Abstract
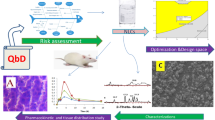
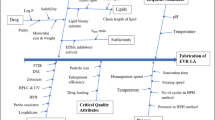
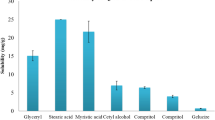
The objective of the present work was to optimize ritonavir (RTV)-loaded nanostructured lipid carriers (NLCs) to improve bioavailability using a quality by design (QbD)-based technique. Risk assessment was studied using “cause and effect” diagram followed by failure mode effect analysis (FMEA) to identify the effective high-risk variables for the formulation development. Quality target product profile (QTPP) and critical quality attributes (CQAs) were initially assigned for the proposed product. Central composite rotatable design (CCRD) was used to identify the individual and combined interactions of formulation variables. RTV-loaded NLC (RTV-NLC) was prepared using emulsification-ultrasonication method. The effect of formulation variables like ultrasound amplitude, lipid concentration, surfactant concentration on their responses like particle size, polydispersity index (PDI), and entrapment efficiency (EE) were studied by CCRD. The optimized formulation was subjected to lyophilization to obtain dry NLCs for solid-state analysis. DSC and PXRD investigations showed that RTV was molecularly dispersed in lipid matrix indicating amorphous form present in the formulation. FESEM and AFM depicted the spherical and uniform particles. The increase in solubility and dissolution rate is expected to be related to the molecular dispersion, amorphous state, of the drug in the nanoparticle. The optimized NLCs showed good physical stability during storage for 6 months. RTV-NLC was further subjected to in vitro studies and found a successful sustained release rate of 92.37 ± 1.03%. The parallel artificial membrane permeability assay (PAMPA) and everted gut sac model have demonstrated the permeation enhancement of RTV. In vivo study observed the enhanced bioavailability with 2.86-fold suggesting optimized NLC successfully overcome the issue of solubility.
Graphical abstract









Similar content being viewed by others
Data availability
Data available on request from the authors.
References
Kaspera R, Kirby BJ, Sahele T, Collier AC, Kharasch ED, Unadkat JD, et al. Investigating the contribution of CYP2J2 to ritonavir metabolism in vitro and in vivo. Biochem Pharmacol. 2014;91:109–18.
Patel D, Patel B, Patel C. Spectrophotometric method for simultaneous estimation of atazanavir sulfate and ritonavir in tablet dosage form. Drug Dev Ther. 2015;6:1.
Sudhakar B, Krishna MC, Murthy KVR. Factorial design studies of antiretroviral drug-loaded stealth liposomal injectable: PEGylation, lyophilization and pharmacokinetic studies. Appl Nanosci [Internet]. Springer Berlin Heidelberg; 2016;6:43–60. Available from: https://doi.org/10.1007/s13204-015-0408-8
Ahammed V, Narayan R, Paul J, Nayak Y, Roy B, Shavi G V, et al. Development and in vivo evaluation of functionalized ritonavir proliposomes for lymphatic targeting. Life Sci [Internet]. Elsevier Inc.; 2017;183:11–20. Available from: https://doi.org/10.1016/j.lfs.2017.06.022
Makwana V, Jain R, Patel K, Nivsarkar M, Joshi A. Solid lipid nanoparticles (SLN) of Efavirenz as lymph targeting drug delivery system: Elucidation of mechanism of uptake using chylomicron flow blocking approach. Int J Pharm [Internet]. Elsevier BV; 2015;495:439–46. Available from: https://doi.org/10.1016/j.ijpharm.2015.09.014
Javan F, Vatanara A, Azadmanesh K, Nabi-Meibodi M, Shakouri M. Encapsulation of ritonavir in solid lipid nanoparticles: in-vitro anti-HIV-1 activity using lentiviral particles. J Pharm Pharmacol. 2017;69:1002–9.
Kumar S, Narayan R, Ahammed V, Nayak Y, Naha A, Nayak UY. Development of ritonavir solid lipid nanoparticles by Box Behnken design for intestinal lymphatic targeting. J Drug Deliv Sci Technol [Internet]. Elsevier BV; 2018;44:181–9. Available from: https://doi.org/10.1016/j.jddst.2017.12.014
Karakucuk A, Celebi N, Teksin ZS. Preparation of ritonavir nanosuspensions by microfluidization using polymeric stabilizers: I. A Design of Experiment approach. Eur J Pharm Sci [Internet]. Elsevier BV; 2016;95:111–21. Available from: https://doi.org/10.1016/j.ejps.2016.05.010
Sinha S, Ali M, Baboota S, Ahuja A, Kumar A, Ali J. Solid dispersion as an approach for bioavailability enhancement of poorly water-soluble drug ritonavir. AAPS PharmSciTech. 2010;11:518–27.
