Abstract
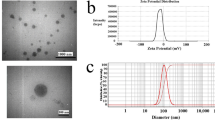
Limited targeted therapies are available for triple-negative breast cancer (TNBC). Thus, the current research focused on developing a targeted protein nanoparticle for TNBC. First, the doxorubicin hydrochloride (Dox)–loaded genipin-crosslinked whey protein nanoparticles (WD) were prepared and optimised by the QbD method using BBD. The hydrodynamic diameter of WD was found to be 364.38 ± 49.23 nm, zeta potential −27.59 ± 1.038 mV, entrapment 63.03 ± 3.625% and Dox loading was found to be 1.419 ± 0.422%. The drug recovery after 18 months of storage was 69%. Then, it was incubated with NAC to obtain modified WD (CyWD). WD followed first-order release kinetics, whereas CyWD followed the Higuchi model. Hemagglutination and hemolysis were not found qualitatively in WD and CyWD. Upon injecting the nanoformulations to 4T1-induced mice, the highest efficacy was found to be in CyWD followed by WD and Dox injection. Upon histopathological observance, it was found that the CyWD group gave the most significant damage to the 4T1 tumour tissue. Thus, NAC-modified protein nanoparticles carrying chemotherapeutic agents can be an excellent targeted therapeutic system against TNBC.
Graphical abstract









Similar content being viewed by others
Availability of data and materials
All data and resource materials are available with the correspondence and first author.
Abbreviations
- ATCC:
-
American Type Culture Collection
- BBD:
-
Box Behnken Design
- CPCSEA:
-
Committee for the Purpose of Control and Supervision of Experiments on Animals
- DMSO:
-
Dimethyl sulfoxide
- Dox:
-
Doxorubicin hydrochloride
- EDTA:
-
Ethylenediamine tetraacetic acid
- FBS:
-
Fetal bovine serum
- FITC:
-
Fluorescein isothiocyanate
- MWCO:
-
Molecular weight cut-off
- MTT:
-
3-(4,5-Dimethylthiazol-2-Yl)-2,5-Diphenyltetrazolium Bromide
- NCCS:
-
National Centre for Cell Science
- PBS:
-
Phosphate buffered saline
- QbD:
-
Quality by Design
- RBC:
-
Red blood cells
- TNBC:
-
Triple-negative breast cancer
References
Hwang S-Y, Park S, Kwon Y. Recent therapeutic trends and promising targets in triple negative breast cancer. Pharmacol Ther. 2019;199:30–57. https://doi.org/10.1016/j.pharmthera.2019.02.006.
Khan I, Saeed K, Khan I. Nanoparticles: properties, applications and toxicities. Arab J Chem. 2019;12(7):908–31. https://doi.org/10.1016/j.arabjc.2017.05.011.
Mudshinge SR, Deore AB, Patil S, Bhalgat CM. Nanoparticles: emerging carriers for drug delivery. Saudi Pharmaceutical Journal. 2011;19(3):129–41. https://doi.org/10.1016/j.jsps.2011.04.001.
Jain A, Singh SK, Arya SK, Kundu SC, Kapoor S. Protein nanoparticles: promising platforms for drug delivery applications. ACS Biomater Sci Eng. 2018;4(12):3939–61. https://doi.org/10.1021/acsbiomaterials.8b01098.
Patel S. Emerging trends in nutraceutical applications of whey protein and its derivatives. J Food Sci Technol. 2015;52(11):6847–58. https://doi.org/10.1007/s13197-015-1894-0.
Souza FN, Gebara C, Ribeiro MCE, Chaves KS, Gigante ML, Grosso CRF. Production and characterization of microparticles containing pectin and whey proteins. Food Res Int. 2012;49(1):560–6. https://doi.org/10.1016/j.foodres.2012.07.041.
Corrochano AR, Buckin V, Kelly PM, Giblin L. Invited review: whey proteins as antioxidants and promoters of cellular antioxidant pathways. J Dairy Sci. 2018;101(6):4747–61. https://doi.org/10.3168/jds.2017-13618.
Kanoujia J, Singh M, Singh P, Parashar P, Tripathi CB, Arya M, et al. Genipin crosslinked soy-whey based bioactive material for atorvastatin loaded nanoparticles: preparation, characterization and in vivo antihyperlipidemic study. RSC Adv. 2016;6(96):93275–87. https://doi.org/10.1039/C6RA16830B.
Wei Y, Zhan X, Dai L, Zhang L, Mao L, Yuan F, et al. Formation mechanism and environmental stability of whey protein isolate-zein core-shell complex nanoparticles using the pH-shifting method. LWT. 2020;110605.
