Abstract
Background and Objective
Vancomycin is often used in the ICU for the treatment of Gram-positive bacterial infection. In critically ill children, there are pathophysiologic changes that affect the pharmacokinetics of vancomycin. A systematic review of vancomycin pharmacokinetics and pharmacodynamics in critically ill children was performed.
Methods
Pharmacokinetic studies of vancomycin in critically ill children published up to May 2021 were included in the review provided they included children aged > 1 month. Studies including neonates were excluded. A search was performed using the PubMed, Scopus, and Google Scholar databases. The Risk of Bias Assessment Tool for Systematic Reviews (ROBIS) was used to check for quality and reduce bias. Data on study characteristics, patient demographics, clinical parameters, pharmacokinetic parameters, outcomes, and study limitations were collected.
Results
Thirteen studies were included in this review. A wide variety of dosing and sampling strategies were used in the studies. Methods for estimating vancomycin pharmacokinetics, especially the area under the curve over 24 h, varied. Vancomycin doses of 20–60 mg/kg were given daily. This resulted in high variability in pharmacokinetic parameters. Vancomycin trough level was less than 15 μg/mL in most of the studies. Vancomycin clearance ranged from 0.05 to 0.38 L/h/kg. Volume of distribution ranged from 0.1 to 1.16 L/kg. Half-life was between 2.4 and 23.6 h. Patients in the study receiving continuous vancomycin infusion had AUC24 < 400 µg·h/mL.
Conclusion
There is large variability in the pharmacokinetics of vancomycin among critically ill patients. Studies to assess the factors responsible for this variability in vancomycin pharmacokinetics are needed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
There was a lack of standardized methods for dosing, sampling and calculation of vancomycin pharmacokinetic parameters. |
Large differences were observed in vancomycin pharmacokinetics in critically ill children. |
Some variability in vancomycin pharmacokinetics has been attributed to age, weight, renal function, protein binding, temperature, fluid balance and concomitant medication. |
Exploration of other factors that may affect vancomycin pharmacokinetics is required. |
1 Introduction
Vancomycin is a tricyclic glycopeptide antibiotic that has been used extensively in adult [1, 2] and paediatric [3, 4] intensive care unit patients for the treatment of infections with Gram-positive organisms such as methicillin-resistant Staphylococcus aureus (MRSA) and penicillin-resistant Streptococcus pneumoniae. It is also frequently used in the context of hospital-acquired infections (including ventilator-associated pneumonia and wound sepsis) while awaiting the identification of specific microorganisms. As it is poorly absorbed from the gut, it is usually administered intravenously, except in situations such as a Clostridium difficile colitis, where it is administered orally (with the intention of limiting its effects to within the gut). The agent has also been used in patients who are allergic to penicillin and cephalosporins [1, 2]
With concerns about increased resistance of bacteria to vancomycin, guidelines have recommended higher therapeutic levels of vancomycin [5]. Specifically, the area under the 24-h concentration-time curve (AUC24), related to the minimal inhibitory concentration (MIC) of the bacteria being treated, has been highlighted as the most accurate reflection of the likelihood of bacterial eradication in a particular patient on vancomycin (for Staphylococcus aureus) [6, 7]. Unfortunately, the MIC is not generally available for microbial isolates, or for presumed pathogens in the clinical setting. Likewise, the AUC24 is generally not available, so trough levels of antibiotics are often used as proxy measures of AUC24 despite the fact that this may be inaccurate [8]. There are concerns about how best to monitor vancomycin clinically [6].
Early forms of vancomycin were associated with severe toxicity related to contaminants in formulations of the drugs, but since that time the toxicity has been substantially modified with new preparations. However, there are concerns that guidelines with high AUC24 or trough levels of vancomycin may be associated with increased toxicity (particularly nephrotoxicity) [9,10,11], so it is important to revisit the toxicity of vancomycin and the adequacy of level monitoring in the clinical setting. In addition, therapeutic drug levels may be strongly affected by protein binding, and children (particularly young infants) may have low protein levels to bind the drug, while illness may also affect protein binding [12].
The paediatric population has a wide range of ages, sizes, and stages of development, which is associated with variations in the pharmacokinetics and pharmacodynamics of medications [13]. In addition, the paediatric critical care environment is characterized by a wide range of baseline pathologies, by rapid changes in clinical symptoms (with associated changes in organ function), by rapid changes in protein levels, and by aggressive therapies that may result in dramatic changes in drug distribution and morbidities [14]. It is likely that critically ill patients present effectively different compartmental models for drug distribution and elimination, which may have a profound effect on the calculations of parameters such as AUC24 using different software models [15, 16].
