Abstract
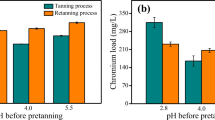
Tanned leather is a product of high intrinsic value and a sign of luxury. Chromium-tanned leather shavings are abundantly produced wastes which contain chromium that could be revalorized. The present research proposes the use of alkaline hydrolysis, aided by an inorganic anion, to promote the hydrolysis of the Cr–O bond. De-chromed solids were employed to synthesize carbon material, which subsequently was applied for dye removal from aqueous solution, while the chromium-rich solution was used as a re-tanning agent. The results demonstrated that it is possible to develop a method of chromium-protein separation, with a minimum degree of hydrolyzation. When an activating agent was employed, the prepared carbon had a surface area of 585 m2/g, with trace levels of chromium (< 0.1%), present mainly as Cr2O3 particles. The materials displayed an adsorption capacity of up to 47.60 mg/g and 27.58 mg/g for methylene blue and Congo red respectively. The chromium-rich solution contained proteins and amino acids that aid in its employment as a re-tanning agent. Leather that was re-tanned with this solution had a similar tear strength and a superior and tensile strength (38%), superior elongation at break (10%), and improved elongation at break (47%) than traditional Cr33-tanned leather. The present study represents a major step in the development of cleaner production of leather.
Graphical abstract






Similar content being viewed by others
Data availability
Data will be available upon request.
References
Pati A, Chaudhary R, Subramani S (2014) A review on management of chrome-tanned leather shavings: a holistic paradigm to combat the environmental issues. Environ Sci Pollut R 21:11266–11282. https://doi.org/10.1007/s11356-014-3055-9
Kolomaznik K, Adamek M, Andel I, Uhlirova M (2008) Leather waste—potential threat to human health, and a new technology of its treatment. J Hazard Mater 160:514–520. https://doi.org/10.1016/j.jhazmat.2008.03.070
Muralidharan V, Palanivel S, Balaraman M (2022) Turning problem into possibility: a comprehensive review on leather solid waste intra-valorization attempts for leather processing. J Clean Prod 133021. https://doi.org/10.1016/j.jclepro.2022.133021
Appala VNSG, Pandhare NN, Bajpai S (2022) Biorefining of leather solid waste to harness energy and materials—a review. Biomass Convers Biorefin. 1–18. https://doi.org/10.1007/s13399-022-02455-8
Long H, Huang X, Liao Y, Ding J (2021) Recovery of Cr(VI) from tannery sludge and chrome-tanned leather shavings by Na2CO3 segmented calcination. J Environ Chem Eng 9:105026. https://doi.org/10.1016/j.jece.2021.105026
Rigueto CVT, Nazari MT, Rosseto M, Massuda LA, Alessandretti IP, Piccin JS, Dettmer A (2021) Emerging contaminants adsorption by beads from chromium (III) tanned leather waste recovered gelatin. J Mol Liq 330:115638. https://doi.org/10.1016/j.molliq.2021.115638
Li C, Xue F, Ding E (2015) Preparation of polyacrylamide grafted collagen extracted from leather wastes and their application in kaolin flocculation. J Appl Polym Sci 132. https://doi.org/10.1002/app.41556
Gao D, Guo S, Zhou Y, Lyu B, Ma J, Zhao P, Pan D, Chen S (2022) Hydrophobic, flexible electromagnetic interference shielding films derived from hydrolysate of waste leather scraps. J Colloid Interface Sci 613:396–405. https://doi.org/10.1016/j.jcis.2022.01.043