Giroux HJ, Britten M. Encapsulation of hydrophobic aroma in whey protein nanoparticles. J Microencapsul. 2011;28(5):337–43. https://doi.org/10.3109/02652048.2011.569761.
Hortobágyi GN. Anthracyclines in the treatment of cancer. An overview Drugs. 1997;54(Suppl 4):1–7. https://doi.org/10.2165/00003495-199700544-00003.
Reddy LH, Murthy R. Pharmacokinetics and biodistribution studies of doxorubicin loaded poly (butyl cyanoacrylate) nanoparticles synthesized by two different techniques. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2004;148(2):161–6.
Hasegawa M, Takahashi H, Rajabi H, Alam M, Suzuki Y, Yin L et al. Functional interactions of the cystine/glutamate antiporter, CD44v and MUC1-C oncoprotein in triple-negative breast cancer cells. Oncotarget. 2016;7(11).
Koppula P, Zhuang L, Gan B. Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell. 2021;12(8):599–620. https://doi.org/10.1007/s13238-020-00789-5.
Alothaim T, Charbonneau M, Tang X. HDAC6 inhibitors sensitize non-mesenchymal triple-negative breast cancer cells to cysteine deprivation. Sci Rep. 2021;11(1):10956. https://doi.org/10.1038/s41598-021-90527-6.
Prajapati R, Garcia-Garrido E, Somoza Á. Albumin-based nanoparticles for the delivery of doxorubicin in breast cancer. Cancers. 2021;13(12):3011.
Kayani Z, Bordbar A-K, Firuzi O. Novel folic acid-conjugated doxorubicin loaded β-lactoglobulin nanoparticles induce apoptosis in breast cancer cells. Biomed Pharmacother. 2018;107:945–56. https://doi.org/10.1016/j.biopha.2018.08.047.
Morshed RA, Muroski ME, Dai Q, Wegscheid ML, Auffinger B, Yu D, et al. Cell-penetrating peptide-modified gold nanoparticles for the delivery of doxorubicin to brain metastatic breast cancer. Mol Pharm. 2016;13(6):1843–54. https://doi.org/10.1021/acs.molpharmaceut.6b00004.
Prados J, Melguizo C, Ortiz R, Vélez C, Alvarez PJ, Arias JL, et al. Doxorubicin-loaded nanoparticles: new advances in breast cancer therapy. Anticancer Agents Med Chem. 2012;12(9):1058–70. https://doi.org/10.2174/187152012803529646.
Singh S, Maurya P, Saraf SA. Cutting edge targeting strategies utilizing nanotechnology in breast cancer therapy. Frontiers in Anti-Cancer Drug Discovery 2019;10:180.
Singh S, Singh P, Mishra N, Maurya P, Singh N, Nisha R, et al. Advanced drug delivery systems in breast cancer. Advanced Drug Delivery Systems in the Management of Cancer. Elsevier; 2021;107–26.
Mohamed AI, Abd-Motagaly AME, Ahmed OAA, Amin S, Mohamed Ali AI. Investigation of drug–polymer compatibility using chemometric-assisted UV-spectrophotometry. Pharmaceutics. 2017;9(1):7.
Teng Z, Luo Y, Wang Q. Nanoparticles synthesized from soy protein: preparation, characterization, and application for nutraceutical encapsulation. J Agric Food Chem. 2012;60(10):2712–20. https://doi.org/10.1021/jf205238x.
Guideline I. Stability testing of new drug substances and products. Q1A (R2), current step. 2003;4:1–24.
Li Y, Angelova A, Hu F, Garamus VM, Peng C, Li N, et al. PH responsiveness of hexosomes and cubosomes for combined delivery of Brucea javanica oil and doxorubicin. Langmuir. 2019;35(45):14532–42.
Tripathi CB, Parashar P, Arya M, Singh M, Kanoujia J, Kaithwas G, et al. QbD-based development of α-linolenic acid potentiated nanoemulsion for targeted delivery of doxorubicin in DMBA-induced mammary gland carcinoma: in vitro and in vivo evaluation. Drug Deliv Transl Res. 2018;8(5):1313–34. https://doi.org/10.1007/s13346-018-0525-5.
Akbaribazm M, Khazaei MR, Khazaei M. Trifolium pratense L.(red clover) extract and doxorubicin synergistically inhibits proliferation of 4T1 breast cancer in tumor‐bearing BALB/c mice through modulation of apoptosis and increase antioxidant and anti‐inflammatory related pathways. Food Science & Nutrition. 2020;8(8):4276–90.