Relatively few studies have investigated the pharmacokinetics of vancomycin in critically ill children, which may relate to the complexity of doing clinical studies in this environment. There are a number of reviews that include the pharmacodynamics of vancomycin in critically ill children [13, 17]. One is a narrative review of anti-infective agents in critically ill children [13], while the other is a systematic review of anti-infective therapy in the paediatric intensive care unit (PICU) [17].
This study set out to systematically review studies of critically ill children in paediatric intensive care with regard to the pharmacokinetics of vancomycin.
2 Methods
2.1 Data Sources and Searches
The Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guideline [18] was used in the preparation of this systematic review (prospectively registered in PROSPERO with ID: CRD42021272930). Articles were sourced from the PubMed, Scopus, and Google Scholar databases. Studies conducted at any time up to 12 May 2021 were searched for. Potential articles were identified independently using variations of the keywords “Vancomycin” AND “Pharmacokinetics” AND “Children” AND “Paediatrics” AND “Critically-ill” AND “ICU” NOT “Adults” NOT “Neonates”.
2.2 Study Selection
Studies were included irrespective of time and language of publication. The selection criteria included studies (i) performed in children > 1 month old and less than 19 years of age who (ii) were treated with vancomycin, where (iii) the study was conducted in the ICU setting and (iv) presented results for primary vancomycin pharmacokinetic parameters, i.e. clearance (CL) and volume of distribution (Vd).
Our exclusion criteria included: (i) studies carried out in preterm infants and (ii) studies that included neonates or adults.
Primary outcomes extracted from the articles included (i) trough and peak concentrations, (ii) CL, (iii) Vd, (iii) half-life (t1/2) and (iv) area under the concentration-time curve (AUC). Secondary outcomes were the effects of demographic and clinical variables (such as kidney injury, augmented renal clearance, serum albumin and age) on vancomycin pharmacokinetics.
2.3 Quality Assessment
Each study was assessed to check for quality and to reduce bias. The Risk of Bias in Systematic Reviews (ROBIS) assessment tool [19] was used for evaluation. The risk of bias in the articles included in the review was classified in all domains. The outcome of the quality check using the ROBIS tool showed a low risk of bias in the systematic review.
2.4 Data Extraction
Data collected from each of the studies included authors’ names, year of publication, study population, study aim and objectives, study design and setting, sample size (number of patients and number of plasma samples analysed), age, weight, inclusion and exclusion criteria, patient subgroups, vancomycin dosing information, compartmental models, pharmacokinetic parameters assessed, clinical parameters, outcomes and limitations.
3 Results
3.1 Study Selection
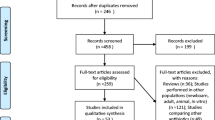
The search for relevant articles as predetermined by the search criteria yielded 652 potential articles. After following the steps in the PRISMA flow diagram, 54 full-text articles were read (see Fig. 1). After screening the 54 articles according to the inclusion and exclusion criteria, 13 articles were included in this study (Fig. 1).
3.2 Study Characteristics
3.2.1 Study Design
Nine studies included in our analysis were retrospective [20,21,22,23,24,25,26,27,28], three were prospective [3, 29, 30], while one study did not indicate the study design [31].
3.2.2 Sample size
Six studies analysed data from less than 30 children [3, 25, 26, 29,30,31], while one study included 250 children [23]. Six studies recruited between 30 and 100 patients [20,21,22, 24, 27, 28].
3.2.3 Clinical Characteristics of the Patients
While most studies included children with ages ranging from 0.1 to 17 years, one study only included infants aged 0.1–0.8 years [30] and another included children aged 1–21 years [23]. The weights of the children ranged from 2.6 to 88.3 kg.
Five studies were carried out in specific groups of children within the PICU, including patients following cardiac arrest [20], burns patients [29], patients on extracorporeal membrane oxygenation (ECMO) [25] and two studies of cancer patients [27, 28].
Most of the studies, with the exception of the studies of ECMO patients [26] and cancer patients [27], excluded patients with renal dysfunction. However, renal dysfunction was defined in different ways in the studies, including (i) creatinine > normal limit for age or/and estimated creatinine clearance <50 ml/min/1.73 m2 (calculated by the Schwartz formula) [22] and/or (ii) diagnosis of chronic kidney failure and/or use of renal replacement therapies and/or patients with a history (up to 2 weeks before monitoring) of an increase of more than 50% in creatinine over baseline [22, 27] or a twofold increase in baseline serum creatinine (SCr) [25] or according to plasma creatinine adjusted by age [31], a baseline serum creatinine of < 0.5 mg/dL in children aged 1 to < 2 years, < 1 mg/dL in children 2 to < 12 years old, < 1.3 mg/dL in children ≥ 12 years [23], or any degree of the paediatric RIFLE (Risk of Renal Dysfunction, Injury to the Kidney, Failure of Kidney Function, Loss of Kidney Function, and End-Stage Renal Disease) [26]. Table 1 summarizes these study characteristics.
