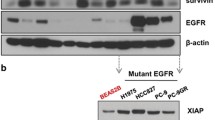
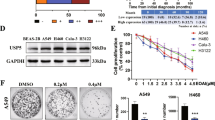
Abstract
Purpose
Chemoresistance is a primary factor for treatment failure and tumor recurrence in non-small cell lung cancer (NSCLC) patients. The oncoprotein survivin is commonly upregulated in human malignancies and is associated with poor prognosis, but its effect on carcinogenesis and chemoresistance in NSCLC is not yet evident, and to explore an effective inhibitor targeting survivin expression is urgently needed.
Methods
The protumor characteristics of survivin and antitumor activities of bergenin in NSCLC cells were examined by MTS, colony formation assays, immunoblot, immunohistochemistry, and in vivo xenograft development.
Results
Survivin was upregulated in non-small cell lung cancer (NSCLC) tissues, while its depletion inhibited NSCLC tumorigenesis. The current study focused on bergenin, identifying its effective antitumor effect on NSCLC cells both in vivo and in vitro. The results showed that bergenin could inhibit cell proliferation and induce the intrinsic pathway of apoptosis via downregulating survivin. Mechanistically, bergenin reduced the phosphorylation of survivin via inhibiting the Akt/Wee1/CDK1 signaling pathway, thus resulting in enhanced interaction between survivin and E3 ligase Fbxl7 to promote survivin ubiquitination and degradation. Furthermore, bergenin promoted chemoresistance in NSCLC cells re-sensitized to pemetrexed treatment.
Conclusions
Survivin overexpression is required for maintaining multiple malignant phenotypes of NSCLC cells. Bergenin exerts a potent antitumor effect on NSCLC via targeting survivin, rendering it a promising agent for the treatment of NSCLC.








Similar content being viewed by others
Data availability
The datasets used and analyzed in this study are available from the corresponding authors on request.
References
V. Arora, H.H. Cheung, S. Plenchette, O.C. Micali, P. Liston, R.G. Korneluk, Degradation of survivin by the X-linked inhibitor of apoptosis (XIAP)-XAF1 complex. J. Biol. Chem. 282(36), 26202–26209 (2007)
F. Bray, J. Ferlay, I. Soerjomataram, R.L. Siegel, L.A. Torre, A. Jemal, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: Cancer J. Clin. 68(6), 394–424 (2018)
D. Camuzi, L.A. Buexm, Sd.Q.C. Lourenço, R. Grazziotin, S. Guaraldi, P. Valverde, D. Rapozo, J.M. Brooks, H. Mehanna, L.F. Ribeiro Pinto, S.C. Soares-Lima, Body hypomethylation is frequent in tumors from the digestive and respiratory tracts and is associated with risk-factor exposure. Int. J. Mol. Sci. 23(14), 7801 (2022)
E. Cella, L. Zullo, S. Marconi, G. Rossi, S. Coco, C. Dellepiane, A. Alama, L. Rozeboom, E. Bennicelli, F. Parisi, G. Sacco, G. Barletta, L. Zinoli, M. Tagliamento, P. Pronzato, C. Genova, Immunotherapy-chemotherapy combinations for non-small cell lung cancer: current trends and future perspectives. Expert Opin. Biol. Ther. 22(10), 1259–1273 (2022)
M. Césaire, J. Montanari, H. Curcio, D. Lerouge, R. Gervais, P. Demontrond, J. Balosso, F. Chevalier, Radioresistance of non-small cell lung cancers and therapeutic perspectives. Cancers (Basel) 14(12), 2829 (2022)
S. Chaithongyot, M. Naumann, Helicobacter pylori-induced reactive oxygen species direct turnover of CSN-associated STAMBPL1 and augment apoptotic cell death. Cell. Mol. Life Sci.: CMLS 79(2), 86 (2022)
