Abstract
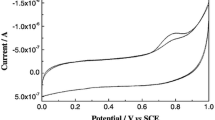
A simple one-pot approach for the synthesis of gold-loaded copper oxide (Au–CuOx) nanoplates was employed to develop a sensitive electrochemical surface enhanced Raman scattering (EC-SERS) substrate for the detection of anti-cancer drug hydroxycarbamide (HC). HC has been used for decades as a therapeutic agent against sickle cell anaemia and various cancers. The Au–CuOx substrate was characterized using various spectroscopic and imaging techniques, including XRD, XPS, FTIR, TEM and SEM. The electrochemical impact on the SERS-based sensing capability was investigated by immobilizing the bimetallic material on a conductive glass support acting as a working electrode. First-principle calculations were carried out to predict the electrostatic properties, electron transfer contribution and adsorption energetics of HC molecules on the nanostructured surface. Spectroscopic and theoretical investigations suggested that Au-loaded copper oxide exhibits inherent surface plasmon resonance (SPR) properties that can be notably enhanced by incorporating the electrochemical factor. Several prominent Raman peaks associated with HC and ascribed by the NCN bending, CN stretching, NO stretching, CO stretching, and NH2 wagging vibrational modes showed substantial enhancements upon the electrochemically induced interaction between the SERS substrate and the drug molecules. A limit of detection of one-tenth of nanomolar concentration could be achieved under optimum conditions. The presented methodology could be potential in the context of biomedical laboratories for the sake of quick and reliable assessment.








Similar content being viewed by others
References
Teng, S.; Ma, C.; Yu, Y.; Yi, C.: Hydroxyurea promotes TET1 expression and induces apoptosis in osteosarcoma cells. Biosci. Rep. (2019). https://doi.org/10.1042/BSR20190456/219136
Teng, J.; Hejazi, S.; Hiddingh, L.; Carvalho, L.; De Gooijer, M.C.; Wakimoto, H.; Barazas, M.; Tannous, M.; Chi, A.S.; Noske, D.P.; Wesseling, P.; Wurdinger, T.; Batchelor, T.T.; Tannous, B.A.: Recycling drug screen repurposes hydroxyurea as a sensitizer of glioblastomas to temozolomide targeting de novo DNA synthesis, irrespective of molecular subtype. Neuro Oncol. 20, 642–654 (2018). https://doi.org/10.1093/NEUONC/NOX198
Angona, A.; Bellosillo, B.; Alvarez-Larrán, A.; Martínez-Avilés, L.; Camacho, L.; Pairet, S.; Fernández-Rodriguez, M.C.; Ancochea, À.; Besses, C.: Genetic predisposition to molecular response in patients with myeloproliferative neoplasms treated with hydroxycarbamide. Leuk. Res. 37, 917–921 (2013). https://doi.org/10.1016/J.LEUKRES.2013.03.013
Berthaut, I.; Bachir, D.; Kotti, S.; Chalas, C.; Stankovic, K.; Eustache, F.; Ravel, C.; Habibi, A.; Brailly-Tabard, S.; Lévy-Dutel, L.; Bleibtreu, A.; Simon, T.; Galactéros, F.; Lionnet, F.; Mandelbaum, J.: Adverse effect of hydroxyurea on spermatogenesis in patients with sickle cell anemia after 6 months of treatment. Blood 130, 2354–2356 (2017). https://doi.org/10.1182/BLOOD-2017-03-771857