The serum creatinine of the patients in the studies ranged between 8.84 and 65.42 μmol/L [20,21,22,23, 25,26,27,28, 30] (Table 1). Creatinine clearance (CLCR) was between 7.8 and 164.1 mL/min/1.73 m2 [3, 20, 21, 23, 25,26,27,28,29], with very low CLCR observed in the study by Zane et al. [20]. Patients with AKI and RRT in the study by Zylbersztajn et al. [26] had low CLCR compared to patients with only AKI.
Only three studies reported mean serum albumin concentrations: 5.20 ± 1.13, 3.91 ± 0.49 and 2.44 ± 0.34 g/L [21, 27, 28].
3.3 Dosing Regimens
Studies utilized a wide range of dosing strategies, including 5–15 mg/kg every 6 h [22, 24, 26, 29, 30] or every 6–12 h [21], 20 mg/kg every 8 h [3], continuous infusion (a 15 mg/kg loading dose followed by 45 mg/kg/day as continuous infusion [25]), or variable doses with 40–60 mg/kg/day [23], or 10–156 mg/kg/day [28], or 60 mg/kg/day given over variable dosing intervals ranging from 6 to 48 h [27]. Dose adjustments made according to the physician’s judgement were mentioned in four studies [22, 25, 26, 29]; these adjustments were made to achieve optimal plasma concentrations.
3.4 Blood Sampling/Sampling Procedure
Blood sampling was done after a steady state was achieved in eight studies [3, 22, 23, 26,27,28,29,30]. In those studies, the steady state was variously defined as after the third dose [22, 30], after the fourth dose [27,28,29], after 30 h [29], or after 48 h [3, 22, 26]. In one study, blood was collected after the first dose and after the third dose [20].
Peak concentration blood samples were typically collected 30 min to 1 h after vancomycin infusion [22, 24, 29, 30], and trough concentrations were collected not more than 30 min before the next vancomycin dose [22, 24, 26, 29, 30]. However, in two studies, vancomycin trough concentration blood samples were obtained 1 h before the next vancomycin administration [27, 28]. Avedissian et al. [23] collected peak concentration samples up to 2 h post-infusion and trough concentration samples up to 2 h before the next vancomycin dose.
Gomez et al. [29] collected blood samples 1, 2, 3 and 4 h after the fourth vancomycin dose, at 30 h, or at the time point equal to at least five times the drug half-life. They also collected trough concentrations immediately before the fifth dose. Mali et al. [3] collected the first blood samples 48 h after the first infusion (baseline), with other samples collected at 1, 1.25, 1.5, 2, 2.5, 3, and 6 h and at 72 h from baseline. Gous et al. [30] collected blood samples immediately before vancomycin infusion and at 30, 60, 120 and 300 min after infusion on days 3 and 9 after the first vancomycin infusion.
Analysis was performed using 30–658 plasma samples [20,21,22,23, 27,28,29]. The number of samples included in the pharmacokinetic analysis was not mentioned in six studies [3, 24,25,26, 30, 31]. One study included patients with only one serum trough concentration in the pharmacokinetic analysis [21].
3.5 Measurement of Vancomycin
A wide variety of different techniques were used to measure vancomycin levels, with somewhat variable characteristics (when reported). Inter-day and intra-day coefficients of variation of test results were 8.4 ± 7.4% and 4.8 ± 3.5%, respectively, in one study [29]; another study reported a daily deviation of < 15% [3]. The lower limits of detection reported by three studies were 0.2 μg/mL [23], 0.67 μg/mL [25] and 1.7 μg/mL [29], respectively. Two studies indicated upper limits of detection of 90 μg/mL [23] and 80 μg/mL [25], respectively.
3.6 Pharmacokinetic Modelling of Vancomycin
Six studies used compartmental modelling for the analysis of vancomycin pharmacokinetics [20, 21, 23,24,25, 30]. Two studies [23, 25] used the one-compartment model from Le et al. [32], while one study used a one-compartment model built by Wu and Furlanut [15]. Only Zane et al. [20] used a two-compartment model (with a proportional error model to describe the random residual variability) to describe the pharmacokinetics of vancomycin. Other studies used noncompartmental models [3, 22, 26, 27, 29].
