Abstract
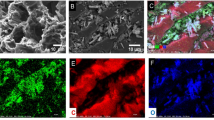
The utilization of renewable and cost-effective biomass for the production of activated carbon represents an innovative approach to environmental remediation. In this work, environmentally friendly carbon materials derived from cocopeat were employed to create a cocopeat-based magnetic activated carbon (CPAC-Fe3O4) nanocomposite for the removal of mercury from aqueous solutions. The CPAC-Fe3O4 nanocomposite underwent comprehensive characterization using SEM, FTIR, BET, XRD, and VSM analyses. The optimization process revealed a maximum adsorption capacity of 204.08 mg/g under specific conditions: initial Hg concentration of 20 mg/L, pH of 6, temperature of 25 °C, and adsorbent dose of 0.01 g within 60 min. Isotherm and kinetic modeling exhibited strong agreement with the Freundlich isotherm (0.9749) and pseudo-second-order (0.9997) kinetic models, indicating a favorable chemisorption process. Furthermore, thermodynamic analysis suggested that the adsorption process is endothermic and spontaneous. The adsorption mechanism was elucidated based on FTIR analysis. The results highlight the CPAC-Fe3O4 nanocomposite as a promising and sustainable candidate for effective water purification.








Similar content being viewed by others
Data availability
Not applicable.
References
Abyar H, Nowrouzi M, Rostami A (2022) A comprehensive study of biological phosphorus removal systems from economic and environmental perspectives based on the optimization approach. Environ Technol Innov 28:102811
Nowrouzi M, Abyar H, Rohani S (2023) A comparison of nitrogen removal systems through cost-coupled life cycle assessment and energy efficiency analysis. Sci Total Environ 858:159787
J Rajapakse M Otoo G Danso 2023 Progress in delivering SDG6: safe water and sanitation Cambridge Prisms: Water 1 e6
Barati AA, Pour MD, Sardooei MA (2023) Water crisis in Iran: a system dynamics approach on water, energy, food, land and climate (WEFLC) nexus. Sci Total Environ 882:163549
AMMA Caretta, RBM Arfanuzzaman, SMR Morgan, M Kumar Water. In: Climate change 2022: impacts, adaptation, and vulnerability. Contribution of Working Group II to the in: I.P.o.C. Change (Ed.) 2022
He C, Liu Z, Wu J, Pan X, Fang Z, Li J, Bryan BA (2021) Future global urban water scarcity and potential solutions. Nat Commun 12(1):4667
Mekonnen MM, Hoekstra AY (2016) Four billion people facing severe water scarcity. Sci Adv 2(2):e1500323
Dixit A, Madhav S, Mishra R, Srivastav AL, Garg P (2022) Impact of climate change on water resources, challenges and mitigation strategies to achieve sustainable development goals. Arab J Geosci 15(14):1296
Mikulčić H, Baleta J, Wang X, Duić N, Dewil R (2022) Sustainable development in period of climate crisis. J Environ Manage 303:114271
Stringer LC, Mirzabaev A, Benjaminsen TA, Harris RM, Jafari M, Lissner TK, Stevens N, Tirado-von Der Pahlen C (2021) Climate change impacts on water security in global drylands. One Earth 4(6):851–864
Attari M, Bukhari SS, Kazemian H, Rohani S (2017) A low-cost adsorbent from coal fly ash for mercury removal from industrial wastewater. J Environ Chem Eng 5(1):391–399
Monjane-Mabuie A, Mondlane-Milisse A, Pedro O, Leão-Buchir J, Correia D (2022) Mercury pollution assessment and metallothionein gene expression in tilapia (Oreochromis mossambicus): a case study of Revuè River in Manica, Mozambique. Rendiconti Lincei Scienze Fisiche e Naturali 33(3):513–526
Zarei S, Raanaei H, Niad M (2023) Investigation of mercury removal by Fe3O4@ SiO2-NH2-GO-NC as magnetic nanocomposite. Inorg Chem Commun 152:110665
