Abstract
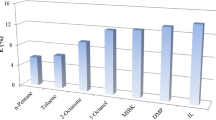
Due to high demands for the production of propionic from fermentation broth, the reactive extraction of the acid using green and non-toxic and microorganism-friendly diluent instead of toxic and harmful conventional organic solvents is considered in this work over the temperature range of 298–318 K. Soybean oil and rice bran oils which reduce the toxic effect of the used reactive extractant tri-octyl amine (TOA) are used as the diluents. The achieved overall distribution coefficient and extraction efficiency are found higher in soybean oil (0.73, 42.25%, respectively) as compared to rice bran oil (0.66, 40.00%, respectively). Due to the low cost and microorganism-friendly nature, soybean and rice bran oils are anticipated to use in bio-refinery industries to recover propionic acid from fermentation broth. The estimated \({K}_{D}^{chem}/{K}_{D}^{phy}\) ratio greater than 1.0 shows that the extraction of the acid is dominated by the chemical extraction at the relatively higher initial concentration of TOA. The chemical reaction equilibrium constant, \({K}_{E(m:n)}\), and complexation stoichiometries \(m\) and \(n\) are determined by applying differential evaluation (DE) technique. \({K}_{E\left(m:n\right)}\) is found higher in the case of soybean oil. Despite the loading ratio Z < 0.5, \(m\) is found greater than 1.0. The concentration of the complex \({[{\left(HA\right)}_{m}{S}_{n}]}_{org}\) is higher in soybean oil than rice bran oil. The negative enthalpy change due to the complexation reaction shows that it is exothermic. The positive entropy changes due to only mass transfer direct to supply external energy to mix the studied immiscible phases.






Similar content being viewed by others
Abbreviations
- \([S]\) :
-
Concentration of extractant (mol/Kg)
- \([HA]\) :
-
concentration of acid (mol/Kg)
- \(P\) :
-
Partition coefficient
- \(D\) :
-
Dimerization constant
- \({K}_{D}\) :
-
Distribution coefficient
- \(\%E\) :
-
Extraction efficiency
- \({K}_{E}\) :
-
Overall equilibrium complexation constant
- \({K}_{HA}\) :
-
Dissociation constant of propionic acid
- \(m\) :
-
Number of acid molecules
- \(n\) :
-
Number of extractant molecules
- \(Z\) :
-
Loading ratio
- μ :
-
Acid extractant by diluents alone
- ν :
-
Diluent fraction
- \(aq\) :
-
Aqueous solution
- \(org\) :
-
Organic solution
- \(exp\) :
-
Experimental value
- \(\Delta H\) :
-
Enthalpy (KJ/mol)
- \(\Delta S\) :
-
Entropy (J/mol.K)
- \(0\) :
-
Initial concentration of butyric acid
- \(tot\) :
-
Total concentration
- \(free\) :
-
Free acid concentration
- \(dil\) :
-
Diluent
- \(che\) :
-
Chemical
- \(overall\) :
-
Overall acid concentration or distribution coefficient
- \(mod\) :
-
Model value
- \(rxn\) :
-
Reaction
- \(mass tr\) :
-
Mass transfer
References
Ayan E, Baylan N, Cehreli N (2020) Optimization of reactive extraction of propionic acid with ionic liquids using central composite design. Chem Eng Res Des 153:666–676
Feng X-H, Chen F, Xu H, Wu B, Yao J, Ying H-J, Ouyang P-K (2010) Propionic acid fermentation by Propionibacterium freudenreichii CCTCC M207015 in a multi-point fibrous-bed bioreactor. Bioprocess Biosyst Eng 33:1077–1085
Lopes HJS, Ramos LR, Silva EL (2017) Co-fermentation of cheese whey and crude glycerol in EGSB reactor as a strategy to enhance continuous hydrogen and propionic acid production. Appl Technol Biotechnol 183:712–728
Coral J, Karp SG, de Souza LP, José Parada JL, Pandey A, Soccol CR (2008) “Batch fermentation model of propionic acid production by Propionibacterium acidipropionici in different carbon sources.” Appl Biochem Biotechnol 151:333–341
Kagliwal LD, Survase SA, Singhal RS (2013) Granström, T “Wheat flour based propionic acid fermentation: an economic approach” Biores. Technol 129:694–699
Chanakya HN, Khuntia HK, Mukherjee N, Aniruddha R, Mudakavi JR (2015) Thimmaraju, P “The physicochemical characteristics and anaerobic degradability of desiccated coconut industry waste water” Env. Monit Assess 187(772):1–12
Roy, H; Simon, C; James, K “Bacterial additives for biological and/or chemical contaminants within water-based fluids”, US 2015/0315052 A1, 2015.
Kurniawan A, Kwon SY, Shin J, Hur J (2016) Cho, J “Acid fermentation process combined with post denitrification for the treatment of primary sludge and wastewater with high strength nitrate.” Water 8:1–11
Skalsky DS, Daigger GT (1995) “Wastewater solids fermentation for volatile acid production and enhanced biological phosphorus removal”, Water Env. Res 230 – 237.
Woskow SA, Glatz BA (1991) “Propionic acid production by a propionic acid-tolerant strain of Propionibacterium acidipropionici in batch and semicontinuous fermentation”, Appl Env Microb. 2821 – 2828.