Liebelt, E.L.; Balk, S.J.; Faber, W.; Fisher, J.W.; Hughes, C.L.; Lanzkron, S.M.; Lewis, K.M.; Marchetti, F.; Mehendale, H.M.; Rogers, J.M.; Shad, A.T.; Skalko, R.G.; Stanek, E.J.; Shelby, M.D.: NTP-CERHR expert panel report on the reproductive and developmental toxicity of hydroxyurea. Birth Defects Res. B Dev. Reprod. Toxicol. 80, 259–366 (2007). https://doi.org/10.1002/BDRB.20123
Marahatta, A.; Ware, R.E.: Hydroxyurea: analytical techniques and quantitative analysis. Blood Cells Mol. Dis. 67, 135–142 (2017). https://doi.org/10.1016/J.BCMD.2017.08.009
Pujari, M.P.; Barrientos, A.; Muggia, F.M.; Koda, R.T.: Determination of hydroxyurea in plasma and peritoneal fluid by high-performance liquid chromatography using electrochemical detection. J. Chromatogr. B Biomed. Sci. Appl. 694, 185–191 (1997). https://doi.org/10.1016/S0378-4347(97)00120-5
Hai, X.; Guo, M.; Gao, C.; Zhou, J.: Quantification of hydroxyurea in human plasma by HPLC–MS/MS and its application to pharmacokinetics in patients with chronic myeloid leukaemia. J. Pharm. Biomed. Anal. 137, 213–219 (2017). https://doi.org/10.1016/J.JPBA.2017.01.008
Cheuquepán, W.; Orts, J.M.; Rodes, A.: Hydroxyurea electrooxidation at gold electrodes. In situ infrared spectroelectrochemical and DFT characterization of adsorbed intermediates. Electrochim. Acta 246, 951–962 (2017). https://doi.org/10.1016/J.ELECTACTA.2017.06.091
Pathak, P.K.; Kumar, A.; Prasad, B.B.: Functionalized nitrogen doped graphene quantum dots and bimetallic Au/Ag core-shell decorated imprinted polymer for electrochemical sensing of anticancerous hydroxyurea. Biosens. Bioelectron. 127, 10–18 (2019). https://doi.org/10.1016/J.BIOS.2018.11.055
Haroon, M.; Tahir, M.; Nawaz, H.; Majeed, M.I.; Al-Saadi, A.A.: Surface-enhanced Raman scattering (SERS) spectroscopy for prostate cancer diagnosis: a review. Photodiagnosis Photodyn. Ther. 37, 102690 (2022). https://doi.org/10.1016/J.PDPDT.2021.102690
Haroon, M.; Iali, W.; Al-Saadi, A.A.: Conformational analysis and concentration detection of linuron: spectroscopic NMR and SERS study. Spectrochim. Acta A Mol. Biomol. Spectrosc. 263, 120223 (2021). https://doi.org/10.1016/J.SAA.2021.120223
Haroon, M.; Iali, W.; Saleh, T.A.; Al-Saadi, A.A.: NMR evidence for hydrogen bonding stabilized anti conformation of 1-methoxy-1-methyl-3-phenylurea and the concentration detection by SERS. J. Mol. Liq. 357, 119096 (2022). https://doi.org/10.1016/J.MOLLIQ.2022.119096
Mahar, N.; Haroon, M.; Saleh, T.A.; Al-Saadi, A.A.: Spectroanalytical SERS-based detection of trace-level procainamide using green-synthesized gold nanoparticles. Surf. Interf. 31, 102059 (2022). https://doi.org/10.1016/J.SURFIN.2022.102059
Schuepfer, D.B.; Badaczewski, F.; Guerra-Castro, J.M.; Hofmann, D.M.; Heiliger, C.; Smarsly, B.; Klar, P.J.: Assessing the structural properties of graphitic and non-graphitic carbons by Raman spectroscopy. Carbon N Y. 161, 359–372 (2020). https://doi.org/10.1016/J.CARBON.2019.12.094
Zhang, W.; Ma, J.; Sun, D.W.: Raman spectroscopic techniques for detecting structure and quality of frozen foods: principles and applications. Crit. Rev. Food Sci. Nutr. 61, 2623–2639 (2020). https://doi.org/10.1080/10408398.2020.1828814
Thomas, G.J.: Raman spectroscopy of protein and nucleic acid assemblies. Annu. Rev. Biophys. Biomol. Struct. 28, 1–27 (2003). https://doi.org/10.1146/ANNUREV.BIOPHYS.28.1.1