Adibkia M, Tajjarod S, Talavari A, Noviery E, Azimi Maleki S (2017) The effect of amino acid coating on the performance of magnetic nanoparticles for the elimination of mercury from waste. Chem Metall Eng J 4(1):1–11
EC Emenike, AG Adeniyi, KO Iwuozor, CJ Okorie, AU Egbemhenghe, PE Omuku, KC Okwu, OD Saliu 2023 A critical review on the removal of mercury (Hg2+) from aqueous solution using nanoadsorbents Environ Nanotechnol Monit Manag 100816
Behjati M, Baghdadi M, Karbassi A (2018) Removal of mercury from contaminated saline wasters using dithiocarbamate functionalized-magnetic nanocomposite. J Environ Manage 213:66–78
EC Emenike, KO Iwuozor, SU Anidiobi Heavy metal pollution in aquaculture: sources, impacts and mitigation techniques Biol Trace Elem Res (2021) 1–17
Awual MR (2017) Novel nanocomposite materials for efficient and selective mercury ions capturing from wastewater. Chem Eng J 307:456–465
Chen J, Wang Y, Wei X, Xu P, Xu W, Ni R, Meng J (2018) Magnetic solid-phase extraction for the removal of mercury from water with ternary hydrosulphonyl-based deep eutectic solvent modified magnetic graphene oxide. Talanta 188:454–462
Igwegbe CA, Obiora-Okafo IA, Iwuozor KO, Ghosh S, Kurniawan SB, Rangabhashiyam S, Kanaoujiya R, Ighalo JO (2022) Treatment technologies for bakers’ yeast production wastewater. Environ Sci Pollut Res 29(8):11004–11026
Iwuozor KO (2019) Prospects and challenges of using coagulation-flocculation method in the treatment of effluents. Adv J Chem A 2(2):105–127
Saleh TA, Mustaqeem M, Khaled M (2022) Water treatment technologies in removing heavy metal ions from wastewater: a review. Environ Nanotechnol Monit Manag 17:100617
Talebi J, Halladj R, Askari S (2010) Sonochemical synthesis of silver nanoparticles in Y-zeolite substrate. J Mater Sci 45:3318–3324
Yaqoob AA, Ahmad H, Parveen T, Ahmad A, Oves M, Ismail IM, Qari HA, Umar K, Mohamad Ibrahim MN (2020) Recent advances in metal decorated nanomaterials and their various biological applications: a review. Front Chem 8:341
Nejati B, Adami P, Bozorg A, Tavasoli A, Mirzahosseini AH (2020) Catalytic pyrolysis and bio-products upgrading derived from Chlorella vulgaris over its biochar and activated biochar-supported Fe catalysts. J Anal Appl Pyrol 152:104799
Nowrouzi M, Younesi H, Bahramifar N (2017) High efficient carbon dioxide capture onto as-synthesized activated carbon by chemical activation of Persian Ironwood biomass and the economic pre-feasibility study for scale-up. J Clean Prod 168:499–509
Zhao Y, Xia K, Zhang Z, Zhu Z, Guo Y, Qu Z (2019) Facile synthesis of polypyrrole-functionalized CoFe2O4@ SiO2 for removal for Hg (II). Nanomaterials 9(3):455
Wang X, Zhang Z, Zhao Y, Xia K, Guo Y, Qu Z, Bai R (2018) A mild and facile synthesis of amino functionalized CoFe2O4@ SiO2 for Hg (II) removal. Nanomaterials 8(9):673
Bao S, Li K, Ning P, Peng J, Jin X, Tang L (2017) Highly effective removal of mercury and lead ions from wastewater by mercaptoamine-functionalised silica-coated magnetic nano-adsorbents: behaviours and mechanisms. Appl Surf Sci 393:457–466
Zhang S, Qian L, Zhou Y, Guo Y (2023) High selective removal towards Hg (II) from aqueous solution with magnetic diatomite-based adsorbent functionalized by poly (3-aminothiophenol): conditional optimization, application, and mechanism. Environ Sci Pollut Res 30(19):56121–56136
Lin Z, Pan Z, Zhao Y, Qian L, Shen J, Xia K, Guo Y, Qu Z (2020) Removal of Hg2+ with polypyrrole-functionalized Fe3O4/kaolin: synthesis, performance and optimization with response surface methodology. Nanomaterials 10(7):1370
S. Sireesha, I. Sreedhar 2023 Holistic and parametric optimization study on Cr (VI) removal using acid-treated coco peat biochar adsorbent, Bioresource Technology Reports 101486