3.7 Covariate Analysis in Models
Three studies included the effects of covariates (including weight, age, sex, SCr, renal function (CLCR), urea, albumin, temperature, use of concurrent nephrotoxic medications) on vancomycin pharmacokinetics in their models [20, 23, 27].
Zane et al. [20] included the allometric scaled weight (reference weight 70 kg) in their base model in order to take into account size-related changes in vancomycin clearance from childhood to adulthood. That study included patients on therapeutic hypothermia following cardiac resuscitation, hence the inclusion of temperatures below 37 °C (measured at the time of drug sampling). The goodness-of-fit of the model was improved by incorporating weight effects on CL, Q, V1 and V2. The effect of renal impairment on CL was also assessed. Glomerular filtration rate (GFR), calculated using height and serum creatinine (the Schwartz formula) at the time closest to the sampling time for vancomycin plasma concentration, was used for estimation. Patients were classified as having poor (≤ 30 mL/min/1.73 m2), reduced (31–89 mL/min/1.73 m2) or normal (90–140 mL/min/1.73 m2) renal function.
Avedissian et al. [23] included the effects of serum creatinine level and weight on CL and the effect of weight on Vd in their final model. Seixas et al. [27] included weight, serum level of vancomycin (> 15, 20, and 25 μg/mL), transplantation of haematopoietic stem cells, vancomycin doses exceeding 70 mg and 80 mg/kg/day, and use of diuretics and/or vasoactive drugs (dopamine, dobutamine, epinephrine, norepinephrine) and/or cyclosporine and/or tacrolimus in their model. The effect of a leukaemia diagnosis, haematopoietic stem cell transplantation, neutropenia, positive blood culture (any isolate), and failure to obtain AUC/MIC ≥ 400 were also assessed.
3.8 Pharmacokinetics of Vancomycin in Critically Ill Children
3.8.1 Vancomycin Serum Concentrations
Trough vancomycin concentrations were reported in 10 of the 13 studies (Table 2), and ranged from 9 to 15.6 μg/mL. Vancomycin trough concentrations were less than 15 μg/mL in 8 of the 10 studies [3, 21,22,23,24, 29,30,31], and only 2 studies reported a trough concentration of >15 μg/mL [27, 28]. In one study, trough concentrations measured after 48 h and 72 h [3] were comparable, as were trough concentrations after 24 h and 72 h [31] and after 72 h and 216 h [30]. Trough levels in children < 2 years and children ≥ 2 years were comparable [24].
Six studies measured the vancomycin peak concentration [22, 24, 25, 27, 28, 30]. The average (mean or median) peak concentration ranged from 21.67 to 35.5 μg/mL. Day 1 and day 3 peak concentrations were comparable in one study [31]. Another study showed a significantly higher peak concentration on day 9 compared to day 3 [30]. Children < 2 years and children ≥ 2 years had comparable peak concentrations [24].
3.8.2 Vancomycin Clearance
Vancomycin clearance ranged from 0.05 to 0.38 L/h/kg [3, 20, 21, 23,24,25,26, 28,29,30,31]. One study did not report CL [22]. Interindividual variability in vancomycin clearance was between 38 and 49.7% in three studies [20, 23, 25] and as high as 90.5% in one study [29].
Higher vancomycin clearance was observed in studies carried out by Zane et al. [20] (median (range): 0.34 (0.31–0.38) L/h/kg) and Sridharan et al. [21] (median (range): 0.34 (0.31–0.38) L/h/kg). Children < 2 years and children ≥ 2 years had comparable vancomycin clearance (median (range): 0.10 (0.06–0.18) L/h/kg vs. 0.10 (0.06–0.14) L/h/kg) [24]. Vancomycin clearance was similar on day 1 (mean ± SD: 0.12 ± 0.07 L/h/kg) and day 3 (mean ± SD: 0.15 ± 0.06 L/h/kg) of treatment in one study [31]. Another study showed similarity in vancomycin CL on day 3 (mean ± SD: 0.09 ± 0.03 L/h/kg) and day 9 (mean ± SD: 0.07 ± 0.02 L/h/kg) of treatment [30]. In the study of Zylbersztajn et al. [26], children without AKI or RRT had higher vancomycin clearance (median (range): 0.10 (0.06–0.10) L/h/kg) than children with AKI and RRT (median (range): 0.05 (0.02–0.06) L/h/kg) and children with AKI only (median (range): 0.07 (0.04–0.09) L/h/kg).
The intercompartmental distribution from the two-compartment model was 0.65 (0.53–0.77) L/h/kg [20].
3.8.3 Vancomycin Volume of Distribution
The average (mean or median) volume of distribution of vancomycin was between 0.1 and 1.16 L/kg [3, 20, 21, 23,24,25,26,27,28,29,30].