Dhore PW, Dave VS, Saoji SD, Bobde YS, Mack C, Raut NA. Enhancement of the aqueous solubility and permeability of a poorly water soluble drug ritonavir via lyophilized milk-based solid dispersions. Pharm Dev Technol. 2017;22:90–102.
Maniyar MG, Kokare CR. Formulation and evaluation of spray dried liposomes of lopinavir for topical application. J Pharm Investig. 2019;49:259–70. https://doi.org/10.1007/s40005-018-0403-7.
Gairetta M, Bianchin MD, Kanis LA, Contri RV, Külkamp-Guerreiro IC. Development of innovative polymer-based matricial nanostructures for ritonavir oral administration. J Nanomater. 2019.
Yogananda R, Chowdary KPR. Enhancement of solubility, dissolution rate and bioavailability of efavirenz by Cyclodextrins and Solutol HS15 - a factorial study. Int J Res Pharm Chem. 2013;3:235–9.
Shete H, Patravale V. Long chain lipid based tamoxifen NLC. Part I: Preformulation studies, formulation development and physicochemical characterization. Int J Pharm [Internet]. Elsevier BV; 2013;454:573–83. Available from: https://doi.org/10.1016/j.ijpharm.2013.03.034
Patil-Gadhe A, Pokharkar V. Montelukast-loaded nanostructured lipid carriers : Part I Oral bioavailability improvement. Eur J Pharm Biopharm [Internet]. Elsevier BV; 2014;88:160–8. Available from: https://doi.org/10.1016/j.ejpb.2014.05.019
Bhise J, Kashaw SK, Sau S, Iyer AK. Nanostructured lipid carriers employing polyphenols as promising anticancer agents: Quality by design (QbD) approach. Elsevier BV; 2017;506–15. https://doi.org/10.1016/j.ijpharm.2017.04.078.
Mendes AI, Silva AC, Catita JAM, Cerqueira F, Gabriel C, Lopes CM. Miconazole-loaded nanostructured lipid carriers (NLC) for local delivery to the oral mucosa : Improving antifungal activity. Colloids Surfaces B Biointerfaces [Internet]. Elsevier BV; 2013;111:755–63. Available from: https://doi.org/10.1016/j.colsurfb.2013.05.041
Chen CC, Tsai TH, Huang ZR, Fang JY. Effects of lipophilic emulsifiers on the oral administration of lovastatin from nanostructured lipid carriers : Physicochemical characterization and pharmacokinetics. Eur J Pharm Biopharm [Internet]. Elsevier BV; 2010;74:474–82. Available from: https://doi.org/10.1016/j.ejpb.2009.12.008
Geszke-Moritz M, Moritz M. Solid lipid nanoparticles as attractive drug vehicles: Composition, properties and therapeutic strategies. Mater Sci Eng C [Internet]. Elsevier BV.; 2016; Available from: https://doi.org/10.1016/j.msec.2016.05.119
Bolhassani A, Javanzad S, Saleh T, Hashemi M, Aghasadeghi MR, Sadat SM. Polymeric nanoparticles Potent vectors for vaccine delivery targeting cancer and infectious diseases. Hum Vaccines Immunother. 2014;10:321–32.
Lembo D, Donalisio M, Civra A, Argenziano M, Cavalli R. Nanomedicine formulations for the delivery of antiviral drugs: a promising solution for the treatment of viral infections [Internet]. Expert Opin Drug Deliv. Taylor & Francis; 2018;93–114. Available from: https://doi.org/10.1080/17425247.2017.1360863
Kumbhar DD, Pokharkar VB. Engineering of a nanostructured lipid carrier for the poorly water-soluble drug , bicalutamide : Physicochemical investigations. Colloids Surfaces A Physicochem Eng Asp [Internet]. Elsevier BV; 2013;416:32–42. Available from: https://doi.org/10.1016/j.colsurfa.2012.10.031
Gurumukhi VC, Bari SB. Fabrication of efavirenz loaded nano-formulation using quality by design (QbD) based approach: Exploring characterizations and in vivo safety. J Drug Deliv Sci Technol [Internet]. Elsevier; 2020;56:101545. Available from: https://doi.org/10.1016/j.jddst.2020.101545
Patil SS, Kumbhar DD, Manwar JV, Jadhao RG, Bakal RL, Wakode S. Ultrasound-Assisted Facile Synthesis of Nanostructured Hybrid Vesicle for the Nasal Delivery of Indomethacin: Response Surface Optimization, Microstructure, and Stability. AAPS PharmSciTech. 2019;20:3–17.