Kahoush M, Behary N, Cayla A, Mutel B, Guan J, Nierstrasz V. Genipin-mediated immobilization of glucose oxidase enzyme on carbon felt for use as heterogeneous catalyst in sustainable wastewater treatment. J Environ Chem Eng. 2021;9. https://doi.org/10.1016/j.jece.2021.105633
Michel P, Abedinzadeh Z, Grajcar L, Baron M. Spectroscopic study of N-acetylcysteine and N-acetylcystine/hydrogen peroxide complexation. Chem Phys. 1998;228:279–91. https://doi.org/10.1016/S0301-0104(97)00337-6.
Shen H, Gao Q, Ye Q, Yang S, Wu Y, Huang Q, et al. Peritumoral implantation of hydrogel-containing nanoparticles and losartan for enhanced nanoparticle penetration and antitumor effect. Int J Nanomedicine. 2018;13:7409–26. https://doi.org/10.2147/ijn.s178585.
Tang X, Ding CK, Wu J, Sjol J, Wardell S, Spasojevic I, et al. Cystine addiction of triple-negative breast cancer associated with EMT augmented death signaling. Oncogene. 2017;36(30):4235–42. https://doi.org/10.1038/onc.2016.394.
Bounous G. Whey protein concentrate (WPC) and glutathione modulation in cancer treatment. Anticancer Res. 2000;20(6c):4785–92.
Kaushik D, Bansal G. Four new degradation products of doxorubicin: an application of forced degradation study and hyphenated chromatographic techniques. Journal of pharmaceutical analysis. 2015;5(5):285–95. https://doi.org/10.1016/j.jpha.2015.05.003.
Kim A, Ng WB, Bernt W, Cho N-J. Validation of size estimation of nanoparticle tracking analysis on polydisperse macromolecule assembly. Sci Rep. 2019;9(1):2639. https://doi.org/10.1038/s41598-019-38915-x.
Kanoujia J, Singh M, Singh P, Saraf SA. Novel genipin crosslinked atorvastatin loaded sericin nanoparticles for their enhanced antihyperlipidemic activity. Mater Sci Eng C. 2016;69:967–76. https://doi.org/10.1016/j.msec.2016.08.011.
Zhao L, Zhang B. Doxorubicin induces cardiotoxicity through upregulation of death receptors mediated apoptosis in cardiomyocytes. Sci Rep. 2017;7(1):44735. https://doi.org/10.1038/srep44735.
Persi E, Duran-Frigola M, Damaghi M, Roush WR, Aloy P, Cleveland JL, et al. Systems analysis of intracellular pH vulnerabilities for cancer therapy. Nat Commun. 2018;9(1):2997. https://doi.org/10.1038/s41467-018-05261-x.
England CG, Miller MC, Kuttan A, Trent JO, Frieboes HB. Release kinetics of paclitaxel and cisplatin from two and three layered gold nanoparticles. Eur J Pharm Biopharm. 2015;92:120–9. https://doi.org/10.1016/j.ejpb.2015.02.017.
Delneste Y, Jeannin P, Potier L, Romero P, Bonnefoy J-Y. N-acetyl-L-cysteine exhibits antitumoral activity by increasing tumor necrosis factor α-dependent T-cell cytotoxicity. Blood. 1997;90(3):1124–32. https://doi.org/10.1182/blood.V90.3.1124.
Acknowledgements
Deshpande Labs, Bhopal, India for in-vivo studies (CPCSEA approved); University Sophisticated Instrumentation Centre-Babasaheb Bhimrao Ambedkar University: Confocal microscopy; Dr Dinesh Kumar and his scholars (Ritu Raj and Umesh Kumar), Center of Biomedical Research, Lucknow, India – centrifuge and lyophilisation.
Funding
Indian Council of Medical Research (No. 3/2/2/42/2020-NCD-III), dated 17.06.2021, awarded to Samipta Singh (SRF) and Prof. Shubhini A. Saraf (Principal Investigator). UGC-Non NET (2016–2020) and ICMR-SRF (2021 onwards) fellowship for financial support to the first author.
Author information
Authors and Affiliations
Contributions
Prof. Shubhini A. Saraf designed and supervised; Samipta Singh: wrote and performed experiments; Priyanka Maurya, Raquibun Nisha, Priya Singh, Soniya Rani and Nidhi Mishra: research co-ordination.
Corresponding author
Ethics declarations
Ethics approval
Animals utilised in the study were approved by the Institutional Animal Ethics Committee (IAEC no. DL/SS/11/20/615).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Singh, S., Maurya, P., Rani, S. et al. Development of doxorubicin hydrochloride–loaded whey protein nanoparticles and its surface modification with N-acetyl cysteine for triple-negative breast cancer. Drug Deliv. and Transl. Res. 12, 3047–3062 (2022). https://doi.org/10.1007/s13346-022-01169-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-022-01169-8