Al-Saadi, A.A.; Haroon, M.; Popoola, S.A.; Saleh, T.A.: Sensitive SERS detection and characterization of procaine in aqueous media by reduced gold nanoparticles. Sens. Actuators B Chem. 304, 127057 (2020). https://doi.org/10.1016/J.SNB.2019.127057
Haroon, M.; Abdulazeez, I.; Saleh, T.A.; Al-Saadi, A.A.: SERS-based trace-level quantification of sulindac: spectroscopic and molecular modeling evaluation. J. Mol. Liq. 312, 113402 (2020). https://doi.org/10.1016/j.molliq.2020.113402
Haroon, M.; Abdulazeez, I.; Saleh, T.A.; Al-Saadi, A.A.: Electrochemically modulated SERS detection of procaine using FTO electrodes modified with silver-decorated carbon nanosphere. Electrochim. Acta 387, 138463 (2021). https://doi.org/10.1016/J.ELECTACTA.2021.138463
Mahar, N.; Haroon, M.; Saleh, T.A.; Al-Saadi, A.A.: Fast and sensitive detection of procainamide: combined SERS and DFT modeling studies. J. Mol. Liq. 343, 117633 (2021). https://doi.org/10.1016/J.MOLLIQ.2021.117633
Kamran, M.; Haroon, M.; Popoola, S.A.; Almohammedi, A.R.; Al-Saadi, A.A.; Saleh, T.A.: Characterization of valeric acid using substrate of silver nanoparticles with SERS. J. Mol. Liq. 273, 536–542 (2019). https://doi.org/10.1016/J.MOLLIQ.2018.10.037
Puente, C.; Sánchez-Domínguez, M.; Brosseau, C.L.; López, I.: Silver-chitosan and gold-chitosan substrates for surface-enhanced Raman spectroscopy (SERS): effect of nanoparticle morphology on SERS performance. Mater. Chem. Phys. 260, 124107 (2021). https://doi.org/10.1016/J.MATCHEMPHYS.2020.124107
Haroon, M.; Ashraf, M.; Ullah, N.; Nawaz Tahir, M.; Al-Saadi, A.A.: SERS and EC-SERS detection of local anesthetic procaine using Pd loaded highly reduced graphene oxide nanocomposite substrate. Spectrochim. Acta A Mol. Biomol. Spectrosc. 278, 121381 (2022). https://doi.org/10.1016/J.SAA.2022.121381
Ferrando, R.; Jellinek, J.; Johnston, R.L.: Nanoalloys: from theory to applications of alloy clusters and nanoparticles. Chem. Rev. 108, 845–910 (2008). https://doi.org/10.1021/CR040090G
Liu, Y.C.; Yang, K.H.; Yang, S.J.: Sonoelectrochemical synthesis of spike-like gold–silver alloy nanoparticles from bulk substrates and the application on surface-enhanced Raman scattering. Anal. Chim. Acta 572, 290–294 (2006). https://doi.org/10.1016/J.ACA.2006.05.038
Yang, L.; Jiang, X.; Ruan, W.; Zhao, B.; Xu, W.; Lombardi, J.R.: Observation of enhanced Raman scattering for molecules adsorbed on TiO2 nanoparticles: charge-transfer contribution. J. Phys. Chem. C 112, 20095–20098 (2008). https://doi.org/10.1021/JP8074145
Lei, D.Y.; Li, J.; Ong, H.C.: Tunable surface plasmon mediated emission from semiconductors by using metal alloys. Appl. Phys. Lett. 91, 021112 (2007). https://doi.org/10.1063/1.2752770
Shan, G.; Zheng, S.; Chen, S.; Chen, Y.; Liu, Y.: Multifunctional ZnO/Ag nanorod array as highly sensitive substrate for surface enhanced Raman detection. Colloids Surf. B Biointerfaces 94, 157–162 (2012). https://doi.org/10.1016/J.COLSURFB.2012.01.037
Yang, L.; Jiang, X.; Ruan, W.; Yang, J.; Zhao, B.; Xu, W.; Lombardi, J.R.: Charge-transfer-induced surface-enhanced Raman scattering on Ag−TiO2 nanocomposites. J. Phys. Chem. C 113, 16226–16231 (2009). https://doi.org/10.1021/JP903600R
