Abstract
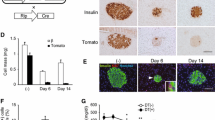
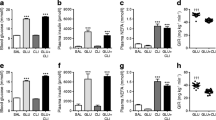
Gamma-aminobutyric acid (GABA) is an inhibitory neurotransmitter that is strongly and selectively synthesized in and secreted from pancreatic beta cells. Exogenously delivered GABA has been proposed to induce beta cell regeneration in type 1 diabetes, but these results have been difficult to replicate and may depend on the specifics of the animal model and drug delivery method used. Here, we developed a GABA-releasing ethylene-vinyl acetate polymer implant for sustained GABA delivery to the intraperitoneal space as an alternative to injected or oral GABA. We explored the effect of the GABA-releasing polymer implants compared to implanted osmotic pumps loaded with GABA on islet size in non-diabetic, outbred mice. We also attempted to monitor in vivo GABA release using HPLC on blood samples, but these measurements were confounded by high variability within treatment groups and unexpectedly high serum GABA levels in mice receiving GABA-negative implants. The ethylene-vinyl acetate polymer implants became heavily fibrosed with abdominal adhesion tissue, while the osmotic pumps had no macroscopic fibrosis. Histological analysis showed no significant effect of the sustained GABA delivery polymer or osmotic pumps on islet size, alpha cell to beta cell ratio, or the number of Ki67-positive islet cells. The GABA treatment time course was limited to two weeks due to the drug-release window of the polymer, while others reported islet-trophic effects of GABA after 10 to 12 weeks of treatment. In summary, our study is consistent with the concept that exogenous GABA administration does not significantly alter islet cell mass in non-diabetic CD-1 mice in the short-term. However, more data are needed including higher GABA doses and more prolonged treatment regimens for a better comparison with contrasting reports.
Graphical abstract






Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Leelarathna L, Guzder R, Muralidhara K, Evans ML. Diabetes: glycaemic control in type 1. BMJ Clin. Evid. 2011.
Wilcox G. Insulin and insulin resistance. Clin Biochem Rev. 2005;26(2):19–39.
Cersosimo E, Solis-Herrera C, Trautmann ME, Malloy J, Triplitt CL. Assessment of pancreatic β-cell function: review of methods and clinical applications. Curr Diabetes Rev. 2014;10(1):2–42.
Pathak V, Pathak NM, O’Neill CL, Guduric-Fuchs J, Medina RJ. Therapies for type 1 diabetes: current scenario and future perspectives. Clin. Med. Insights Endocrinol. Diabetes. 2019;12. https://doi.org/10.1177/1179551419844521.
Dunn TB, Kirchner V, Bellin MD. Beta-cell replacement therapy: current outcomes and future landscape. Curr Opin Organ Transplant. 2015;20(6):681–90. https://doi.org/10.1097/MOT.0000000000000245.
Atkinson MA, Eisenbarth GS, Michels AW. Type 1 diabetes. Lancet. 2014;383(9911):69–82. https://doi.org/10.1016/S0140-6736(13)60591-7.
Franklin IK, Wollheim CB. GABA in the endocrine pancreas. J Gen Physiol. 2004;123(3):185–90. https://doi.org/10.1085/jgp.200409016.
Wan Y, Wang Q, Prud’homme GJ. GABAergic system in the endocrine pancreas: a new target for diabetes treatment. Diabetes Metab Syndr Obes Targets Ther. 2015;8:79–87. https://doi.org/10.2147/DMSO.S50642.
Hagan DW, et al. Mechanism and effects of pulsatile GABA secretion from cytosolic pools in the human beta cell. Nat Metab. 2019;1(11):1110–26. https://doi.org/10.1038/s42255-019-0135-7.
Feng AL, Xiang YY, Gui L, Kaltsidis G, Feng Q, Lu WY. Paracrine GABA and insulin regulate pancreatic alpha cell proliferation in a mouse model of type 1 diabetes. Diabetologia. 2017;60(6):1033–42. https://doi.org/10.1007/s00125-017-4239-x.