In a study comparing the Vd in children < 2 years and children ≥ 2 years, the Vd was similar in the two groups: median (range) 0.67 (0.39–1.15) and 0.62 (0.41–1.04) L/kg, respectively [24]. However, Sridharan et al. [21] evaluated the volume of distribution of vancomycin in 83 children by age group—they distinguished between infants (≤ 1 years), toddlers (> 1–2 years), children (> 2–5 years), and older children (> 5 years)—and found that infants had significantly higher Vd (0.14 L/kg) compared to children in other age groups (median Vd for toddlers: 0.09 L/kg; for children: 0.06 L/kg; for older children: 0.03 L/kg).
Children with AKI and RRT had a higher Vd (median (range): 1.16 (0.68–1.6) L/kg) than children without AKI or RRT (median (range): 0.73 (0.7–0.9) L/kg) and children with AKI only (median (range): 0.88 (0.68–0.92) L/kg) [26].
In the study by Giachetto et al. [31], Vd on day 3 (mean ± SD: 0.86 ± 0.58 L/kg) was higher than Vd on day 1 (mean ± SD: 0.51 ± 0.24 L/kg), and in the study by Gous et al. [30], the Vd on day 3 was almost double the Vd on day 9 (mean ± SD: 0.81 ± 0.6 L/kg vs. 0.44 ± 0.19 L/kg).
In the two-compartment model study [20], Vd in the peripheral compartment was 2.73 (2.24–3.22) L/kg, with the variability attributed to weight, serum creatinine, age, renal function, and temperature. In the same study, the inter-individual variability in the Vd of the central compartment was very high (136 (17.3–254.7) %).
High interindividual variability in Vd was observed in three other studies [23, 25, 29]. Two studies reported residual variabilities in Vd of 20.9% and 21.0%, respectively [20, 23].
3.8.4 Vancomycin Half-Life
The mean or median half-life of vancomycin in the studies ranged from 2.4 to 23.6 h [3, 21,22,23,24, 26, 29,30,31]. Children < 2 years and children ≥ 2 years had comparable half-lives (median (range): 3.6 (2.2–5.5) h and 3.8 (2.7–10.6) h, respectively) [24].
Compared to children without AKI or RRT and children with AKI only (median (range): 6.2 [4.9–8.06] h and 8.69 (5.05–17.52) h, respectively), vancomycin had a longer half-life (median (range): 23.6 (16.2–31) h) in children with AKI and RRT [26]. In the study by Gous et al. [30], the half-life on day 3 was higher than the half-life on day 9 (mean ± SD: 5.3 ± 3.2 h vs. 3.4 ± 1.2 h), and in the study by Giachetto et al. [31], the half-life on day 3 was higher than the half-life on day 1 (mean ± SD: 4.5 ± 3.07 h vs. 3.1 ± 0.78 h, respectively.
3.8.5 Vancomycin Area Under the Concentration–Time Curve
Vancomycin AUC24 was reported in eight studies [3, 21, 23,24,25,26, 29, 31]. Of these, only five indicated the methodology used for the calculation.
Avedissian et al. [23] estimated the AUC24 by dividing the 24-h dose (mg/day) by the vancomycin CL (L/h).
Sridharan et al. [21] calculated AUC0–t as the AUC for the dosing interval between 6 and 12 h. AUC0−∞ was extrapolated by adding AUC0–t to the ratio of the vancomycin concentration at the last time point upon kel. They determined AUC0−24 by multiplying AUC0−∞ by the number of doses prescribed per day.
Acuna et al. [24] used a Bayesian method implemented with the Therapeutic Drug Monitoring System 2000 and LEXI-calc software for AUC calculation.
Mali et al. [3] calculated AUC from 0 to 8 h and then calculated the AUC0-24 by tripling the AUC0-8.
Giachetto et al. [31] used the formula AUC24 = daily dose/clearance. For the AUC0−24/MIC ratio, MIC values of 1 µg/mL and 2 µg/mL were considered.
Five studies reported AUC0−24 < 400 µg·h/mL [3, 21, 23, 25, 31].
In the study by Acuña et al. [24], a therapeutic AUC0-24 (> 400 μg·h/mL) was observed in 39% of the 41 patients with trough plasma concentrations of 5–15 μg/mL. Importantly, a therapeutic vancomycin AUC0−24 (≥ 400 μg·h/mL) was obtained in 16% of patients with initial trough vancomycin concentrations between 5.0 and 9.9 μg/mL and in 81% of patients with initial plasma trough concentrations between 10.0 and 15 μg/mL. The AUC0−24 observed in children < 2 years was similar to that in children ≥ 2 years.
