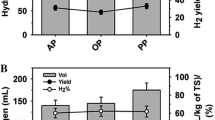
Abstract
Potato waste, which contains starch as a primary carbohydrate, is generated domestically and industrially as solid or liquid waste. Biological fermentation process is a cost-effective and clean way of H2 production. The combined dark and photo fermentation technique is an economical and time-saving process with a higher H2 production rate and yield. The current study highlights the feasibility of using potato waste as substrate for H2 production in combined dark and photo fermentation. The dark and photo fermentative bacteria Enterobacter aerogenes MTCC2822 and Rhodobacter sphaeroides O.U.001 respectively were used at a ratio of 1:7 (w/w) for H2 production. Response surface methodology (RSM) with central composite design (CCD) was employed to optimize carbohydrate concentration, FeCl3 concentration, and pH for enhancing H2 production. During fermentation, pH drop inside photo bioreactor (PBR) was observed due to accumulation of volatile fatty acids (VFAs). The maximum H2 production was 1580 mL H2/L media at 2 g/L carbohydrate concentration and 0.2 g/L FeCl3 concentration at pH 7. In total, 53% of the initial carbohydrate was converted to hydrogen with H2 production yield of 6.31 mol H2/mol carbohydrate.
Graphical abstract












Similar content being viewed by others
References
Al-Mohammedawi HH, Znad H, Eroglu E (2018) Synergistic effects and optimization of photo-fermentative hydrogen production of Rhodobacter sphaeroides DSM 158. Int J Hydrogen Energy 43:15823–15834. https://doi.org/10.1016/j.ijhydene.2018.06.140
Adessi A, McKinlay JB, Harwood CS, De Philippis R (2012) A Rhodopseudomonas palustris nifA* mutant produces H2 from NH4+-containing vegetable wastes. Int J Hydrogen Energy 37:15893–15900. https://doi.org/10.1016/j.ijhydene.2012.08.009
Mahata C, Dhar S, Ray S, Das D (2021) Effect of thermal pretreated organic wastes on the dark fermentative hydrogen production using mixed microbial consortia. Fuel 284:119062. https://doi.org/10.1016/j.fuel.2020.119062
Basak N, Das D (2007) Microbial biohydrogen production by Rhodobacter sphaeroides OU 001 in photobioreactor. Proceedings of world congress on eng. and computer sci., San Francisco, USA, Citeseer. https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.148.6134&rep=rep1&type=pdf.
Trchounian K, Sawers RG, Trchounian A (2017) Improving biohydrogen productivity by microbial dark-and photo-fermentations: novel data and future approaches. Renew Sustain Energy Rev 80:1201–1216. https://doi.org/10.1016/j.rser.2017.05.149
Meky N, Elreedy A, Ibrahim MG, Fujii M, Tawfik A (2021) Intermittent versus sequential dark-photo fermentative hydrogen production as an alternative for bioenergy recovery from protein-rich effluents. Energy 217:119326. https://doi.org/10.1016/j.energy.2020.119326
Moreira F, Rodrigues M, Sousa L, Batista F, Ferreira J, Cardoso V (2022) Single-stage repeated batch cycles using co-culture of Enterobacter cloacae and purple non-sulfur bacteria for hydrogen production. Energy 239:122465. https://doi.org/10.1016/j.energy.2021.122465
Ghosh S, Chowdhury R, Bhattacharya P (2018) A review on single stage integrated dark-photo fermentative biohydrogen production: insight into salient strategies and scopes. Int J Hydrogen Energy 43:2091–2107. https://doi.org/10.1016/j.ijhydene.2017.12.018
Rao R, Basak N (2021) Fermentative molecular biohydrogen production from cheese whey: present prospects and future strategy. Appl Biochem Biotechnol 1–34. https://doi.org/10.1007/s12010-021-03528-6
Sağır E, Yucel M, Hallenbeck PC (2018) Demonstration and optimization of sequential microaerobic dark-and photo-fermentation biohydrogen production by immobilized Rhodobacter capsulatus JP91. Biores Technol 250:43–52. https://doi.org/10.1016/j.biortech.2017.11.018
Lin R, Cheng J, Ding L, Song W, Liu M, Zhou J, Cen K (2016) Enhanced dark hydrogen fermentation by addition of ferric oxide nanoparticles using Enterobacter aerogenes. Biores Technol 207:213–219. https://doi.org/10.1016/j.biortech.2016.02.009
Rao R, Basak N (2021) Process optimization and mathematical modelling of photo-fermentative hydrogen production from dark fermentative cheese whey effluent by Rhodobacter sphaeroides OU 001 in 2-L cylindrical bioreactor. Biomass Conversion and Biorefinery 1–24. https://doi.org/10.1007/s13399-021-01377-1