Tian J, Dang H, Nguyen AV, Chen Z, Kaufman DL. Combined therapy with GABA and proinsulin/alum acts synergistically to restore long-term normoglycemia by modulating T-cell autoimmunity and promoting β-cell replication in newly diabetic NOD mice. Diabetes. 2014;63(9):3128–34. https://doi.org/10.2337/db13-1385.
Soltani N, et al. GABA exerts protective and regenerative effects on islet beta cells and reverses diabetes. Proc Natl Acad Sci U S A. 2011;108(28):11692–7. https://doi.org/10.1073/pnas.1102715108.
Purwana I, et al. GABA promotes human β-cell proliferation and modulates glucose homeostasis. Diabetes. 2014;63(12):4197–205. https://doi.org/10.2337/db14-0153.
Prud’homme GJ, Glinka Y, Hasilo C, Paraskevas S, Li X, Wang Q. GABA protects human islet cells against the deleterious effects of immunosuppressive drugs and exerts immunoinhibitory effects alone. Transplantation. 2013;96(7):616. https://doi.org/10.1097/TP.0b013e31829c24be.
Bhandage AK, et al. GABA regulates release of inflammatory cytokines from peripheral blood mononuclear cells and CD4+ T cells and is immunosuppressive in type 1 diabetes. EBioMedicine. 2018;30:283–94. https://doi.org/10.1016/j.ebiom.2018.03.019.
Weir GC, Bonner-Weir S. GABA signaling stimulates β cell regeneration in diabetic mice. Cell. 2017;168(1):7–9. https://doi.org/10.1016/j.cell.2016.12.006.
Ben-Othman N, et al. Long-term GABA administration induces alpha cell-mediated beta-like cell neogenesis. Cell. 2017;168(1):73-85.e11. https://doi.org/10.1016/j.cell.2016.11.002.
He S, et al. Rapamycin/GABA combination treatment ameliorates diabetes in NOD mice. Mol Immunol. 2016;73:130–7. https://doi.org/10.1016/j.molimm.2016.01.008.
Quoix N, Cheng-Xue R, Guiot Y, Herrera PL, Henquin J-C, Gilon P. The GluCre-ROSA26EYFP mouse: a new model for easy identification of living pancreatic α-cells. FEBS Lett. 2007;581(22):4235–40. https://doi.org/10.1016/j.febslet.2007.07.068.
Li Z, et al. Artesunate prevents type 1 diabetes in NOD mice mainly by inducing protective IL-4-producing T cells and regulatory T cells, FASEB. J Off Publ Fed Am Soc Exp Biol. 2019;33(7):8241–8. https://doi.org/10.1096/fj.201900146R.
Li J, et al. Artemisinins target GABAA receptor signaling and impair α cell identity. Cell. 2017;168(1):86-100.e15. https://doi.org/10.1016/j.cell.2016.11.010.
Collombat P, et al. The ectopic expression of Pax4 in the mouse pancreas converts progenitor cells into α- and subsequently β-cells. Cell. 2009;138(3):449–62. https://doi.org/10.1016/j.cell.2009.05.035.
Courtney M, et al. The inactivation of Arx in pancreatic α-cells triggers their neogenesis and conversion into functional β-like cells. PLoS Genet. 2013;9(10). https://doi.org/10.1371/journal.pgen.1003934.
van der Meulen T, et al. Artemether does not turn α cells into β cells. Cell Metab. 2018;27(1):218-225.e4. https://doi.org/10.1016/j.cmet.2017.10.002.
Ackermann AM, Moss NG, Kaestner KH. GABA and artesunate do not induce pancreatic α-to-β cell transdifferentiation in vivo. Cell Metab. 2018;28(5):787-792.e3. https://doi.org/10.1016/j.cmet.2018.07.002.
Untereiner A, et al. GABA promotes β-cell proliferation, but does not overcome impaired glucose homeostasis associated with diet-induced obesity. FASEB J. 2018;33(3):3968–84. https://doi.org/10.1096/fj.201801397R.
Yang NJ, Hinner MJ. Getting across the cell membrane: an overview for small molecules, peptides, and proteins. Methods Mol Biol Clifton NJ. 2015;1266:29–53. https://doi.org/10.1007/978-1-4939-2272-7_3.