In the study by Zylbersztajn et al. [26], children in all groups had AUC0−24 > 400 µg·h/mL; however, AUC0-24 was higher in children without AKI or RRT compared to children in the AKI and RRT group and the AKI-only group. In the study by Giachetto et al. [31], the AUC0−24 on the first day of treatment was the same as the AUC0-24 on day 3. The study by Mali et al. [3] showed that 65.71% of 12 children had vancomycin trough concentrations below the therapeutic range, and vancomycin AUC0−24 > 400 μg·h/mL was achieved in 50% of the study population. Sridharan et al. [21] found that trough vancomycin concentration was a good predictor of AUC0-24 [0.86; 95% CI 0.8–0.9; P = 0.0001]. The study findings are summarized in Table 2.
3.9 Vancomycin Pharmacodynamics in Critically Ill Children
3.9.1 Vancomycin Therapeutic Concentrations
Therapeutic targets for trough concentration varied in the reviewed studies. An optimal trough concentration of ≥ 15 μg/L is recommended [33].
Acuña et al. [24] evaluated the percentage of patients with trough plasma levels in the therapeutic range of 5–15 μg/mL. Forty-one of the 84 children enrolled in the study had baseline vancomycin plasma concentrations in the therapeutic range after receiving 40 mg/kg/day.
In the study by Gous et al. [30], the therapeutic plasma concentration targets included trough concentrations of 5–10 μg/mL and peak concentrations of 25–40 μg/mL. Only 53% of the children on day 3 and 27% of the children on day 9 had trough concentrations within the therapeutic range, while 53% of the children on day 3 and 60% of the children on day 9 had peak concentrations within the therapeutic range.
In the study by Mali et al. [3], 35 trough concentrations of 12 subjects receiving 20 mg/kg/day were evaluated against the recommended trough range of 10–20 μg/mL. A total of 23 (65.71%) trough concentrations were below the range, 11 (31.43%) trough concentrations were within the range, while 1 (2.86%) trough concentration was above the recommended range.
Zylbersztajn et al. [26] provided initial doses of 40 mg/kg/day (median) every 6 or 8 h. In 15 children with normal renal function, therapeutic trough concentrations (10–20 μg/mL) were observed in 8 (53%) children, supra-therapeutic levels in 4 (27%) children and sub-therapeutic concentrations in 3 (20%) children. In 11 children with AKI, trough levels of vancomycin within the therapeutic range were observed in 3 (27%) children, 7 (64%) had supra-therapeutic levels, and 1 (9%) child had sub-therapeutic trough levels. Although dose adjustment was done in the 4 patients with supra-therapeutic values, 3 of them did not achieve the target values because of death or termination of treatment. Treatment was stopped in the patient with sub-therapeutic concentrations before dose adjustment. Target therapeutic concentrations were achieved in 7 (63%) patients after one or two dose modifications. The final adjusted dose of 20 mg/kg/day every 12 h yielded a median trough level of 15.95 (12.1–18.03) μg/mL. Only 3 (10%) patients out of the 29 on RRT achieved therapeutic vancomycin trough concentrations after the initial dose.
Genuini et al. [25] assessed the number of children on vancomycin continuous infusion who attained target vancomycin plasma concentrations between 15 and 30 μg/mL. Of the 28 children enrolled in the study, 12 patients (43%) achieved therapeutic vancomycin concentrations after the initial dose. Twenty patients had more than one vancomycin trough concentration. Of those, 9 (45%) had the second measured vancomycin trough concentration within the therapeutic range. The median vancomycin trough concentration before dosing adjustment was 12.1 (9.8–17.3) μg/mL; after dosing adjustment it was 15.5 (10–25.7) μg/mL.
Peak vancomycin concentrations of above 20–40 µg/mL and trough vancomycin concentrations above 5–10 µg/mL were considered therapeutic by Giachetto et al. [31]. On the first day, 22 children were treated with a vancomycin dose of 40 mg/kg/day in four divided doses each given as an infusion over 1 h. Of these, 7 had therapeutic peak concentrations (23.9–53.5 µg/mL) and 16 had therapeutic trough concentrations (5.02–2 µg/mL). After dose adjustment (44 mg/kg/day) in 15 children, therapeutic peak (20.56–45.4 µg/mL) and trough (6.65–29.2 µg/mL) concentrations were attained in 7 and 10 children, respectively, on day 3.
In another group of children [22], initial dosing of 47 mg/kg/day in 45 children yielded trough therapeutic concentrations (15–20 μg/mL) in 2 (4.4%) children. Thirty-seven children (82.2%) had sub-therapeutic trough concentrations. Of these, 10 had trough concentrations between 10 and 14.9 μg/mL. Six children (13.3%) had supra-therapeutic concentrations.