Varghese SM, Chowdhury AR, Arnepalli DN, Rao GR (2023) Delineating the effects of pore structure and N-doping on CO2 adsorption using coco peat derived carbon. Carbon Trends 10:100250
Peer FE, Bahramifar N, Younesi H (2018) Removal of Cd (II), Pb (II) and Cu (II) ions from aqueous solution by polyamidoamine dendrimer grafted magnetic graphene oxide nanosheets. J Taiwan Inst Chem Eng 87:225–240
Venkateswarlu S, Yoon M, Kim MJ (2022) An environmentally benign synthesis of Fe3O4 nanoparticles to Fe3O4 nanoclusters: rapid separation and removal of Hg (II) from an aqueous medium. Chemosphere 286:131673
Yang L, Wang Z, Yang L, Li X, Zhang Y, Lu C (2017) Coco peat powder as a source of magnetic sorbent for selective oil–water separation. Ind Crops Prod 101:1–10
Okoli CP, Naidoo EB, Ofomaja AE (2018) Role of synthesis process variables on magnetic functionality, thermal stability, and tetracycline adsorption by magnetic starch nanocomposite. Environ Nanotechnol Monit Manag 9:141–153
Bunaciu AA, UdriŞTioiu EG, Aboul-Enein HY (2015) X-ray diffraction: instrumentation and applications. Crit Rev Anal Chem 45(4):289–299
Verma S, Kujur S, Sharma R, Pathak DD (2022) Cucurbit [6] uril-supported Fe3O4 magnetic nanoparticles catalyzed green and sustainable synthesis of 2-substituted benzimidazoles via acceptorless dehydrogenative coupling. ACS Omega 7(11):9754–9764
Tan YH, Davis JA, Fujikawa K, Ganesh NV, Demchenko AV, Stine KJ (2012) Surface area and pore size characteristics of nanoporous gold subjected to thermal, mechanical, or surface modification studied using gas adsorption isotherms, cyclic voltammetry, thermogravimetric analysis, and scanning electron microscopy. J Mater Chem 22(14):6733–6745
Ncibi M, Jeanne-Rose V, Mahjoub B, Jean-Marius C, Lambert J, Ehrhardt J, Bercion Y, Seffen M, Gaspard S (2009) Preparation and characterisation of raw chars and physically activated carbons derived from marine Posidonia oceanica (L.) fibres. J Hazard Mater 165(1–3):240–249
Fu H, Yan D, Yao C, Su X, Wang X, Wang H, Li Y (2022) Pore structure and multi-scale fractal characteristics of adsorbed pores in marine shale: a case study of the Lower Silurian Longmaxi shale in the Sichuan Basin China. J Earth Sci 33(5):1278–1290
Kianfar E (2018) Synthesis and characterization of AlPO4/ZSM-5 catalyst for methanol conversion to dimethyl ether. Russ J Appl Chem 91(10):1711–1720
Mangi HN, Chi R, DeTian Y, Sindhu L, He D, Ashraf U, Fu H, Zixuan L, Zhou W, Anees A (2022) The ungrind and grinded effects on the pore geometry and adsorption mechanism of the coal particles. J Nat Gas Sci Eng 100:104463
Ebadi M, Rifqi Md Zain A, Tengku Abdul Aziz TH, Mohammadi H, Tee CA, Rahimi Yusop M (2023) Formulation and characterization of Fe3O4@ PEG nanoparticles loaded sorafenib; molecular studies and evaluation of cytotoxicity in liver cancer cell lines. Polymers 15(4):971
Ma Z, Liu F, Liu N, Liu W, Tong M (2021) Facile synthesis of sulfhydryl modified covalent organic frameworks for high efficient Hg (II) removal from water. J Hazard Mater 405:124190
Einollahipeer F, Okati N (2022) High efficient Hg (II) and TNP removal by NH2 grafted magnetic graphene oxide synthesized from Typha latifolia. Environ Technol 43(25):3956–3972
Ahmad M, Wang J, Xu J, Yang Z, Zhang Q, Zhang B (2020) Novel synthetic method for magnetic sulphonated tubular trap for efficient mercury removal from wastewater. J Colloid Interface Sci 565:523–535
Ge H, Hua T, Wang J (2017) Preparation and characterization of poly (itaconic acid)-grafted crosslinked chitosan nanoadsorbent for high uptake of Hg2+ and Pb2+. Int J Biol Macromol 95:954–961