Banu JR, Merrylin J, Usman TM, Kannah RY, Gunasekaran M, Kim S-H, Kumar G (2020) Impact of pretreatment on food waste for biohydrogen production: a review. Int J Hydrogen Energy 45:18211–18225. https://doi.org/10.1016/j.ijhydene.2019.09.176
Hitit ZY, Lazaro CZ, Hallenbeck PC (2017) Hydrogen production by co-cultures of Clostridium butyricum and Rhodospeudomonas palustris: optimization of yield using response surface methodology. Int J Hydrogen Energy 42:6578–6589. https://doi.org/10.1016/j.ijhydene.2016.12.122
Basak N, Jana AK, Das D (2021) Photofermentative biohydrogen generation from organic acids by Rhodobacter sphaeroides OU 001: computational fluid dynamics modeling of hydrodynamics and temperature. Biotechnol Appl Biochem. https://doi.org/10.1002/bab.2151
Adessi A, De Philippis R (2014) Photobioreactor design and illumination systems for H2 production with anoxygenic photosynthetic bacteria: a review. Int J Hydrogen Energy 39:3127–3141. https://doi.org/10.1016/j.ijhydene.2013.12.084
Akhlaghi N, Najafpour-Darzi G (2020) A comprehensive review on biological hydrogen production. Int J Hydrogen Energy. https://doi.org/10.1016/j.ijhydene.2020.06.182
Park J-H, Chandrasekhar K, Jeon B-H, Jang M, Liu Y, Kim S-H (2021) State-of-the-art technologies for continuous high-rate biohydrogen production. Biores Technol 320:124304. https://doi.org/10.1016/j.biortech.2020.124304
Zagrodnik R, Łaniecki M (2017) The effect of pH on cooperation between dark-and photo-fermentative bacteria in a co-culture process for hydrogen production from starch. Int J Hydrogen Energy 42:2878–2888. https://doi.org/10.1016/j.ijhydene.2016.12.150
Zagrodnik R, Laniecki M (2015) The role of pH control on biohydrogen production by single stage hybrid dark-and photo-fermentation. Biores Technol 194:187–195. https://doi.org/10.1016/j.biortech.2015.07.028
Rezaeitavabe F, Saadat S, Talebbeydokhti N, Sartaj M, Tabatabaei M (2020) Enhancing bio-hydrogen production from food waste in single-stage hybrid dark-photo fermentation by addition of two waste materials (exhausted resin and biochar). Biomass Bioenerg 143:105846. https://doi.org/10.1016/j.biombioe.2020.105846
Rai PK, Singh S (2016) Integrated dark-and photo-fermentation: recent advances and provisions for improvement. International Journal of Hydrogen Energy 41:19957–19971. https://doi.org/10.1016/j.ijhydene.2016.08.084
Rao R, Basak N (2021) Development of novel strategies for higher fermentative biohydrogen recovery along with novel metabolites from organic wastes: the present state of the art. Biotechnol Appl Biochem 68:421–444. https://doi.org/10.1002/bab.1964
Arizzi M, Morra S, Gilardi G, Pugliese M, Gullino M, Valetti F (2021) Improving sustainable hydrogen production from green waste:[FeFe]-hydrogenases quantitative gene expression RT-qPCR analysis in presence of autochthonous consortia. Biotechnol Biofuels 14:1–17. https://doi.org/10.1186/s13068-021-02028-3
Taherdanak M, Zilouei H, Karimi K (2015) Investigating the effects of iron and nickel nanoparticles on dark hydrogen fermentation from starch using central composite design. Int J Hydrogen Energy 40:12956–12963. https://doi.org/10.1016/j.ijhydene.2015.08.004
Basak N, Das D (2007) The prospect of purple non-sulfur (PNS) photosynthetic bacteria for hydrogen production: the present state of the art. World J Microbiol Biotechnol 23:31–42. https://doi.org/10.1007/s11274-006-9190-9
Kumar G, Mathimani T, Rene ER, Pugazhendhi A (2019) Application of nanotechnology in dark fermentation for enhanced biohydrogen production using inorganic nanoparticles. Int J Hydrogen Energy 44:13106–13113. https://doi.org/10.1016/j.ijhydene.2019.03.131
Rambabu K, Bharath G, Banat F, Hai A, Show PL, Nguyen THP (2021) Ferric oxide/date seed activated carbon nanocomposites mediated dark fermentation of date fruit wastes for enriched biohydrogen production. Int J Hydrogen Energy 46:16631–16643. https://doi.org/10.1016/j.ijhydene.2020.06.108
Alian M, Saadat S, Rezaeitavabe F (2021) An investigation on the dose-dependent effect of iron shaving on bio-hydrogen production from food waste. Int J Hydrogen Energy 46:19886–19896. https://doi.org/10.1016/j.ijhydene.2021.03.121