A.P. Chandrasekaran, A. Tyagi, N. Poondla, N. Sarodaya, J.K. Karapurkar, K. Kaushal, C.-H. Park, S.-H. Hong, K.-S. Kim, S. Ramakrishna, Dual role of deubiquitinating enzyme USP19 regulates mitotic progression and tumorigenesis by stabilizing survivin. Mol. Ther.: J. Am. Soc. Gene Ther. 30(11), 3414–3429 (2022)
A.J. Cooper, L.V. Sequist, J.J. Lin, Third-generation EGFR and ALK inhibitors: mechanisms of resistance and management. Nat. Rev. Clin. Oncol. 19(8), 499–514 (2022)
X. Ding, Y. Gu, M. Jin, X. Guo, S. Xue, C. Tan, J. Huang, W. Yang, M. Xue, Q. Zhou, W. Wang, Y. Zhang, The deubiquitinating enzyme UCHL1 promotes resistance to pemetrexed in non-small cell lung cancer by upregulating thymidylate synthase. Theranostics 10(13), 6048–6060 (2020)
T. Dohi, E. Beltrami, N.R. Wall, J. Plescia, D.C. Altieri, Mitochondrial survivin inhibits apoptosis and promotes tumorigenesis. J. Clin. Investig. 114(8), 1117–1127 (2004)
T. Dohi, F. Xia, D.C. Altieri, Compartmentalized phosphorylation of IAP by protein kinase A regulates cytoprotection. Mol. Cell 27(1), 17–28 (2007)
N. Duma, R. Santana-Davila, J.R. Molina, Non-small cell lung cancer: epidemiology, screening, diagnosis, and treatment. Mayo Clin. Proc. 94(8), 1623–1640 (2019)
A. FäldtBeding, P. Larsson, K. Helou, Z. Einbeigi, T.Z. Parris, Pan-cancer analysis identifies BIRC5 as a prognostic biomarker. BMC Cancer 22(1), 322 (2022)
R.N. Fuller, J. Kabagwira, P.A. Vallejos, A.D. Folkerts, N.R. Wall, Survivin splice variant 2β enhances pancreatic ductal adenocarcinoma resistance to gemcitabine. Onco. Targets Ther. 15, 1147–1160 (2022)
H. Garg, P. Suri, J.C. Gupta, G.P. Talwar, S. Dubey, Survivin: a unique target for tumor therapy. Cancer Cell Int. 16, 49 (2016)
R. George, S. Hehlgans, M. Fleischmann, C. Rödel, E. Fokas, F. Rödel, Advances in nanotechnology-based platforms for survivin-targeted drug discovery. Expert Opin. Drug Discov. 17(7), 733–754 (2022)
M. Hurtado, U.T. Sankpal, A. Kaba, S. Mahammad, J. Chhabra, D.T. Brown, R.K. Gurung, A.A. Holder, J.K. Vishwanatha, R. Basha, Novel survivin inhibitor for suppressing pancreatic cancer cells growth via downregulating Sp1 and Sp3 transcription factors. Cell. Physiol. Biochem.: Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 51(4), 1894–1907 (2018)
W. Jiang, W. Wang, L. Sun, Y. Xiao, T. Ma, B. Li, X. Yan, Y. Wu, H. Li, J. Lian, F. He, (-)-Gossypol enhances the anticancer activity of epirubicin via downregulating survivin in hepatocellular carcinoma. Chem. Biol. Interact. 364, 110060 (2022)
G.S. Jones, D.R. Baldwin, Recent advances in the management of lung cancer. Clin. Med. (London, England) 18(Suppl 2), s41–s46 (2018)
S. Khan, H.F. Bennit, N.R. Wall, The emerging role of exosomes in survivin secretion. Histol. Histopathol. 30(1), 43–50 (2015)
F. Li, I. Aljahdali, X. Ling, Cancer therapeutics using survivin BIRC5 as a target: what can we do after over two decades of study? J. Exp. Clin. Cancer Res. 38(1), 368 (2019)
F. Li, J. Yang, N. Ramnath, M.M. Javle, D. Tan, Nuclear or cytoplasmic expression of survivin: what is the significance? Int. J. Cancer 114(4), 509–512 (2005)
M. Li, F. Gao, X. Li, Y. Gan, S. Han, X. Yu, H. Liu, W. Li, Correction to: stabilization of MCL-1 by E3 ligase TRAF4 confers radioresistance. Cell Death Dis. 14(1), 36 (2023)