Behbahani ES, Ghaedi M, Abbaspour M, Rostamizadeh K. Optimization and characterization of ultrasound assisted preparation of curcumin-loaded solid lipid nanoparticles: Application of central composite design, thermal analysis and X-ray diffraction techniques. Ultrason Sonochem [Internet]. 2017;38:271–80. Available from: https://doi.org/10.1016/j.ultsonch.2017.03.013
Gurumukhi VC, Bari SB. Quality by design (QbD)–based fabrication of atazanavir-loaded nanostructured lipid carriers for lymph targeting: bioavailability enhancement using chylomicron flow block model and toxicity studies. Drug Deliv Transl Res [Internet]. Springer US; 2021; Available from: https://doi.org/10.1007/s13346-021-01014-4
Hiremath PS, Soppimath KS, Betageri GV. Proliposomes of exemestane for improved oral delivery: Formulation and in vitro evaluation using PAMPA, Caco-2 and rat intestine. Int J Pharm. 2009;380:96–104.
Chiranjeevi K, Channabasavaraj KP, Srinivas Reddy PTN. Development and Validation of Spectrophotometric Method for Quantitative estimation of Ritonavir in Bulk and Pharmaceutical Dosage Forms. Int J ChemTech Res. 2011;3:58–62.
ICH guideline Q2(R1). ICH harmonised tripartite guideline, validation of analytical procedures: text and methodology. Curr. Step 4 version Parent Guidel 1994. 2005;13.
Pallagi E, Ambrus R, Szabó-Révész P, Csóka I. Adaptation of the quality by design concept in early pharmaceutical development of an intranasal nanosized formulation. Int J Pharm [Internet]. Elsevier BV; 2015;491:384–92. Available from: https://doi.org/10.1016/j.ijpharm.2015.06.018
Shekhawat P, Pokharkar V. Risk assessment and QbD based optimization of an Eprosartan mesylate nanosuspension: In-vitro characterization, PAMPA and in-vivo assessment. Int J Pharm [Internet]. Elsevier; 2019;567:118415. Available from: https://doi.org/10.1016/j.ijpharm.2019.06.006
Simões LS, Araújo JF, Vicente AA, Ramos OL. Design of β-lactoglobulin micro- and nanostructures by controlling gelation through physical variables. Food Hydrocoll [Internet]. Elsevier Ltd; 2020;100:105357. Available from: https://doi.org/10.1016/j.foodhyd.2019.105357
Ngan CL, Basri M, Lye FF, Fard Masoumi HR, Tripathy M, Abedi Karjiban R, et al. Comparison of Box-Behnken and central composite designs in optimization of fullerene loaded palm-based nano-emulsions for cosmeceutical application. Ind Crops Prod [Internet]. Elsevier B.V.; 2014;59:309–17. Available from: https://doi.org/10.1016/j.indcrop.2014.05.042
Ahmed S, Gull A, Alam M, Aqil M, Sultana Y. Ultrasonically tailored, chemically engineered and “QbD” enabled fabrication of agomelatine nanoemulsion; optimization, characterization, ex-vivo permeation and stability study. Ultrason Sonochem [Internet]. 2018;41:213–26. Available from: https://doi.org/10.1016/j.ultsonch.2017.09.042
Garg NK, Sharma G, Singh B, Nirbhavane P, Tyagi RK, Shukla R, et al. Quality by Design (QbD)-enabled development of aceclofenac loaded-nano structured lipid carriers (NLCs): An improved dermatokinetic profile for inflammatory disorder(s). Int J Pharm [Internet]. Elsevier BV; 2017;517:413–31. Available from: https://doi.org/10.1016/j.ijpharm.2016.12.010
Abdelwahed W, Degobert G, Stainmesse S, Fessi H. Freeze-drying of nanoparticles: Formulation, process and storage considerations. Adv Drug Deliv Rev. 2006;58:1688–713.