Wang, Y.; Song, W.; Ruan, W.; Yang, J.; Zhao, B.; Lombardi, J.R.: SERS spectroscopy used to study an adsorbate on a nanoscale thin film of CuO coated with Ag. J. Phys. Chem. C 113, 8065–8069 (2009). https://doi.org/10.1021/JP900052Q
Tahir, D.; Tougaard, S.: Electronic and optical properties of Cu, CuO and Cu2O studied by electron spectroscopy. J. Phys. Condens. Matter 24, 175002 (2012). https://doi.org/10.1088/0953-8984/24/17/175002
Issaoui, N.; Ghalla, H.; Bardak, F.; Karabacak, M.; Aouled Dlala, N.; Flakus, H.T.; Oujia, B.: Combined experimental and theoretical studies on the molecular structures, spectroscopy, and inhibitor activity of 3-(2-thienyl)acrylic acid through AIM NBO, FT-IR, FT-Raman, UV and HOMO-LUMO analyses, and molecular docking. J. Mol. Struct. (2017). https://doi.org/10.1016/j.molstruc.2016.11.019
Issaoui, N.; Abdessalem, K.; Ghalla, H.; Yaghmour, S.J.; Calvo, F.; Oujia, B.: Theoretical investigation of the relative stability of Na+Hen (n = 2–24) clusters: many-body versus delocalization effects. J. Chem. Phys. (2014). https://doi.org/10.1063/1.4900873
Kazachenko, A.S.; Akman, F.; Sagaama, A.; Issaoui, N.; Malyar, Y.N.; Vasilieva, N.Y.; Borovkova, V.S.: Theoretical and experimental study of guar gum sulfation. J. Mol. Model. (2021). https://doi.org/10.1007/s00894-020-04645-5
Jomaa, I.; Issaoui, N.; Roisnel, T.; Marouani, H.: Insight into non-covalent interactions in a tetrachlorocadmate salt with promising NLO properties: experimental and computational analysis. J. Mol. Struct. (2021). https://doi.org/10.1016/j.molstruc.2021.130730
Pang, M.; Zeng, H.C.: Highly ordered self-assemblies of submicrometer Cu2O Spheres and their hollow chalcogenide derivatives. Langmuir 26, 5963–5970 (2010). https://doi.org/10.1021/LA904292T/SUPPL_FILE/LA904292T_SI_001.PDF
Hajibabaei, H.; Little, D.J.; Pandey, A.; Wang, D.; Mi, Z.; Hamann, T.W.: Direct deposition of crystalline Ta3 N5 thin films on FTO for PEC Water splitting. ACS Appl. Mater. Interf. 11, 15457–15466 (2019). https://doi.org/10.1021/acsami.8b21194
Ma, H.; Fan, Q.; Fan, B.; Zhang, Y.; Fan, D.; Wu, D.; Wei, Q.: Formation of homogeneous Epinephrine-Melanin solutions to fabricate electrodes for enhanced photoelectrochemical biosensing. Langmuir 34, 7744–7750 (2018). https://doi.org/10.1021/acs.langmuir.8b00264
Becke, A.D.: A new mixing of Hartree-Fock and local density-functional theories. J. Chem. Phys. 98, 1372–1377 (1993). https://doi.org/10.1063/1.464304
Nnadiekwe, C.C.; Abdulazeez, I.; Haroon, M.; Peng, Q.; Jalilov, A.; Al-Saadi, A.: Impact of polypyrrole functionalization on the anodic performance of boron nitride nanosheets: insights from first-principles calculations. Front. Chem. 9, 670833 (2021). https://doi.org/10.3389/FCHEM.2021.670833/FULL
Chang, J.-H.; Antunes, M.; Umar, M.; Nnadiekwe, C.C.; Haroon, M.; Abdulazeez, I.; Alhooshani, K.; Al-Saadi, A.A.; Peng, Q.: A first-principles study on the multilayer graphene nanosheets anode performance for boron-ion battery. Nanomaterials 12, 1280 (2022). https://doi.org/10.3390/NANO12081280
Lee, D.S.; Chen, Y.W.: Au/CuO–CeO2 catalyst for preferential oxidation of CO in hydrogen-rich stream: effect of CuO content. Int. J. Hydrog. Energy 41, 3605–3612 (2016). https://doi.org/10.1016/J.IJHYDENE.2015.12.137