Das S, Samanta A, Kole K, Gangopadhyay G, Jana S (2020) MnO2 flowery nanocomposites for efficient and fast removal of mercury (II) from aqueous solution: a facile strategy and mechanistic interpretation. Dalton Trans 49(20):6790–6800
Zhang Z, Xia K, Pan Z, Yang C, Wang X, Zhang G, Guo Y, Bai R (2020) Removal of mercury by magnetic nanomaterial with bifunctional groups and core-shell structure: synthesis, characterization and optimization of adsorption parameters. Appl Surf Sci 500:143970
Awad FS, AbouZied KM, Abou El-Maaty WM, El-Wakil AM, El-Shall MS (2020) Effective removal of mercury (II) from aqueous solutions by chemically modified graphene oxide nanosheets. Arab J Chem 13(1):2659–2670
Khorshidi P, Shirazi RHSM, Miralinaghi M, Moniri E, Saadi S (2020) Adsorptive removal of mercury (II), copper (II), and lead (II) ions from aqueous solutions using glutathione-functionalized NiFe2O4/graphene oxide composite. Res Chem Intermed 46:3607–3627
Falahian Z, Torki F, Faghihian H (2018) Synthesis and application of polypyrrole/Fe3O4 nanosize magnetic adsorbent for efficient separation of Hg2+ from aqueous solution. Global Chall 2(1):1700078
Asasian N, Kaghazchi T, Soleimani M (2012) Elimination of mercury by adsorption onto activated carbon prepared from the biomass material. J Ind Eng Chem 18(1):283–289
Naushad M, Ahamad T, AlOthman ZA, Ala’a H (2019) Green and eco-friendly nanocomposite for the removal of toxic Hg (II) metal ion from aqueous environment: adsorption kinetics & isotherm modelling. J Mol Liq 279:1–8
Ifthikar J, Jiao X, Ngambia A, Wang T, Khan A, Jawad A, Xue Q, Liu L, Chen Z (2018) Facile one-pot synthesis of sustainable carboxymethyl chitosan–sewage sludge biochar for effective heavy metal chelation and regeneration. Biores Technol 262:22–31
Xia K, Guo Y, Shao Q, Zan Q, Bai R (2019) Removal of mercury (II) by EDTA-functionalized magnetic CoFe2O4@ SiO2 nanomaterial with core-shell structure. Nanomaterials 9(11):1532
Huang S, Ma C, Liao Y, Min C, Du P, Jiang Y (2016) Removal of mercury (II) from aqueous solutions by adsorption on poly (1-amino-5-chloroanthraquinone) nanofibrils: equilibrium, kinetics, and mechanism studies. J Nanomater 2016:7245829
Mensah MB, Lewis DJ, Boadi NO, Awudza JA (2021) Heavy metal pollution and the role of inorganic nanomaterials in environmental remediation. R Soc Open Sci 8(10):201485
Rahmanzadeh L, Ghorbani M, Jahanshahi M (2016) Effective removal of hexavalent mercury from aqueous solution by modified polymeric nanoadsorbent. J Water Environ Nanotechnol 1(1):1–8
AlOmar MK, Alsaadi MA, Jassam TM, Akib S, Hashim MA (2017) Novel deep eutectic solvent-functionalized carbon nanotubes adsorbent for mercury removal from water. J Colloid Interface Sci 497:413–421
Tabatabaiee Bafrooee AA, Ahmad Panahi H, Moniri E, Miralinaghi M, Hasani AH (2020) Removal of Hg2+ by carboxyl-terminated hyperbranched poly (amidoamine) dendrimers grafted superparamagnetic nanoparticles as an efficient adsorbent. Environ Sci Pollut Res 27:9547–9567
Zhang Y, Yan L, Xu W, Guo X, Cui L, Gao L, Wei Q, Du B (2014) Adsorption of Pb (II) and Hg (II) from aqueous solution using magnetic CoFe2O4-reduced graphene oxide. J Mol Liq 191:177–182
Cui L, Guo X, Wei Q, Wang Y, Gao L, Yan L, Yan T, Du B (2015) Removal of mercury and methylene blue from aqueous solution by xanthate functionalized magnetic graphene oxide: sorption kinetic and uptake mechanism. J Colloid Interface Sci 439:112–120
Goci MC, Leudjo Taka A, Martin L, Klink MJ (2023) Chitosan-based polymer nanocomposites for environmental remediation of mercury pollution. Polymers 15(3):482