Zheng J, Gao Q. Tang C he, Ge G, Zhao M, Sun W. Heteroprotein complex formation of soy protein isolate and lactoferrin: Thermodynamic formation mechanism and morphologic structure. Food Hydrocoll. 2020;100.
Das S, Ng WK, Tan RBH. Are nanostructured lipid carriers (NLCs) better than solid lipid nanoparticles (SLNs): Development, characterizations and comparative evaluations of clotrimazole-loaded SLNs and NLCs? Eur J Pharm Sci [Internet]. Elsevier BV; 2012;47:139–51. Available from: https://doi.org/10.1016/j.ejps.2012.05.010
Park S, Baker JO, Himmel ME, Parilla PA, Johnson DK. Cellulose crystallinity index: Measurement techniques and their impact on interpreting cellulase performance. Biotechnol Biofuels. 2010;3:1–10.
Aji Alex MR, Chacko AJ, Jose S, Souto EB. Lopinavir loaded solid lipid nanoparticles (SLN) for intestinal lymphatic targeting. Eur J Pharm Sci. 2011;42:11–8.
Patil P, Khairnar G, Naik J. Preparation and statistical optimization of Losartan Potassium loaded nanoparticles using Box Behnken factorial design: Microreactor precipitation. Chem Eng Res Des [Internet]. Institution of Chemical Engineers; 2015;104:98–109. Available from: https://doi.org/10.1016/j.cherd.2015.07.021
Pardeshi C V., Rajput P V., Belgamwar VS, Tekade AR, Surana SJ. Novel surface modified solid lipid nanoparticles as intranasal carriers for ropinirole hydrochloride: application of factorial design approach. Drug Deliv [Internet]. 2013;20:47–56. Available from: https://doi.org/10.3109/10717544.2012.752421
Jores K, Mehnert W, Mäder K. Physicochemical investigations on solid lipid nanoparticles and on oil-loaded solid lipid nanoparticles: A nuclear magnetic resonance and electron spin resonance study. Pharm Res. 2003;20:1274–83.
Choi JU, Maharjan R, Pangeni R, Jha SK, Lee NK, Kweon S, et al. Modulating tumor immunity by metronomic dosing of oxaliplatin incorporated in multiple oral nanoemulsion. J Control Release [Internet]. Elsevier; 2020;322:13–30. Available from: https://doi.org/10.1016/j.jconrel.2020.03.012
Tiwari R, Pathak K. Nanostructured lipid carrier versus solid lipid nanoparticles of simvastatin: Comparative analysis of characteristics, pharmacokinetics and tissue uptake. Int J Pharm [Internet]. Elsevier B.V.; 2011;415:232–43. Available from: https://doi.org/10.1016/j.ijpharm.2011.05.044
Pardeshi C V., Belgamwar VS. Improved brain pharmacokinetics following intranasal administration of N,N,N-trimethyl chitosan tailored mucoadhesive NLCs. Mater Technol [Internet]. Taylor & Francis; 2020;35:249–66. Available from: https://doi.org/10.1080/10667857.2019.1674522
Kumar R, Singh A, Garg N, Siril PF. Solid lipid nanoparticles for the controlled delivery of poorly water soluble non-steroidal anti-inflammatory drugs. Ultrason Sonochem [Internet]. 2018;40:686–96. Available from: https://doi.org/10.1016/j.ultsonch.2017.08.018
Tefas LR, Rus LM, Achim M, Vlase L, Tomuță I. Application of the quality by design concept in the development of quercetin-loaded polymeric nanoparticles. Farmacia. 2018;66:798–810.
Üstündaǧ-Okur N, Gökçe EH, Bozbiyik DI, Eǧrilmez S, Özer Ö, Ertan G. Preparation and in vitro-in vivo evaluation of ofloxacin loaded ophthalmic nano structured lipid carriers modified with chitosan oligosaccharide lactate for the treatment of bacterial keratitis. Eur J Pharm Sci. 2014;63:204–15.
Shah B, Khunt D, Bhatt H, Misra M, Padh H. Application of quality by design approach for intranasal delivery of rivastigmine loaded solid lipid nanoparticles : Effect on formulation and characterization parameters. Eur J Pharm Sci [Internet]. Elsevier B.V.; 2015;78:54–66. Available from: https://doi.org/10.1016/j.ejps.2015.07.002
Aburahma MH, Badr-Eldin SM. Compritol 888 ATO: a multifunctional lipid excipient in drug delivery systems and nanopharmaceuticals. Expert Opin Drug Deliv. 2014;11:1865–83.