Silberstein GB, Daniel CW. Elvax 40P implants: sustained, local release of bioactive molecules influencing mammary ductal development. Dev Biol. 1982;93(1):272–8. https://doi.org/10.1016/0012-1606(82)90259-7.
Almeida A, et al. Sustained release from hot-melt extruded matrices based on ethylene vinyl acetate and polyethylene oxide. Eur J Pharm Biopharm. 2012;82(3):526–33. https://doi.org/10.1016/j.ejpb.2012.08.008.
Fiorani L, Maccarone R, Fernando N, Colecchi L, Bisti S, Valter K. Slow-release drug delivery through Elvax 40W to the rat retina: implications for the treatment of chronic conditions. J Vis Exp JoVE 2014;(91). https://doi.org/10.3791/51563.
Genina N, Holländer J, Jukarainen H, Mäkilä E, Salonen J, Sandler N. Ethylene vinyl acetate (EVA) as a new drug carrier for 3D printed medical drug delivery devices. Eur J Pharm Sci. 2016;90:53–63. https://doi.org/10.1016/j.ejps.2015.11.005.
Schneider C, Langer R, Loveday D, Hair D. Applications of ethylene vinyl acetate copolymers (EVA) in drug delivery systems. J Controlled Release. 2017;262:284–95. https://doi.org/10.1016/j.jconrel.2017.08.004.
Okabe K, Kimura H, Okabe J, Kato A, Kunou N, Ogura Y. Intraocular tissue distribution of betamethasone after intrascleral administration using a non-biodegradable sustained drug delivery device. Invest Ophthalmol Vis Sci. 2003;44(6):2702–7. https://doi.org/10.1167/iovs.02-0956.
The Dow Chemical Company, ELVAXTM 40W ethylene vinyl acetate copolymer technical data sheet. The Dow Chemical Company, 2019, [Online]. Available: https://www.dow.com/en-us/document-viewer.html?ramdomVar=2253541954360773183&docPath=/content/dam/dcc/documents/en-us/productdatasheet/914/914-20801-01-elvax-40w-ethylene-vinyl-acetate-copolymer-tds.pdf.
Tallury P, Alimohammadi N, Kalachandra S. Poly(ethylene-co-vinyl acetate) copolymer matrix for delivery of chlorhexidine and acyclovir drugs for use in the oral environment: effect of drug combination, copolymer composition and coating on the drug release rate. Dent Mater Off Publ Acad Dent Mater. 2007;23(4):404–9. https://doi.org/10.1016/j.dental.2006.02.011.
Fu Y, Kao WJ. Drug release kinetics and transport mechanisms of non-degradable and degradable polymeric delivery systems. Expert Opin Drug Deliv. 2010;7(4):429–44. https://doi.org/10.1517/17425241003602259.
DURECT Corporation, Alzet MICRO-OSMOTIC PUMP MODEL 1002. DURECT Corporation, Oct. 2016, Accessed: Apr. 20, 2020. [Online]. Available: https://www.alzet.com/wp-content/uploads/2019/06/Specs-1002.pdf.
Thammacharoen S, Kitchanukitwattana P, Suwanapaporn P, Chaiyabutr N. Effects of hindbrain infusion of an estrogen receptor antagonist on estrogenic modulation of eating behavior. Neurophysiology. 2017;49(1):72–7. https://doi.org/10.1007/s11062-017-9631-0.
Zhidkov N, De Souza R, Ghassemi AH, Allen C, Piquette-Miller M. Continuous intraperitoneal carboplatin delivery for the treatment of late-stage ovarian cancer. Mol Pharm. 2013;10(9):3315–22. https://doi.org/10.1021/mp400345h.
Hippocampal MicroRNA-124 enhances chronic stress resilience in mice| Journal of Neuroscience. https://www.jneurosci.org/content/36/27/7253 (accessed Apr. 20, 2020).
Zandy SL , Doherty JM, Wibisono ND, Gonzales RA. High sensitivity HPLC method for analysis of in vivo extracellular GABA using optimized fluorescence parameters for o-phthalaldehyde (OPA)/sulfite derivatives. J Chromatogr B Analyt Technol Biomed Life Sci. 2017;15:1055–1056, p. 1–7. https://doi.org/10.1016/j.jchromb.2017.04.003.