Tang Y, Zhao J, Zhang Y, Zhou J, Shi B (2021) Conversion of tannery solid waste to an adsorbent for high-efficiency dye removal from tannery wastewater: a road to circular utilization. Chemosphere 263:127987. https://doi.org/10.1016/j.chemosphere.2020.127987
Manera C, Tonello AP, Perondi D, Godinho M (2019) Adsorption of leather dyes on activated carbon from leather shaving wastes: kinetics, equilibrium and thermodynamics studies. Environ Technol 40:2756–2768. https://doi.org/10.1080/09593330.2018.1452984
Rao JR, Thanikaivelan P, Sreeram KJ, Nair BU (2002) Green route for the utilization of chrome shavings (chromium-containing solid waste) in tanning industry. Environ Sci Technol 36:1372–1376. https://doi.org/10.1021/es015635s
Senthil R, Hemalatha T, Kumar BS, Uma TS, Das BN, Sastry TP (2015) Recycling of finished leather wastes: a novel approach. Clean Technol Environ Policy 17:187–197. https://doi.org/10.1007/s10098-014-0776-x
Malek A, Hachemi M, Didier V (2009) New approach of depollution of solid chromium leather waste by the use of organic chelates: economical and environmental impacts. J Hazard Mater 170:156–162. https://doi.org/10.1016/j.jhazmat.2009.04.118
Ferreira MJ, Almeida MF, Pinho SC, Gomes JR, Rodrigues JL (2014) Alkaline hydrolysis of chromium tanned leather scrap fibers and anaerobic biodegradation of the products. Waste Biomass Valori 5:551–562. https://doi.org/10.1007/s12649-013-9252-9
Wionczyk B, Apostoluk W, Charewicz WA, Adamski Z (2011) Recovery of chromium(III) from wastes of uncolored chromium leathers. Part II. Solvent extraction of chromium(III) from alkaline protein hydrolyzate. Sep Purif Technol 81:237–242. https://doi.org/10.1016/j.seppur.2011.07.024
Beltrán-Prieto JC, Veloz-Rodríguez R, Pérez-Pérez MC, Navarrete-Bolaños JL, Vázquez-Nava E, Jiménez-Islas H, Botello-Álvarez JE (2012) Chromium recovery from solid leather waste by chemical treatment and optimisation by response surface methodology. Chem Ecol 28:89–102. https://doi.org/10.1080/02757540.2011.628016
Malek A, Hachemi M (2008) Effect of the detoxification on the shrinkage temperature and pH of chromium leather waste, another promising way for the tannery pollution. Am J Appl Sci 5:1329–1335. https://doi.org/10.3844/ajassp.2008.1329.1335
Yahya MA, Al-Qodah Z, ZanariahNgah CW (2015) Agricultural bio-waste materials as potential sustainable precursors used for activated carbon production: a review. Renew Sust Energ Rev 46:218–235. https://doi.org/10.1016/j.rser.2015.02.051
Arcibar-Orozco JA, Barajas-Elias BS, Caballero-Briones F, Nielsen L, Rangel-Mendez JR (2019) Hybrid carbon nanochromium composites prepared from chrome-tanned leather shavings for dye adsorption. Water, Air, Soil Pollut 230:142. https://doi.org/10.1007/s11270-019-4194-x
Arcibar-Orozco JA, Rangel-Mendez JR, Bandosz TJ (2013) Desulfurization of model diesel fuel on activated carbon modified with iron oxyhydroxide nanoparticles: effect of tert-butylbenzene and naphthalene concentrations. Energy Fuels 27:5380–5387. https://doi.org/10.1021/ef400999g
Jagiello J (1994) Stable numerical solution of the adsorption integral equation using splines. Langmuir 10:2778–2785. https://doi.org/10.1021/la00020a045
Hammond JBW, Kruger NJ (1988) The Bradford method for protein quantitation. In: Walker JM (ed) New protein techniques. Humana Press, Totowa, NJ, pp 25–32
ISO, 2017. ISO 20137:2017(en) Leather — chemical tests — guidelines for testing critical chemicals in leather. https://www.iso.org/obp/ui/#iso:std:iso:20137:ed-1:v1:en. (Accessed 09/09/2022)