In the study by Seixas et al. [27], a mean vancomycin dose of 59.23 ± 49.85 mg/kg/day yielded vancomycin trough concentrations between 15 and 20 μg/mL in 35 (13.6%) of the 256 plasma measurements, while trough concentrations of > 20 μg/mL were observed among 73 (28.5%) concentrations.
3.9.2 Vancomycin Concentration–Area Under the Curve (AUC)/Minimum Inhibitory Concentration (MIC)
A target of AUC24/MIC ≥ 400 is recommended for bacteria with a MIC ≤ 1 µg/mL [33]. The target concentration is increased in the treatment of infection caused by bacteria with a higher MIC.
In the study by Zylbersztajn et al. [26], children on ECMO with no AKI and no RRT (N = 15) who received 40 mg/kg/day of vancomycin in four divided doses every 6 h achieved AUC24/MIC > 400.
Assuming a MIC ≤ 1 μg/mL, 25% of the 28 of patients in the study by Genuini et al. [25] reached the target AUC24/MIC of > 400. Vancomycin serum concentrations in these children were all > 15 μg/mL (17.1–23.7 μg/mL).
In the study by Giachetto et al. [31], most patients (>70%) attained minimum therapeutic trough concentrations (5–10 µg/mL). However, on day 1, only 9 of 18 children attained AUC24/MIC > 400 when MIC = 1 µg/mL, and when MIC = 2 µg/mL, only 1 patient attained AUC24/MIC > 400. On day 3, 7 children attained an AUC24/MIC < 400 when MIC = 1 µg/mL, and 1 patient did when MIC = 2 µg/mL.
In the study by Silva et al. [28], an AUC/MIC > 400 was observed in 18% of plasma samples with trough concentrations of < 15 µg/mL. In all patients with trough concentrations of > 15 µg/mL, the AUC/MIC was > 400. In the study by Seixas et al. [27], when MIC = 1 µg/mL, a trough serum concentration of ≥ 15 µg/mL had a 94% positive predictive value for AUC24/MIC > 400, with a negative predictive value of 75% (sensitivity = 73%; specificity = 95%). For a MIC of 1.5 µg/mL, a trough serum concentration of ≥ 15 µg/mL showed a 64% positive predictive value for AUC24/MIC > 400, with a negative predictive value of 97% (sensitivity = 95%; specificity = 79%). When MIC = 2 µg/mL, trough levels of ≥ 15 µg/mL had a 33.6% positive predictive value for AUC24/MIC > 400, with a negative predictive value of 100% (sensitivity = 100%; specificity = 67%).
3.9.3 Vancomycin Concentration–Toxicity
Only the study by Seixas et al. [27] assessed vancomycin-related nephrotoxicity in patients. Vancomycin-related nephrotoxicity was observed in 21 (22.3%) of the 94 study participants. Nineteen of those patients received cyclosporine, tacrolimus, amikacin and amphotericin. Bivariate logistical regression showed that serum trough level > 20 μg/mL (P = 0.0001, odds ratio (OR) = 20.23) and duration of vancomycin therapy > 10 days (P = 0.016, OR = 4.95) or > 14 days (P = 0.001, OR = 5.35) were predictive of nephrotoxicity. The multivariate model showed that only a serum trough level of > 20 μg/mL was an independent predictor of nephrotoxicity. Creatinine levels at the beginning of vancomycin treatment, the day before the observation of an elevated vancomycin serum concentration, and 24 h after the reduction of the dose or serum level were evaluated to establish causality between the occurrence of elevated serum levels of vancomycin and nephrotoxicity. In 7 of the 21 children with nephrotoxicity, creatinine levels had a direct incremental relationship with serum vancomycin concentration. Hence, increased vancomycin levels lead to worsened renal function. Vancomycin-related nephrotoxicity was a possible risk factor for death (P = 0.006, OR = 5), as was creatinine clearance < 70 mL/min/1.73 m2 on at least one occasion (P = 0.0008, OR = 8.75). Failure to obtain AUC/MIC > 400 was not a significant possible risk factor for death. Ten deaths occurred during vancomycin treatment; one of these deaths was of a child with sepsis caused by Staphylococcus epidermidis with a MIC of 1 µg/mL, where AUC/MIC > 400 was attained after three dose adjustments (of up to 80 mg/kg/day) but was associated with renal deterioration.
3.10 Known Factors Affecting Vancomycin Pharmacokinetics
The effects of renal function, protein binding, temperature, fluid balance and concomitant medication on vancomycin pharmacokinetics were evaluated in seven studies [3, 20, 21, 23, 25, 29, 31].