Adessi A, Venturi M, Candeliere F, Galli V, Granchi L, De Philippis R (2018) Bread wastes to energy: sequential lactic and photo-fermentation for hydrogen production. Int J Hydrogen Energy 43:9569–9576. https://doi.org/10.1016/j.ijhydene.2018.04.053
Torres M, Fradinho P, Rodríguez P, Falqué E, Santos V, Domínguez H (2020) Biorefinery concept for discarded potatoes: recovery of starch and bioactive compounds. J Food Eng 275:109886. https://doi.org/10.1016/j.jfoodeng.2019.109886
Kothari R, Kumar V, Pathak VV, Ahmad S, Aoyi O, Tyagi V (2017) A critical review on factors influencing fermentative hydrogen production. Front Biosci 22:1195–1220. https://doi.org/10.2741/4542
Singh V, Singh H, Das D (2019) Optimization of the medium composition for the improvement of hydrogen and butanol production using Clostridium saccharoperbutylacetonicum DSM 14923. Int J Hydrogen Energy 44:26905–26919. https://doi.org/10.1016/j.ijhydene.2019.08.125
Yahya M, Herrmann C, Ismaili S, Jost C, Truppel I, Ghorbal A (2021) Development and optimization of an innovative three-stage bioprocess for converting food wastes to hydrogen and methane. Biochem Eng J 170:107992. https://doi.org/10.1016/j.bej.2021.107992
Basak N, Das D (2009) Photofermentative hydrogen production using purple non-sulfur bacteria Rhodobacter sphaeroides OU 001 in an annular photobioreactor: a case study. Biomass Bioenerg 33:911–919. https://doi.org/10.1016/j.ijhydene.2014.02.093
Cakır A, Ozmihci S, Kargi F (2010) Comparison of bio-hydrogen production from hydrolyzed wheat starch by mesophilic and thermophilic dark fermentation. Int J Hydrogen Energy 35:13214–13218. https://doi.org/10.1016/j.ijhydene.2010.09.029
Argun H, Kargi F, Kapdan IK (2009) Hydrogen production by combined dark and light fermentation of ground wheat solution. Int J Hydrogen Energy 34:4305–4311. https://doi.org/10.1016/j.ijhydene.2009.03.033
Rao R, Basak N (2021) Optimization and modelling of dark fermentative hydrogen production from cheese whey by Enterobacter aerogenes 2822. Int J Hydrogen Energy 46:1777–1800. https://doi.org/10.1016/j.ijhydene.2020.10.142
Basak N, Jana AK, Das D (2014) Optimization of molecular hydrogen production by Rhodobacter sphaeroides OU 001 in the annular photobioreactor using response surface methodology. Int J Hydrogen Energy 39:11889–11901. https://doi.org/10.1016/j.ijhydene.2014.05.108
Nielsen SS (2010) Phenol-sulfuric acid method for total carbohydrates. Food analysis laboratory manual Springer. https://doi.org/10.1007/978-1-4419-1463-7_6
Argun H, Kargi F (2010) Bio-hydrogen production from ground wheat starch by continuous combined fermentation using annular-hybrid bioreactor. Int J Hydrogen Energy 35:6170–6178. https://doi.org/10.1016/j.ijhydene.2010.03.132
Das SR, Basak N (2021) Molecular biohydrogen production by dark and photo fermentation from wastes containing starch: recent advancement and future perspective. Bioprocess Biosyst Eng 44:1–25. https://doi.org/10.1007/s00449-020-02422-5
Boboescu IZ, Ilie M, Gherman VD, Mirel I, Pap B, Negrea A, Kondorosi É, Bíró T, Maróti G (2014) Revealing the factors influencing a fermentative biohydrogen production process using industrial wastewater as fermentation substrate. Biotechnol Biofuels 7:1–15. https://doi.org/10.1186/s13068-014-0139-1
Basak N, Jana AK, Das D, Saikia D (2014) Photofermentative molecular biohydrogen production by purple-non-sulfur (PNS) bacteria in various modes: the present progress and future perspective. Int J Hydrogen Energy 39:6853–6871. https://doi.org/10.1016/j.ijhydene.2014.02.093
Xia A, Jacob A, Herrmann C, Murphy JD (2016) Fermentative bio-hydrogen production from galactose. Energy 96:346–354. https://doi.org/10.1016/j.energy.2015.12.087
Whiteman J, Kana EG (2014) Comparative assessment of the artificial neural network and response surface modelling efficiencies for biohydrogen production on sugar cane molasses. BioEnergy Research 7:295–305. https://doi.org/10.1007/s12155-013-9375-7
Nath K, Muthukumar M, Kumar A, Das D (2008) Kinetics of two-stage fermentation process for the production of hydrogen. Int J Hydrogen Energy 33:1195–1203. https://doi.org/10.1016/j.ijhydene.2007.12.011
