-
Homeostasis model assessment of insulin resistance
- HOMA2-β:
-
Homeostasis model assessment of beta cell functionality
- LDL-C:
-
Low-density lipoprotein
- T2DM:
-
Type 2 diabetes mellitus
- TCH:
-
Total cholesterol
- TGs:
-
Triglycerides
- TNF-α:
-
Tumor necrosis factor alpha
References
Cernea S, Dobreanu M. Diabetes and beta cell function: from mechanisms to evaluation and clinical implications. Biochem Med (Zagreb). 2013;23(3):266–80.
Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444:860–7.
Ye J. Mechanisms of insulin resistance in obesity. Front Med. 2013;7(1):14–24.
Uslu S, Kebapçi N, Kara M, Bal C. Relationship between adipocytokines and cardiovascular risk factors in patients with type 2 diabetes mellitus. Exp Ther Med. 2012;4(1):113–20.
Raucci R, Rusolo F, Sharma A, Colonna G, Castello G, Costantini S. Functional and structural features of adipokine family. Cytokine. 2013;61(1):1–14.
Konheim YL, Wolford JK. Association of a promoter variant in the inducible cyclooxygenase-2 gene (PTGS2) with type 2 diabetes mellitus in Pima Indians. Hum Genet. 2003;113:377–81.
Helmersson J, Vessby B, Larsson A, Basu S. Association of type 2 diabetes with cyclooxygenase-mediated inflammation and oxidative stress in an elderly population. Circulation. 2004;109:1729–34.
Hsieh PS, Tsai HC, Kuo CH, Chan JY, Shyu JF, Cheng WT, et al. Selective COX-2 inhibition improves whole body and muscular insulin resistance in fructose-fed rats. Eur J Clin Invest. 2008;38(11):812–9.
Hsieh PS, Jin JS, Chiang CF, Chan PC, Chen CH, Shih KC. COX-2-mediated inflammation in fat is crucial for obesity-linked insulin resistance and fatty liver. Obesity (Silver Spring). 2009;17:1150–7.
Alpert E, Gruzman A, Tennenbaum T, Sasson S. Selective cyclooxygenase-2 inhibitors stimulate glucose transport in L6 myotubes in a protein kinase C[delta]-dependent manner. Biochem Pharmacol. 2007;73(3):368–77.
Coll T, Palomer X, Blanco-Vacca F, Escola-Gil JC, Sanchez RM, Laguna JC, et al. Cyclooxygenase 2 inhibition exacerbates palmitate-induced inflammation and insulin resistance in skeletal muscle cells. Endocrinology. 2010;151(2):537–48.
Gokalp D, Bahceci M, Ozmen S, Arikan S, Tuzcu A, Danıs R. Adipocyte volumes and levels of adipokines in diabetes and obesity. Diabetes Metab Syndr Clin Res Rev. 2008;2:253–8.
Aubrée-Lecat A, Hervagault C, Delacour A, Beaude P, Bourdillon C, Remy MH. Direct electrochemical determination of glucose oxidase in biological samples. Anal Biochem. 1989;178(2):427–30.
Gonen G, Rubenstein AH. Haemoglobin A1 and diabetes mellitus. Diabetologia. 1978;15(1):1–8.
Kim MK. Crystal structure of visfatin/pre-B cell colony-enhancing factor 1/nicotinamide phosphoribosyltransferase, free and in complex with the anti- cancer agent FK-866. J Mol Biol. 2006;362(1):66–77.
Brouckaert P, Libert C, Everaerdt B, Takahashi N, Cauwels A, Fiers W. Tumor necrosis factor, its receptors and the connection with interleukin 1 and interleukin 6. Immunobiology. 1993;187(3–5):317–29.
Tsao TS, Lodish HF, Fruebis J. ACRP30, a new hormone controlling fat and glucose metabolism. Eur J Pharmacol. 2002;440(2-3):213–21.
Turkington RW, Estkowski A, Link M. Secretion of insulin or connecting peptide: a predictor of insulin dependence of obese "diabetics". Arch Intern Med. 1982;142(6):1102–5.