Batchtold, A.; Strunk, C.; Salvetat, J.P.; Bonard, J.M.; Forro, L.; Nussbau-mer, T.; Schonenberger, C.; Li, Z.; Xie, S.S.; Qian, L.X.; Chang, B.H.; Zou, B.S.; Zhou, W.Y.; Zhao, A.; Wang, G.; Ren, F.; Huang, Z.P.; Wang, D.Z.; Wen, J.G.; Xu, J.W.; Wang, J.H.; Calvet, L.E.; Chen, J.; Klemic, J.F.; Reed, M.A.; Phys Lett, A.; Bower, C.; Zhou, O.; Zhu, W.; Werder, D.J.; Jin, S.H.; Sonoda, S.; Tanaka, C.; Murakami, H.; Yamakawa, H.; Wenzhong Wang, B.; Wang, G.; Wang, X.; Zhan, Y.; Liu, Y.; Zheng, C.: Synthesis and characterization of Cu2O nanowires by a novel reduction route**. Phys. Lett. 277, 705 (1996). https://doi.org/10.1002/1521-4095
Ghijsen, J.; Tjeng, L.H.; Van Elp, J.; Eskes, H.; Westerink, J.; Sawatzky, G.A.; Czyzyk, M.T.: Electronic structure of Cu2O and CuO. Phys. Rev. B 38, 11322 (1988). https://doi.org/10.1103/PhysRevB.38.11322
Zhou, L.J.; Zou, Y.C.; Zhao, J.; Wang, P.P.; Feng, L.L.; Sun, L.W.; Wang, D.J.; Li, G.D.: Facile synthesis of highly stable and porous Cu2O/CuO cubes with enhanced gas sensing properties. Sens. Actuators B Chem. 188, 533–539 (2013). https://doi.org/10.1016/J.SNB.2013.07.059
Tajammul Hussain, S.; Iqbal, M.; Mazhar, M.: Size control synthesis of starch capped-gold nanoparticles. J. Nanoparticle Res. 11, 1383–1391 (2009). https://doi.org/10.1007/S11051-008-9525-6/FIGURES/10
Torkzadeh-Mahani, A.; Mohammadi, A.; Torkzadeh-Mahani, M.; Mohamadi, M.: Voltammetric determination of anticancer drug hydroxyurea using a carbon paste electrode, incorporating TiO2 nanoparticles Anal. Bioanal Electrochem. 9, 117–125 (2017)
Naik, K.M.; Alagur, M.M.; Nandibewoor, S.T.: Electrochemical response of hydroxyurea by different voltammetric techniques at carbon paste electrode. Anal. Methods (2013). https://doi.org/10.1039/c3ay41680a
Torkzadeh-Mahani, A.; Mohammadi, A.; Torkzadeh-Mahani, M.; Mohamadi, M.: Voltammetric determination of the anticancer drug hydroxyurea using a carbon paste electrode incorporating TiO2 nanoparticles. Anal. Bioanal. Electrochem. 9, 117–125 (2017)
Naik, K.M.; Ashi, C.R.; Nandibewoor, S.T.: Anodic voltammetric behavior of hydroxyurea and its electroanalytical determination in pharmaceutical dosage form and urine. J. Electroanal. Chem. 755, 109–114 (2015). https://doi.org/10.1016/J.JELECHEM.2015.07.038
Legrand, T.; Rakotoson, M.-G.; Galactéros, F.; Bartolucci, P.; Hulin, A.: Determination of hydroxyurea in human plasma by HPLC-UV using derivatization with xanthydrol. J. Chromatogr. B 1064, 85–91 (2017). https://doi.org/10.1016/j.jchromb.2017.09.008
James, H.; Nahavandi, M.; Wyche, M.Q.; Taylor, R.E.: Quantitative analysis of trimethylsilyl derivative of hydroxyurea in plasma by gas chromatography–mass spectrometry. J. Chromatogr. B 831, 42–47 (2006). https://doi.org/10.1016/J.JCHROMB.2005.11.033
Jong, Y.J.; Hsu, H.O.; Wu, H.L.; Kou, H.S.; Wu, S.M.: Analysis of hydroxyurea in human plasma by high performance liquid chromatography with electrochemical detection. Anal. Chim. Acta 488, 223–230 (2003). https://doi.org/10.1016/S0003-2670(03)00702-5
Haghshenas, M.; Mazloum-Ardakani, M.; Alizadeh, Z.; Vajhadin, F.; Naeimi, H.: A sensing platform using ag/pt core-shell nanostructures supported on multiwalled carbon nanotubes to detect hydroxyurea. Electroanalysis 32, 2137–2145 (2020). https://doi.org/10.1002/ELAN.202060020