“CD-1®IGS Mice.” Charles River Laboratories International, Inc, 2011, [Online]. Available: https://www.criver.com/sites/default/files/resources/CD-1IGSMouseModelInformationSheet.pdf.
Hickman DL, Johnson J, Vemulapalli TH, Crisler JR, Shepherd R. Commonly used animal models. Princ Anim Res Grad Undergrad Stud. 2017; p. 117–175. https://doi.org/10.1016/B978-0-12-802151-4.00007-4.
Tuttle AH, Philip VM, Chesler EJ, Mogil JS. Comparing phenotypic variation between inbred and outbred mice. Nat Methods 2018;15(12). https://doi.org/10.1038/s41592-018-0224-7.
Kilimnik G, Jo J, Periwal V, Zielinski MC, Hara M. Quantification of islet size and architecture. Islets. 2012;4(2):167–72. https://doi.org/10.4161/isl.19256.
Kim A, Miller K, Jo J, Kilimnik G, Wojcik P, Hara M. Islet architecture. Islets. 2009;1(2):129–36. https://doi.org/10.4161/isl.1.2.9480.
Siepmann J, Peppas NA. Higuchi equation: derivation, applications, use and misuse. Int J Pharm. 2011;418(1):6–12. https://doi.org/10.1016/j.ijpharm.2011.03.051.
Brophy MR, Deasy PB. Application of the Higuchi model for drug release from dispersed matrices to particles of general shape. Int J Pharm. 1987;37(1):41–7. https://doi.org/10.1016/0378-5173(87)90008-1.
Shin SC, Byun SY. Controlled release of ethinylestradiol from ethylene-vinyl acetate membrane. Int J Pharm. 1996;137(1):95–102. https://doi.org/10.1016/0378-5173(96)04497-3.
Higuchi T. Rate of release of medicaments from ointment bases containing drugs in suspension. J Pharm Sci. 1961;50(10):874–5. https://doi.org/10.1002/jps.2600501018.
Mircioiu C, et al. Mathematical modeling of release kinetics from supramolecular drug delivery systems. Pharmaceutics. 2019;11(3). https://doi.org/10.3390/pharmaceutics11030140.
Baskin DG. A historical perspective on the identification of cell types in pancreatic islets of langerhans by staining and histochemical techniques. J Histochem Cytochem. 2015;63(8):543–58. https://doi.org/10.1369/0022155415589119.
Graefe C, et al. Optimized Ki-67 staining in murine cells: a tool to determine cell proliferation. Mol Biol Rep. 2019;46(4):4631–43. https://doi.org/10.1007/s11033-019-04851-2.
Upton G, Cook I. “ANOVA,” in A dictionary of statistics, Oxford University Press, 2008.
Kim A, Miller K, Jo J, Kilimnik G, Wojcik P, Hara M. Islet architecture: a comparative study. Islets. 2009;1(2):129–36. https://doi.org/10.4161/isl.1.2.9480.
Acknowledgments
We thank the Molecular Pathology Core, Department of Pathology, Immunology and Laboratory Medicine, University of Florida College of Medicine, for tissue processing and staining service.
Funding
Funding support was provided by the intramural funding programs of the J. Crayton Pruitt Family Department of Biomedical Engineering and the Herbert Wertheim College of Engineering at the University of Florida (E.P.) and by the University of Florida Center for Undergraduate Research, University Scholars Program (K.L.).
Author information
Authors and Affiliations
Contributions
Data collection and analysis were performed by Kevin C. Ling, D. Walker Hagan, Jorge Santini-González, and Edward A. Phelps. The manuscript was written by Kevin C. Ling and Edward A. Phelps. All authors read and commented on previous versions of the manuscript and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Ethical approval
The study did not involve human subjects. All institutional and national guidelines for the care and use of laboratory animals were followed.
Consent for publication
All authors read and approved the final manuscript.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ling, K.C., Hagan, D.W., Santini-González, J. et al. Effects of sustained GABA releasing implants on pancreatic islets in mice. Drug Deliv. and Transl. Res. 11, 2198–2208 (2021). https://doi.org/10.1007/s13346-020-00886-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-020-00886-2