Liu F, Liu Y, Xu Y, Ni L, Meng X, Hu Z, Zhong G, Meng M, Wang Y, Han J (2015) Efficient static and dynamic removal of Sr (II) from aqueous solution using chitosan ion-imprinted polymer functionalized with dithiocarbamate. J Environ Chem Eng 3(2):1061–1071
Zhao R, Jia L, Yao Y-X, Huo R-P, Qiao X-L, Fan B-G (2019) Study of the effect of adsorption temperature on elemental mercury removal performance of iron-based modified biochar. Energy Fuels 33(11):11408–11419
Johari K, Saman N, Song ST, Cheu SC, Kong H, Mat H (2016) Development of coconut pith chars towards high elemental mercury adsorption performance–effect of pyrolysis temperatures. Chemosphere 156:56–68
Fuente-Cuesta A, Diaz-Somoano M, Lopez-Anton M, Cieplik M, Fierro J, Martínez-Tarazona M (2012) Biomass gasification chars for mercury capture from a simulated flue gas of coal combustion. J Environ Manage 98:23–28
Safari N, Ghanemi K, Buazar F (2020) Selenium functionalized magnetic nanocomposite as an effective mercury (II) ion scavenger from environmental water and industrial wastewater samples. J Environ Manage 276:111263
Bhatnagar P, Sireesha S, Siddiqui S, Sreedhar I (2023) Novel pectin-cellulose-biochar composite with SDS modification for copper removal: optimization, characterization, and regeneration. Bioresour Technol Rep 21:101382
Samaniego JO, Tanchuling MAN (2019) Removal of heavy metals from an actual small scale gold mining wastewater by sorption onto Cocopeat. ASEAN J Sci Technol Dev 36(1):1–7
Hosseini-Bandegharaei A, Hosseini MS, Jalalabadi Y, Sarwghadi M, Nedaie M, Taherian A, Ghaznavi A, Eftekhari A (2011) Removal of Hg (II) from aqueous solutions using a novel impregnated resin containing 1-(2-thiazolylazo)-2-naphthol (TAN). Chem Eng J 168(3):1163–1173
Jena KK, Reddy KSK, Karanikolos GN, Choi DS (2023) l-Cysteine and silver nitrate based metal sulfide and Zeolite-Y nano adsorbent for efficient removal of mercury (II) ion from wastewater. Appl Surf Sci 611:155777
Arshadi M, Mousavinia F, Khalafi-Nezhad A, Firouzabadi H, Abbaspourrad A (2017) Adsorption of mercury ions from wastewater by a hyperbranched and multi-functionalized dendrimer modified mixed-oxides nanoparticles. J Colloid Interface Sci 505:293–306
Chen F, Ma N, Peng G, Xu W, Zhang Y, Meng F, Huang Q, Hu B, Wang Q, Guo X (2023) Camellia oleifera shell biochar as a robust adsorbent for aqueous mercury removal. Fermentation 9(3):295
Esrafili A, Ghambarian M, Tajik M, Baharfar M (2020) Adsorptive removal of Hg2+ from environmental water samples using thioglycerol-intercalated magnetic layered double hydroxides. Anal Methods 12(17):2279–2286
Hadi P, To M-H, Hui C-W, Lin CSK, McKay G (2015) Aqueous mercury adsorption by activated carbons. Water Res 73:37–55
Sun X, Hwang J-Y, Xie S (2011) Density functional study of elemental mercury adsorption on surfactants. Fuel 90(3):1061–1068
Duan L, Hu X, Sun D, Liu Y, Guo Q, Zhang T, Zhang B (2020) Rapid removal of low concentrations of mercury from wastewater using coal gasification slag. Korean J Chem Eng 37:1166–1173
Funding
This work was supported by Gorgan University of Agricultural Sciences and Natural Resources, Iran [grant number 01–474-96].
Author information
Authors and Affiliations
Contributions
Hassan Rezaei: conceptualization, investigation, methodology, and supervision; Negar Movazzaf Rostami: investigation, methodology, original draft; Hajar Abyar: data analysis, original draft, and draft review and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rezaei, H., Rostami, N.M. & Abyar, H. Magnetic nanocomposite synthesized from cocopeat for highly efficient mercury removal from aqueous solutions. Biomass Conv. Bioref. (2024). https://doi.org/10.1007/s13399-024-05425-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-024-05425-4