Cazelles, R.; Shukla, R.P.; Ware, R.E.; Vinks, A.A.; Ben-Yoav, H.: Electrochemical determination of hydroxyurea in a complex biological matrix using MoS2-modified electrodes and chemometrics. Biomedicines 9, 6 (2020). https://doi.org/10.3390/BIOMEDICINES9010006
Lombardi, J.R.; Birke, R.L.: A unified view of surface-enhanced raman scattering. Acc. Chem. Res. 42, 734–742 (2009). https://doi.org/10.1021/AR800249Y/ASSET/IMAGES/AR-2008-00249Y_M003.GIF
Fu, X.; Jiang, T.; Zhao, Q.; Yin, H.: Charge-transfer contributions in surface-enhanced Raman scattering from Ag, Ag2S and Ag2Se substrates. J. Raman Spectrosc. 43, 1191–1195 (2012). https://doi.org/10.1002/JRS.4033
Willets, K.A.: Probing nanoscale interfaces with electrochemical surface-enhanced Raman scattering. Curr. Opin. Electrochem. 13, 18–24 (2019). https://doi.org/10.1016/J.COELEC.2018.10.005
Wu, D.Y.; Li, J.F.; Ren, B.; Tian, Z.Q.: Electrochemical surface-enhanced Raman spectroscopy of nanostructures. Chem. Soc. Rev. 37, 1025–1041 (2008). https://doi.org/10.1039/B707872M
Pearson, R.G.: Absolute electronegativity and hardness: application to inorganic chemistry. Inorg. Chem. 27, 734–740 (1988)
Al-Saadi, A.A.: Understanding the influence of electron-donating and electron-withdrawing substituents on the anticorrosive properties of imidazole: a quantum-chemical approach. Arab. J. Sci. Eng. 45, 153–166 (2020). https://doi.org/10.1007/S13369-019-04167-0/TABLES/10
Zarrouk, A.; El Ouali, I.; Bouachrine, M.; Hammouti, B.; Ramli, Y.; Essassi, E.M.; Warad, I.; Aouniti, A.; Salghi, R.: Theoretical approach to the corrosion inhibition efficiency of some quinoxaline derivatives of steel in acid media using the DFT method. Res. Chem. Intermed. 39, 1125–1133 (2013). https://doi.org/10.1007/S11164-012-0671-1/FIGURES/3
Obot, I.B.: HSAB descriptors of thiadiazole derivatives calculated by DFT: possible relationship as mild steel corrosion inhibitors special issue on “material and corrosion”-sustainability journal-impact factor-2.576. View project Discovery of Novel Molecules for Corrosion Protection Using Computational Chemistry View project, (n.d.).
Wang, Y.; Ji, W.; Sui, H.; Kitahama, Y.; Ruan, W.; Ozaki, Y.; Zhao, B.: Exploring the effect of intermolecular H-bonding: a study on charge-transfer contribution to surface-enhanced Raman scattering of p-mercaptobenzoic acid. J. Phys. Chem. C 118, 10191–10197 (2014). https://doi.org/10.1021/JP5025284/SUPPL_FILE/JP5025284_SI_001.PDF
Rekik, N.; Issaoui, N.; Ghalla, H.; Oujia, B.; Wójcik, M.J.: IR spectral density of H-bonds. Both intrinsic anharmonicity of the fast mode and the H-bond bridge. Part I: Anharmonic coupling parameter and temperature effects. J. Mol. Struct. Theochem. 821, 9–21 (2007). https://doi.org/10.1016/J.THEOCHEM.2007.06.016
Acknowledgements
Authors thank King Fahd University of Petroleum & Minerals (KFUPM) for its support through Project No. DF191043.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Haroon, M., Al-Saadi, A.A. The Role of Gold-Loaded Copper Oxide Nanoplates in the Formulation of Sensitive EC-SERS Substrates for the Detection of Anti-Cancer Drugs: Spectro–Electro and DFT Studies. Arab J Sci Eng 49, 673–684 (2024). https://doi.org/10.1007/s13369-023-08384-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-023-08384-6