Policastro G, Luongo V, Frunzo L, Fabbricino M (2021) A comprehensive review of mathematical models of photo fermentation. Crit Rev Biotechnol 41:628–648. https://doi.org/10.1080/07388551.2021.1873241
Hitam C, Jalil A (2020) A review on biohydrogen production through photo-fermentation of lignocellulosic biomass. Biomass Conversion and Biorefinery 1–19. https://doi.org/10.1007/s13399-020-01140-y
Ghosh S, Dutta S, Chowdhury R (2020) Ameliorated hydrogen production through integrated dark-photo fermentation in a flat plate photobioreactor: mathematical modelling and optimization of energy efficiency. Energy Convers Manage 226:113549. https://doi.org/10.1016/j.enconman.2020.113549
Basak N, Jana AK, Das D (2016) CFD modeling of hydrodynamics and optimization of photofermentative hydrogen production by Rhodopseudomonas palustris DSM 123 in annular photobioreactor. Int J Hydrogen Energy 41:7301–7317. https://doi.org/10.1016/j.ijhydene.2016.02.126
Hitit ZY, Lazaro CZ, Hallenbeck PC (2017) Single stage hydrogen production from cellulose through photo-fermentation by a co-culture of Cellulomonas fimi and Rhodopseudomonas palustris. Int J Hydrogen Energy 42:6556–6566. https://doi.org/10.1016/j.ijhydene.2016.12.035
Dolly S, Pandey A, Pandey BK, Gopal R (2015) Process parameter optimization and enhancement of photo-biohydrogen production by mixed culture of Rhodobacter sphaeroides NMBL-02 and Escherichia coli NMBL-04 using Fe-nanoparticle. International Journal of Hydrogen Energy 40:16010–16020. https://doi.org/10.1016/j.ijhydene.2015.09.089
Sun Q, Xiao W, Xi D, Shi J, Yan X, Zhou Z (2010) Statistical optimization of biohydrogen production from sucrose by a co-culture of Clostridium acidisoli and Rhodobacter sphaeroides. Int J Hydrogen Energy 35:4076–4084. https://doi.org/10.1016/j.ijhydene.2010.01.145
Zagrodnik R, Łaniecki M (2017) Hydrogen production from starch by co-culture of Clostridium acetobutylicum and Rhodobacter sphaeroides in one step hybrid dark-and photofermentation in repeated fed-batch reactor. Biores Technol 224:298–306. https://doi.org/10.1016/j.biortech.2016.10.060
Takagi D, Okamura S, Tanaka K, Ikenaga N, Iwashima M, Haghparast SMA, Tanaka N, Miyake J (2016) Characterization of hydrogen production by the co-culture of dark-fermentative and photosynthetic bacteria. Res Chem Intermed 42:7713–7722. https://doi.org/10.1007/s11164-016-2656-y
Argun H, Kargi F (2010) Photo-fermentative hydrogen gas production from dark fermentation effluent of ground wheat solution: effects of light source and light intensity. Int J Hydrogen Energy 35:1595–1603. https://doi.org/10.1016/j.ijhydene.2009.12.040
Argun H, Kargi F (2010) Effects of light source, intensity and lighting regime on bio-hydrogen production from ground wheat starch by combined dark and photo-fermentations. Int J Hydrogen Energy 35:1604–1612. https://doi.org/10.1016/j.ijhydene.2009.12.033
Acknowledgements
The first author Satya Ranjan Das wishes to thank Ministry of Education, Government of India, for providing the fellowships. The authors are thankful to the Department of Biotechnology, Dr. B R Ambedkar National Institute of Technology Jalandhar (India), for providing facilities to carry out the research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Single-stage combined dark and photo fermentation was carried out using potato waste as feedstock in a custom-made 2 L photo bioreactor.
• Optimization was carried out by a central composite design matrix to study the interactive effect of the independent variable on response.
• The optimum value for carbohydrate and FeCl3 concentration was found to be 2 g/L and 0.2 g/L at pH 7.
• Maximum H2 production was 1580 mL H2/L media.
• Fifty-three percent of the initial carbohydrate was converted to hydrogen with yield of 6.31 mol H2/mol carbohydrate.
Rights and permissions
About this article
Cite this article
Das, S.R., Basak, N. Optimization of process parameters for enhanced biohydrogen production using potato waste as substrate by combined dark and photo fermentation. Biomass Conv. Bioref. 14, 4791–4811 (2024). https://doi.org/10.1007/s13399-022-02588-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-02588-w