M. Li, F. Gao, X. Yu, Q. Zhao, L. Zhou, W. Liu, W. Li, Promotion of ubiquitination-dependent survivin destruction contributes to xanthohumol-mediated tumor suppression and overcomes radioresistance in human oral squamous cell carcinoma. J. Exp. Clin. Canc. Res. 39(1), 88 (2020)
M. Li, H. Liu, Q. Zhao, S. Han, L. Zhou, W. Liu, W. Li, F. Gao, Targeting Aurora B kinase with Tanshinone IIA suppresses tumor growth and overcomes radioresistance. Cell Death Dis. 12(2), 152 (2021)
W. Li, X. Yu, Z. Xia, X. Yu, L. Xie, X. Ma, H. Zhou, L. Liu, J. Wang, Y. Yang, H. Liu, Repression of Noxa by Bmi1 contributes to deguelin-induced apoptosis in non-small cell lung cancer cells. J. Cell Mol. Med. 22(12), 6213–6227 (2018)
X. Li, Y. Wang, J. Liang, Z. Bi, H. Ruan, Y. Cui, L. Ma, Y. Wei, B. Zhou, L. Zhang, H. Zhou, C. Yang, Bergenin attenuates bleomycin-induced pulmonary fibrosis in mice via inhibiting TGF-β1 signaling pathway. Phytother. Res. 35(10), 5808–5822 (2021)
Y. Li, L.-R. He, Y. Gao, N.-N. Zhou, Y. Liu, X.-K. Zhou, J.-F. Liu, X.-Y. Guan, N.-F. Ma, D. Xie, CHD1L contributes to cisplatin resistance by upregulating the ABCB1-NF-κB axis in human non-small-cell lung cancer. Cell Death Dis. 10(2), 99 (2019)
R. Madaan, R.K. Singla, S. Kumar, A.K. Dubey, D. Kumar, P. Sharma, R. Bala, S. Singla, B. Shen, Bergenin - A biologically active scaffold: nanotechnological perspectives. Curr. Top. Med. Chem. 22(2), 132–149 (2022)
H. Mamdani, S. Matosevic, A.B. Khalid, G. Durm, S.I. Jalal, Immunotherapy in lung cancer: current landscape and future directions. Front. Immunol. 13, 823618 (2022)
D. Martínez-García, N. Manero-Rupérez, R. Quesada, L. Korrodi-Gregório, V. Soto-Cerrato, Therapeutic strategies involving survivin inhibition in cancer. Med. Res. Rev. 39(3), 887–909 (2019)
H.-Y. Min, H.-Y. Lee, Mechanisms of resistance to chemotherapy in non-small cell lung cancer. Arch. Pharmacal. Res. 44(2), 146–164 (2021)
H.-Y. Min, H.J. Lee, Y.-A. Suh, H. Pei, H. Kwon, H.-J. Jang, H.J. Yun, H.-G. Moon, H.-Y. Lee, Targeting epidermal growth factor receptor in paclitaxel-resistant human breast and lung cancer cells with upregulated glucose-6-phosphate dehydrogenase. Br. J. Cancer 127(4), 661–674 (2022)
X.W. Nan, L.H. Gong, X. Chen, H.H. Zhou, P.P. Ye, Y. Yang, Z.H. Xing, M.N. Wei, Y. Li, S.T. Wang, K. Liu, Z. Shi, X.J. Yan, Survivin promotes piperlongumine resistance in ovarian cancer. Front. Oncol. 9, 1345 (2019)
D.S. O’Connor, D. Grossman, J. Plescia, F. Li, H. Zhang, A. Villa, S. Tognin, P.C. Marchisio, D.C. Altieri, Regulation of apoptosis at cell division by p34cdc2 phosphorylation of survivin. Proc. Natl. Acad. Sci. U.S.A. 97(24), 13103–13107 (2000)
R. Peery, Q. Cui, K. Kyei-Baffour, S. Josephraj, C. Huang, Z. Dong, M. Dai, J.-T. Zhang, J.-Y. Liu, A novel survivin dimerization inhibitor without a labile hydrazone linker induces spontaneous apoptosis and synergizes with docetaxel in prostate cancer cells. Bioorg. Med. Chem. 65, 116761 (2022)
A. Rafatmanesh, M. Behjati, N. Mobasseri, M. Sarvizadeh, T. Mazoochi, M. Karimian, The survivin molecule as a double-edged sword in cellular physiologic and pathologic conditions and its role as a potential biomarker and therapeutic target in cancer. J. Cell. Physiol. 235(2), 725–744 (2020)