Wiesner G, Vaz M, Collier G, Seals D, Kaye D, Jennings G, et al. Leptin is released from the human brain: influence of adiposity and gender. J Clin Endocrinol Metab. 1999;84(7):2270–4.
Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care. 2004;27(6):1487–95.
Bucolo G, David H. Quantitative determination of serum triglycerides by the use of enzymes. Clin Chem. 1973;19(5):476–82.
Allian CC, Poon LS, Chan CS, Richmond W, Fu PC. Enzymatic determination of total serum cholesterol. Clin Chem. 1974;20(4):470–5.
Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma without use of preparative ultracentrifuge. Clin Chem. 1972;18(6):499–502.
Hermans MP, Ahn SA, Rousseau MF. The atherogenic dyslipidemia ratio [log(TG)/HDL-C] is associated with residual vascular risk, beta-cell function loss and microangiopathy in type 2 diabetes females. Lipids Health Dis. 2012;11(132):1–8.
Aroor A, McKarns S, Nistala R, Demarco V, Gardner M, Garcia-Touza M, et al. DPP-4 inhibitors as therapeutic modulators of immune cell function and associated cardiovascular and renal insulin resistance in obesity and diabetes. Cardiorenal Med. 2013;3(1):48–56.
Wajchenberg BL. Beta-cell failure in diabetes and preservation by clinical treatment. Endocr Rev. 2007;28(2):187–218.
Tian YF, Hsia TL, Hsieh CH, Huang DW, Chen CH, Hsieh PS. The importance of cyclooxygenase 2-mediated oxidative stress in obesity-induced muscular insulin resistance in high-fat-fed rats. Life Sci. 2011;89(3–4):107–14.
Liu TT, Shih KC, Kao CC, Cheng WT, Hsieh PS. Importance of cyclooxygenase 2-mediated low-grade inflammation in the development of fructose-induced insulin resistance in rats. Chin J Physiol. 2009;52(2):65–71.
Sone H, Takahashi A, Yamada N. Ibuprofen-related hypoglycemia in a patient receiving sulfonylurea. Ann Intern Med. 2001;134:344.
Perucca E. Drug interactions with nimesulide. Drugs. 1993;46 Suppl 1:79–82.
Matsuda M, Shimomura I. Roles of adiponectin and oxidative stress in obesity-associated metabolic and cardiovascular diseases. Rev Endocr Metab Disord. 2014;15(1):1–10.
Shoelson SE, Lee J, Goldfine AB. Inflammation and insulin resistance. J Clin Invest. 2006;116(7):1793–801.
Jacob S, Machann J, Rett K, Brechtel K, Volk A, Renn W, et al. Association of increased intramyocellular lipid content with insulin resistance in lean nondiabetic offspring of type 2 diabetic subjects. Diabetes. 1999;48(5):1113–9.
Jeusette IC, Lhoest ET, Istasse LP, Diez MO. Influence of obesity on plasma lipid and lipoprotein concentrations in dogs. Am J Vet Res. 2005;66(1):81–6.
Acknowledgments
The author gratefully acknowledges Prof. Dr. Osama M. Ibrahim, Chairman of the Clinical Pharmacy Department, Faculty of Pharmacy, Tanta University, for his support and helpful assistance in instruments supply and lab availability.
Conflict of interest
The authors declared that there are no conflicts of interest.
Author’s contribution
Hoda El-Bahrawy did the conception and design of the study and final approval of the version to be published. Sahar Hegazy followed up the patients and drafted the article or revised it critically for important intellectual content. Wael Farrag took the detailed history from patients, examined the patients clinically, and analyzed and interpreted the data. Rehab Werida followed up the patients and collected samples from them; performed anthropometric evaluations and biochemical assay for selected parameters; analyzed obtained data; and drafted the article or revised it critically for important intellectual content, and gave final approval of the version to be published.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Bahrawy, H., Hegazy, S., Farrag, W. et al. Targeting inflammation using celecoxib with glimepiride in the treatment of obese type 2 diabetic Egyptian patients. Int J Diabetes Dev Ctries 37, 97–102 (2017). https://doi.org/10.1007/s13410-015-0355-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13410-015-0355-7