Lewis VP, Yang S (1992) A “Novel extractive fermentation process for propionic acid production from whey lactose.” Biotechnol Prog 8:104–110
Rodriguez BA, Stowers CC, Phama V, Coxb BM (2014) “The production of propionic acid, propanol and propylene via sugar fermentation: an industrial perspective on the progress, technical challenges and future outlook”, Green Chem 1066 – 1076.
Castilla-Archilla J, Heiberger J, Mills S, Hilbig J, Collins G, Lens PN (2021) L “Continuous volatile fatty acid production from acid brewery spent grain leachate in expanded granular sludge bed reactors” Front. Sus Food Sys 664944:1–16
Stowers CC, Cox BM, Rodriguez B (2014) A “Development of an industrializable fermentation process for propionic acid production.” J Ind Microb Biotechnol 41:837–852
Wang X, Salvachúa D, Nogué VS, Michener WE, Bratis AD, Dorgan JR, Beckham GT (2017) “Propionic acid production from corn stover hydrolysate by Propionibacterium Acidipropionici” Biotechnol Biofuel 1–13
Hong YK, Hong WH, Han DH (2001) Application of reactive extraction to recovery of carboxylic acids. Biotechnol Bioprocess Eng 6:386–394
Keshav A, Wasewar KL, Chand S (2009) Extraction of acrylic, propionic, and butyric acid using Aliquat 336 in oleyl alcohol: equilibria and effect of temperature. Ind Eng Chem Res 48:888–893
Keshav A, Chand S, Wasewar KL (2009) Recovery of propionic acid from aqueous phase by reactive extraction using Quaternary amine (Aliquat 336) in various diluents. Chem Eng J 152:95–102
Keshav A, Wasewar KL, Chand S (2008) Extraction of propionic acid using different extractants (tri-n-butylphosphate, tri-n-octylamine, and Aliquat 336). Ind Eng Chem Res 47:6192–6196
Keshav A, Chand S, Wasewar KL (2008) Equilibrium studies for extraction of propionic acid using tri-n-butyl phosphate in different solvents. J Chem Eng Data 53:1424–1430
Harris RD, Geankoplis CJ (1962) Extraction of mixed solutes separation of propionic and sulfuric acids. J Chem Eng Data 7(2):218–223
Wasewar KL, Keshav A, Seema (2010) “Physical extraction of propionic acid”, Int. J Recent Research and Appl. Stud, 3(3):290–302.
Henczka M, Djas M (2016) Reactive extraction of acetic acid and propionic acid using supercritical carbon dioxide. J Supercritical Fluids 110:154–160
Inyang VM, Lokhat D (2020) Reactive extraction of propionic acid using trioctylamine in 1–decanol by response surface methodology optimization technique. Int J Low-Carbon Technol 15:171–179
Kumar S, Babu BV, Wasewar KL (2012) Investigations of biocompatible systems for reactive extraction of propionic acid using aminic extractants (TOA and Aliquat 336). Biotechnol Bioprocess Eng 17:1252–1260
Fahim MA, Qader A, Hughes MA (1992) Extraction equilibria of acetic and propionic acids from dilute aqueous solution by several solvents. Sep Sci Technol 27(13):1809–1821
Zhong G, Bonita AG, Charles EG (1998) Propionic acid production by extractive fermentation I Solvent considerations. Biotechnol BioEng 57:454
Keshav A, Wasewar KL, Chand S (2009) Reactive extraction of propionic acid using tri-n-octylamine, tri-n-butyl phosphate and Aliquat 336 in sunflower oil as diluent. J Chem Technol Biotechnol 84:484–489
Mukherjee S, Nayak SS, Munshi B “Reactive extraction of propionic acid using non-toxic diluents” Chem. Eng. Technol (Manuscript id: ceat.202100511)
Mukherjee S, Munshi B (2022) “Reactive extraction of butyric acid from effluent streams and fermentation broth by using tri-n-octyl amine in decanol/natural oils” Biomass Convers Biorefin. https://doi.org/10.1007/s13399-022-02504-2
Storn, R.; “Differential evolution- a simple and efficient heuristic for global optimization over continuous spaces” Journal of Global Optimization 1997, 341–359
Mukherjee S, Munshi B (2020) Experimental and theoretical analysis of reactive extraction of caproic acid by using TBP in green diluents. Chem Eng Process 153:107926
Mukherjee S, Negi D, Nagraj M, Munshi B (2021) Reactive extraction of butyric acid from water using trioctyl amine in 1-decanol and green natural oils. J Chem Eng Data 66:2733–2753
Thakre N, Prajapati AK, Mahapatra SP, Kumar A, Khapre A (2016) Modeling and optimization of reactive extraction of citric acid. J Chem Eng Data. https://doi.org/10.1021/acs.jced.6b00274
Acknowledgements
We acknowledge the NIT Rourkela for infrastructural and analytical facilities.
Author information
Authors and Affiliations
Contributions
All authors have participated in the (a) conception and design or analysis and interpretation of the data; (b) drafting the article or revising it critically for important intellectual content; and (c) approval of the final version.
Corresponding author
Ethics declarations
Consent for publication
This manuscript has not been submitted to, nor is under review at, another journal or other publishing venue.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mukherjee, S., Munshi, B. Reactive extraction of propionic acid by using tri-octyl amine in edible oils. Biomass Conv. Bioref. 14, 8029–8039 (2024). https://doi.org/10.1007/s13399-022-02883-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-02883-6