S.A. Salama, M.M. Elshafey, Cross-talk between PPARγ, NF-κB, and p38 MAPK signaling mediates the ameliorating effects of bergenin against the iron overload-induced hepatotoxicity. Chem. Biol. Interact. 368, 110207 (2022)
L. Shi, W. Zhu, Y. Huang, L. Zhuo, S. Wang, S. Chen, B. Zhang, B. Ke, Cancer-associated fibroblast-derived exosomal microRNA-20a suppresses the PTEN/PI3K-AKT pathway to promote the progression and chemoresistance of non-small cell lung cancer. Clin. Transl. Med. 12(7), e989 (2022)
R.K. Singla, K. Dhonchak, R.K. Sodhi, M. ArockiaBabu, J. Madan, R. Madaan, S. Kumar, R. Sharma, B. Shen, Bergenin ameliorates cognitive deficits and neuropathological alterations in sodium azide-induced experimental dementia. Front. Pharmacol. 13, 994018 (2022)
Q. Sun, X. Wei, Z. Wang, Y. Zhu, W. Zhao, Y. Dong, Primary and acquired resistance against immune check inhibitors in non-small cell lung cancer. Cancers (Basel) 14(14), 3294 (2022)
M. Šutić, A. Vukić, J. Baranašić, A. Försti, F. Džubur, M. Samaržija, M. Jakopović, L. Brčić, J. Knežević, Diagnostic, predictive, and prognostic biomarkers in Non-Small Cell Lung Cancer (NSCLC) management. J. Pers. Med. 11(11), 1102 (2021)
S. Tan, Q. Gou, W. Pu, C. Guo, Y. Yang, K. Wu, Y. Liu, L. Liu, Y.-Q. Wei, Y. Peng, Circular RNA F-circEA produced from EML4-ALK fusion gene as a novel liquid biopsy biomarker for non-small cell lung cancer. Cell Res. 28(6), 693–695 (2018)
W.D. Travis, E. Brambilla, A.G. Nicholson, Y. Yatabe, J.H.M. Austin, M.B. Beasley, L.R. Chirieac, S. Dacic, E. Duhig, D.B. Flieder, K. Geisinger, F.R. Hirsch, Y. Ishikawa, K.M. Kerr, M. Noguchi, G. Pelosi, C.A. Powell, M.S. Tsao, I. Wistuba, The 2015 World Health Organization Classification of Lung Tumors: Impact of Genetic, Clinical and Radiologic Advances Since the 2004 Classification. J. Thorac. Oncol.: Off. Publ. Int. Assoc. Study Lung Cancer 10(9), 1243–1260 (2015)
Y. Wang, X. Li, H. Wang, G. Zhang, CircCAMSAP1 promotes non-small cell lung cancer proliferation and inhibits cell apoptosis by sponging miR-1182 and regulating BIRC5. Bioengineered 13(2), 2428–2439 (2022)
N.M. Warrier, R.K. Krishnan, V. Prabhu, R.C. Hariharapura, P. Agarwal, P. Kumar, Survivin inhibition by piperine sensitizes glioblastoma cancer stem cells and leads to better drug response. Int. J. Mol. Sci. 23(14), 7604 (2022)
S.P. Wheatley, A.J. Henzing, H. Dodson, W. Khaled, W.C. Earnshaw, Aurora-B phosphorylation in vitro identifies a residue of survivin that is essential for its localization and binding to inner centromere protein (INCENP) in vivo. J. Biol. Chem. 279(7), 5655–5660 (2004)
S.M. Woo, S. Kim, S.U. Seo, S. Kim, J.-W. Park, G. Kim, Y.-R. Choi, K. Hur, T.K. Kwon, Inhibition of USP1 enhances anticancer drugs-induced cancer cell death through downregulation of survivin and miR-216a-5p-mediated upregulation of DR5. Cell Death Dis. 13(9), 821 (2022)
Y. Wu, Y. Zhang, D. Wang, Y. Zhang, J. Zhang, Y. Zhang, L. Xu, T. Wang, S. Wang, Q. Zhang, F. Liu, M.Y. Zaky, Q. Li, Q. Sun, D. Guo, S. Liu, L. Zou, Q. Yang, H. Liu, USP29 enhances chemotherapy-induced stemness in non-small cell lung cancer via stabilizing Snail1 in response to oxidative stress. Cell Death Dis. 11(9), 796 (2020)
W. Xiao, P. Ahluwalia, L. Wang, J. Howard, R. Kolhe, A.M. Rojiani, M.V. Rojiani, TIMP-1 dependent modulation of metabolic profiles impacts chemoresistance in NSCLC. Cells 11(19), 3036 (2022)