Wionczyk B, Apostoluk W, Charewicz WA, Adamski Z (2011) Recovery of chromium(III) from wastes of uncolored chromium leathers. Part I. Kinetic studies on alkaline hydrolytic decomposition of the wastes. Sep Purif Technol 81:223–236. https://doi.org/10.1016/j.seppur.2011.07.036
Ke L, Zhao K, Yan X, Cao X, Wu X, Zhang C, Luo T, Ding T, Yan N (2022) Facile mineralization and valorization of Cr-containing leather shavings for electrocatalytic H2O2 generation and organic pollutant removal. Chem Eng J 437:135036. https://doi.org/10.1016/j.cej.2022.135036
Appala VNS, Gunasri P, Naresh N, Bajpai S (2022) Biorefining of leather solid waste to harness energy and materials—a review. Biorefin. In press, Biomass Convers. https://doi.org/10.1007/s13399-022-02455-8
Gauza-Włodarczyk M, Kubisz L, Mielcarek S, Włodarczyk D (2017) Comparison of thermal properties of fish collagen and bovine collagen in the temperature range 298–670K. Mater Sci Eng C 80:468–471. https://doi.org/10.1016/j.msec.2017.06.012
Yılmaz O, CemKantarli I, Yuksel M, Saglam M, Yanik J (2007) Conversion of leather wastes to useful products. Resour Conserv Recycl 49:436–448. https://doi.org/10.1016/j.resconrec.2006.05.006
Oliveira LCA, Guerreiro MC, Gonçalves M, Oliveira DQL, Costa LCM (2008) Preparation of activated carbon from leather waste: a new material containing small particle of chromium oxide. Mater Lett 62:3710–3712. https://doi.org/10.1016/j.matlet.2008.04.064
Kong J, Yue Q, Huang L, Gao Y, Sun Y, Gao B, Li Q, Wang Y (2013) Preparation, characterization and evaluation of adsorptive properties of leather waste based activated carbon via physical and chemical activation. Chem Eng J 221:62–71. https://doi.org/10.1016/j.cej.2013.02.021
Chaudhary R, Pati A (2017) Adsorption isotherm and kinetics of tannic acid on to carbonized chrome tanned leather solid waste. J Soc Leath Tech Ch 112:198–206
Karmegam PM, Natarajan P, Somasundaram S (2023) Effect of activating agents on the photocatalytic activity of chromium oxide based porous carbon photocatalysts derived from chrome-tanned leather buffing dust waste for the degradation of 2-chlorophenol. Chem Eng J 451:138553. https://doi.org/10.1016/j.cej.2022.138553
Li K, Liu Q, Cheng H, Hu M, Zhang S (2021) Classification and carbon structural transformation from anthracite to natural coaly graphite by XRD, Raman spectroscopy, and HRTEM. Spectrochm Acta A 249:119286. https://doi.org/10.1016/j.saa.2020.119286
Manoj B, Kunjomana AG (2012) Study of stacking structure of amorphous carbon by X-ray diffraction technique. Int J Electrochem Sci 7:3127–3134
Aguilera C, González JC, Borrás A, Margineda D, González JM, González-Elipe AR, Espinós JP (2013) Preparation and characterization of CrO2 films by Low Pressure Chemical Vapor Deposition from CrO3. Thin Solid Films 539:1–11. https://doi.org/10.1016/j.tsf.2013.04.118
Fuente E, Menéndez JA, Suárez D, Montes-Morán MA (2003) Basic surface oxides on carbon materials: a global view. Langmuir 19:3505–3511. https://doi.org/10.1021/la026778a
Laboratory, Measurement Services Division of the National Institute of Standards and Technology Material Measurement (2012) NIST, XPS database. https://srdata.nist.gov/xps. (Accessed 23/06/2022)
Chen Y-d, Lin Y-C, Ho S-H, Zhou Y, Ren N-q (2018) Highly efficient adsorption of dyes by biochar derived from pigments-extracted macroalgae pyrolyzed at different temperature. Bioresour Technol 259:104–110. https://doi.org/10.1016/j.biortech.2018.02.094