Kumbhar DD, Pokharkar VB. Physicochemical investigations on an engineered lipid – polymer hybrid nanoparticle containing a model hydrophilic active, zidovudine. Colloids Surfaces A Physicochem Eng Asp [Internet]. Elsevier BV; 2013;436:714–25. Available from: https://doi.org/10.1016/j.colsurfa.2013.07.044
Tran TTD, Tran PHL, Nguyen MNU, Tran KTM, Pham MN, Tran PC, et al. Amorphous isradipine nanosuspension by the sonoprecipitation method. Int J Pharm [Internet]. Elsevier BV; 2014;474:146–50. Available from: https://doi.org/10.1016/j.ijpharm.2014.08.017
Khosa A, Reddi S, Saha RN. Nanostructured lipid carriers for site-specific drug delivery. Biomed Pharmacother [Internet]. Elsevier; 2018;103:598–613. Available from: https://doi.org/10.1016/j.biopha.2018.04.055
Singh H, Jindal S, Singh M, Sharma G, Kaur IP. Nano-formulation of rifampicin with enhanced bioavailability: Development, characterization and in-vivo safety. Int J Pharm [Internet]. Elsevier BV; 2015;485:138–51. Available from: https://doi.org/10.1016/j.ijpharm.2015.02.050
Neupane YR, Srivastava M, Ahmad N, Kumar N, Bhatnagar A, Kohli K, Kanchan A. Lipid based nanocarrier system for the potential oral delivery of decitabine: Formulation design, characterization, ex vivo, and in vivo assessment. Int J Pharm [Internet]. Elsevier BV; 2014;477:601–12. Available from: https://doi.org/10.1016/j.ijpharm.2014.11.001
Wang Y, Zheng Y, Zhang L, Wang Q, Zhang D. Stability of nanosuspensions in drug delivery [Internet]. J Control Release. Elsevier BV; 2013;1126–41. Available from: https://doi.org/10.1016/j.jconrel.2013.08.006
Tan SW, Billa N, Roberts CR, Burley JC. Surfactant effects on the physical characteristics of Amphotericin B-containing nanostructured lipid carriers. Colloids Surfaces A Physicochem Eng Asp [Internet]. Elsevier BV; 2010;372:73–9. Available from: https://doi.org/10.1016/j.colsurfa.2010.09.030
Kim JK, Park JS, Kim CK. Development of a binary lipid nanoparticles formulation of itraconazole for parenteral administration and controlled release. Int J Pharm. 2010;383:209–15.
Qiao H, Chen L, Rui T, Wang J, Chen T, Fu T, et al. Fabrication and in vitro/in vivo evaluation of amorphous andrographolide nanosuspensions stabilized by D-α-tocopheryl polyethylene glycol 1000 succinate/sodium lauryl sulfate. Int J Nanomedicine. 2017;12:1033–46.
Zeng C, Zheng R, Yang X, Du Y, Xing J, Lan W. Improved oral delivery of tilianin through lipid–polymer hybrid nanoparticles to enhance bioavailability. Biochem Biophys Res Commun [Internet]. Elsevier Ltd; 2019;519:316–22. Available from: https://doi.org/10.1016/j.bbrc.2019.09.004
Veszelka S, Bocsik A, Walter FR, Hantosi D, Deli MA. Blood-brain barrier co-culture models to study nanoparticle penetration: Focus on co-culture systems. Acta Biol Szeged. 2015;59:157–68.
Dangre P, Gilhotra R, Dhole S. Formulation and statistical optimization of self-microemulsifying drug delivery system of eprosartan mesylate for improvement of oral bioavailability. Drug Deliv Transl Res. 2016;6:610–21. https://doi.org/10.1007/s13346-016-0318-7.
Acknowledgements
The authors would like to acknowledge the support of Management, Principal, and my colleagues of R. C. Patel Institute of Pharmaceutical Education and Research (RCPIPER), Shirpur.
Author information
Authors and Affiliations
Contributions
All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All animal experiments were carried out according to the NIH publication No. 8023, revised 1923 guidelines for Care and Use of Laboratory animals.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gurumukhi, V.C., Bari, S.B. Development of ritonavir-loaded nanostructured lipid carriers employing quality by design (QbD) as a tool: characterizations, permeability, and bioavailability studies. Drug Deliv. and Transl. Res. 12, 1753–1773 (2022). https://doi.org/10.1007/s13346-021-01083-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-021-01083-5