F. Xu, J. Gu, C. Lu, W. Mao, L. Wang, Q. Zhu, Z. Liu, Y. Chu, R. Liu, D. Ge, Calpain-2 enhances non-small cell lung cancer progression and chemoresistance to paclitaxel via EGFR-pAKT pathway. Int. J. Biol. Sci. 15(1), 127–137 (2019)
S. Yang, Z. Yu, L. Wang, T. Yuan, X. Wang, X. Zhang, J. Wang, Y. Lv, G. Du, The natural product bergenin ameliorates lipopolysaccharide-induced acute lung injury by inhibiting NF-kappaB activition. J. Ethnopharmacol. 200, 147–155 (2017)
X. Yu, R. Wang, Y. Zhang, L. Zhou, W. Wang, H. Liu, W. Li, Skp2-mediated ubiquitination and mitochondrial localization of Akt drive tumor growth and chemoresistance to cisplatin. Oncogene 38(50), 7457–7472 (2019)
M. Yuan, L.-L. Huang, J.-H. Chen, J. Wu, Q. Xu, The emerging treatment landscape of targeted therapy in non-small-cell lung cancer. Signal Transduct. Target. Ther. 4, 61 (2019)
Q. Zeng, T. Zhou, F. Zhao, D. Xiong, B. He, Q. Hua, M. Lin, L. Deng, X. Sang, W. Xie, J. Chen, Z. Wang, L. Ren, Z. Luo, X. Huang, W. Liu, S. Tang, p62-Nrf2 regulatory loop mediates the anti-pulmonary fibrosis effect of bergenin. Antioxidants (Basel) 11(2), 307 (2022)
J. Zhang, Y. Nishimoto, H. Tokuda, N. Suzuki, K. Yasukawa, W. Kitdamrongtham, H. Akazawa, A. Manosroi, J. Manosroi, T. Akihisa, Cancer chemopreventive effect of bergenin from Peltophorum pterocarpum wood. Chem. Biodivers. 10(10), 1866–1875 (2013)
Y. Zhang, Y. Sun, Y. Jia, Q. Zhang, P. Zhu, X. Ma, α5-nAChR and survivin: Two potential biological targets in lung adenocarcinoma. J. Cell. Physiol. 236(3), 1787–1797 (2021)
H. Zhou, Q. Guan, X. Hou, L. Liu, L. Zhou, W. Li, H. Liu, Epithelial-mesenchymal reprogramming by KLF4-regulated Rictor expression contributes to metastasis of non-small cell lung cancer cells. Int. J. Biol. Sci. 18(13), 4869–4883 (2022)
Y. Zhu, Z. Ouyang, H. Du, M. Wang, J. Wang, H. Sun, L. Kong, Q. Xu, H. Ma, Y. Sun, New opportunities and challenges of natural products research: When target identification meets single-cell multiomics. Acta Pharm. Sinica B 12(11), 4011–4039 (2022)
Funding
This work was supported by the National Natural Science Foundation of China (No. 81972837 and 82003203), the Natural Science Foundation of Hunan Province (Nos. 2021JJ31011 and 2021JJ41058) and the Hunan Province Natural Science Foundation Youth Fund Project, China (No. 2021JJ40943).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Material preparation, data collection and analysis were performed by [Xiaoying Li], [Qi Liang], [Li Zhou], [Gaoyan Deng], [Yeqing Xiao], [Yu Gan], [Shuangze Han], [Jinzhuang Liao], [Ruirui Wang], [Xiang Qing] and [Wei Li]. Writing, review, and/or revision of the manuscript was written by [Xiaoying Li], [Qi Liang], [Li Zhou], [Xiang Qing] and [Wei Li]. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
The animal experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of the Third Xiangya Hospital of Central South University (Changsha, China). Informed consent was obtained from all patients.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, X., Liang, Q., Zhou, L. et al. Survivin degradation by bergenin overcomes pemetrexed resistance. Cell Oncol. 46, 1837–1853 (2023). https://doi.org/10.1007/s13402-023-00850-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-023-00850-5