Raposo F, De La Rubia MA, Borja R (2009) Methylene blue number as useful indicator to evaluate the adsorptive capacity of granular activated carbon in batch mode: influence of adsorbate/adsorbent mass ratio and particle size. J Hazard Mater 165:291–299. https://doi.org/10.1016/j.jhazmat.2008.09.106
Rafatullah M, Sulaiman O, Hashim R, Ahmad A (2010) Adsorption of methylene blue on low-cost adsorbents: a review. J Hazard Mater 177:70–80. https://doi.org/10.1016/j.jhazmat.2009.12.047
Raval NP, Shah PU, Shah NK (2016) Adsorptive amputation of hazardous azo dye Congo red from wastewater: a critical review. Environ Sci Pollut R 23:14810–14853. https://doi.org/10.1007/s11356-016-6970-0
Tahiri S, Messaoudi A, Albizane A, Azzi M, Bouhria M, Younssi SA, Bennazha J, Mabrour J (2003) Removal of dyes from aqueous solutions by adsorption on chrome-tanned solid wastes generated in the leather industry. Water Qual Res J 38:393–411. https://doi.org/10.2166/wqrj.2003.025
Zubair M, Mùazu ND, Jarrah N, Blaisi NI, Aziz HA, Al-Harthi MA (2020) Adsorption behavior and mechanism of methylene blue, crystal violet, eriochrome black T, and methyl orange dyes onto biochar-derived date palm fronds waste produced at different pyrolysis conditions. Water, Air, Soil Pollut 231:240. https://doi.org/10.1007/s11270-020-04595-x
Fan S, Wang Yi, Wang Z, Tang J, Tang J, Li X (2017) Removal of methylene blue from aqueous solution by sewage sludge-derived biochar: adsorption kinetics, equilibrium, thermodynamics and mechanism. J Environ Chem Eng 5:601–611. https://doi.org/10.1016/j.jece.2016.12.019
Lonappan L, Rouissi T, Das RK, Brar SK, Ramirez AA, Verma M, Surampalli RY, Valero JR (2016) Adsorption of methylene blue on biochar microparticles derived from different waste materials. Waste Manage (Oxford) 49:537–544. https://doi.org/10.1016/j.wasman.2016.01.015
Gürses A, Doğar Ç, Karaca S, Acikyildiz M, Bayrak R (2006) Production of granular activated carbon from waste Rosa canina sp. seeds and its adsorption characteristics for dye. J Hazard Mater 131:254–259. https://doi.org/10.1016/j.jhazmat.2005.09.014
Tseng R-L, Tseng S-K, Wu F-C (2006) Preparation of high surface area carbons from Corncob with KOH etching plus CO2 gasification for the adsorption of dyes and phenols from water. Colloids Surf A 279:69–78. https://doi.org/10.1016/j.colsurfa.2005.12.042
Li Z, Hanafy H, Zhang L, Sellaoui L, Schadeck N, Matias O, Marcos LS, Seliem MK, Luiz Dotto G, Bonilla-Petriciolet A, Li Q (2020) Adsorption of congo red and methylene blue dyes on an ashitaba waste and a walnut shell-based activated carbon from aqueous solutions: experiments, characterization and physical interpretations. Chem Eng J 388:124263. https://doi.org/10.1016/j.cej.2020.124263
Harja M, Buema G, Bucur D (2022) Recent advances in removal of Congo Red dye by adsorption using an industrial waste. Sci Rep 12:6087. https://doi.org/10.1038/s41598-022-10093-3
Singh P, Raizada P, Pathania D, Sharma G, Sharma P (2013) Microwave induced KOH activation of guava peel carbon as an adsorbent for congo red dye removal from aqueous phase. Indian J Chem Technol 20:305–311
Sivakumar V, Asaithambi M, Sivakumar P, Gopal N (2014) Removal of Congo red dye using an adsorbent prepared from Martynia annua. L seeds Am Chem Sci J 4:424–442. https://doi.org/10.9734/ACSJ/2014/6680
Ghaedi M, Tavallali H, Sharifi M, Kokhdan SN, Asghari A (2012) Preparation of low cost activated carbon from Myrtus communis and pomegranate and their efficient application for removal of Congo red from aqueous solution. Spectrochm Acta A 86:107–114. https://doi.org/10.1016/j.saa.2011.10.012
Extross A, Waknis A, Tagad C, Gedam VV, Pathak PD (2022) Adsorption of congo red using carbon from leaves and stem of water hyacinth: equilibrium, kinetics, thermodynamic studies. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-022-03938-x
Kanagaraj J, Chandra Babu NK, Mandal AB (2008) Recovery and reuse of chromium from chrome tanning waste water aiming towards zero discharge of pollution. J Clean Prod 16:1807–1813. https://doi.org/10.1016/j.jclepro.2007.12.005
Pati A, Chaudhary R, Subramani S (2013) Biochemical method for extraction and reuse of protein and chromium from chrome leather shavings: a waste to wealth approach. J Soc Leath Tech Ch 108:365–372
Kim W (2021) A study on the subjective feeling affecting tactile satisfaction of leather in automobile: a structural equation modeling approach. Int J Ind Ergon 84:103167. https://doi.org/10.1016/j.ergon.2021.103167
Dayanandan A, Kanagaraj J, Sounderraj L, Govindaraju R, Rajkumar GS (2003) Application of an alkaline protease in leather processing: an ecofriendly approach. J Clean Prod 11:533–536. https://doi.org/10.1016/S0959-6526(02)00056-2
Habib AB, Noor IA, Musa AE (2015) Effect of some skin defects on physical properties of the leather. J Appl Ind Sci 3:112–119
Ali F, Kamal M, Shoriful IM (2020) Comparative study on physical properties of different types of leather in Bangladesh. Int J Eng Res Appl 10:55–63. https://doi.org/10.9790/9622-1002035563
Acknowledgements
The authors are grateful with Ernesto Ornelas and Catalina de la Rosa for their technical assistance at CIATEC. The assistance of national laboratories LINAN and LANBAMA as well as the help of Ana Iris Maldonado during the microscopy measurements and Elizabeth Isaacs for the physisorption of nitrogen is appreciated. XPS analysis was performed by Engineer Wilian Javier Cauich at the National Laboratory of Nano and Biomaterials at CINVESTAV-Merida, sponsored by the following projects: FOMIX-Yucatan, 2008-1081160 and CONACYT LA-2009-01-123913, 292692, 294643, 188345, and 204822. Ionic chromatography analysis was sponsored by CONACYT through the project 269990.
Funding
This work was supported by the Ministry for Innovation of Guanajuato (Secretaría de Innovación del Estado de Guanajuato, SICES) through the SICES/058/2018 grant and CONACYT (Ciencia de Frontera CF-2019 1727999).
Author information
Authors and Affiliations
Contributions
JA Arcibar-Orozco was in charge of the experimentation and planning of the manuscript, wrote the majority, and thoroughly reviewed it, A. Saldaña-Robles performed the design experimental analysis and wrote the corresponding discussion, R. Rangel-Mendez was in charge of most of the material’s characterization, Lilja Nielsen wrote part of the manuscript and edited thoroughly, H. Baltazar-Campos performed all of the extraction and adsorption experiments, A. Garduño-Cruces performed the chromatography analysis and characterization of the protein, V. Hernandez-López assisted in the experimentation related to the leather re-tanning, and F. Caballero-Briones performed the XPS analysis and discussion.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arcibar-Orozco, J.A., Saldaña-Robles, A., Rangel-Méndez, R. et al. Revalorization of chromium-tanned leather shavings into carbon materials and re-tanning solution. Biomass Conv. Bioref. (2023). https://doi.org/10.1007/s13399-023-04014-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-023-04014-1