3.10.1 Renal Function
Vancomycin trough and peak concentrations increased as urea and creatinine serum levels increased [20, 27] and CL decreased [27]. However, CL increased with increased creatinine clearance.
Sridharan et al. [21] compared vancomycin pharmacokinetics between children with augmented renal clearance (ARC, defined as CRCL > 130 ml/min/1.73 m2) and those with normal renal clearance (NRC). In the 40% of the patients with ARC, there was significantly higher CL, a shorter half-life and a lower AUC24. None of the children with ARC achieved AUC24 > 400 μg·h/mL after the first dose, while only 26.5% of the children achieved this concentration at steady state.
Avedissian et al. [23] found that the trough level in 79% of children with ARC (29 children) was sub-therapeutic (< 10 μg/mL) compared to 53% of children with NRC (250 children), although the dose per weight was similar in the two groups. There was no significant difference in AUC24 between the groups.
3.10.2 Protein Binding
Only Sridharan et al. [21] studied the effect of protein binding in critically ill children. The free vancomycin concentration was 77.5% of the total vancomycin concentration. Younger children (1 month to 5 years) had significantly lower protein-free vancomycin concentrations than older children (6–17 years). Infants (1–12 months) had higher Vd compared to older children (13 months to 17 years), which could be associated with altered plasma binding. Protein binding did not affect the attainment of AUC 24 > 400 μg·h/mL.
3.10.3 Temperature
Zane et al. [20] studied vancomycin pharmacokinetics in normothermic (NT, body temperature between 36.3 and 37.6 °C) patients and children undergoing therapeutic hypothermia (TH, body temperature between 32 and 34°C) for up to 12 h, followed by up to 10–12 h of rewarming.
They found a direct relationship between vancomycin CL and hypothermia. Vancomycin CL was reduced by 25%, 10% and 0% when the body temperature was 32 °C, 35 °C and 37 °C, respectively. Children with renal dysfunction treated with TH showed a reduction in vancomycin clearance of about 84%.
3.10.4 Fluid Balance
Giachetto et al. [31] studied the effect of water balance on vancomycin pharmacokinetics. Of the 16 children included in the study, only 2 had a negative water balance prior to vancomycin administration. Children with a negative water balance had higher peak and trough levels compared with children with a positive fluid balance on day 1 and day 3 of vancomycin therapy. The Vd of vancomycin on day 1 and day 3 of therapy was also lower in these children.
In the study by Gous et al. [30], 3 patients receiving fluid resuscitation in the early stages of their therapy had a large Vd and a prolonged half-life on day 3, but the Vd and half-life were within normal ranges on day 9.
3.10.5 Concomitant Medication
The effect of concomitant nephrotoxic drugs in critically ill children on continuous vancomycin infusion was studied by Genuini et al. [25]. Children included in this study had normal renal function before vancomycin initiation. Renal dysfunction in this study was defined as serum creatinine ≥ 2 times the upper limit of normal concentration for age or a twofold increase in the baseline creatinine. Three (aged 3.7, 7.6 and 11.9 years, respectively) out of the 20 children on concomitant nephrotoxic drugs (aminoglycosides, cyclosporin, tacrolimus, or diuretics) developed renal dysfunction. They had vancomycin plasma concentrations of 25.6, 15.5 and 22.9 μg/mL, respectively, at the beginning of their renal dysfunction. The child aged 7.6 years received two concomitant nephrotoxic drugs while the others received one. The patients recovered without renal replacement therapy.
3.11 Dose Recommendation
Most studies did not recommend doses, but low vancomycin serum concentrations were observed at current doses. Villena et al. [13] suggested that higher doses of vancomycin are necessary, especially in children between 1 and 12 years. A median trough concentration of 10.4 (1.4–25.5) μg/mL was observed. They, however, did not specify an appropriate dose.
Gomez et al. [20] suggested an increased vancomycin dose to 80 mg/kg/day in critically ill children and recommended a dose of 90–100 mg/kg/day in paediatric burn patients with sepsis. This finding was based on their study of 13 children aged 1–11 years with severe thermal injuries and a sepsis diagnosis. The vancomycin trough concentration increased significantly from 7.4 ± 6.0 μg/mL to 13.0 ± 4.8 μg/mL after dose adjustment from 43.4 ± 9.0 mg/kg to 98.0 ± 17.9 mg/kg.
4 Discussion
4.1 Main Findings
As expected, this systematic review has included data from a wide range of patients in terms of age and weight, underlying state of health, reason for admission to the PICU, therapies that were being administered, indication for antibiotic therapy, organ dysfunction (particular attention was paid to renal dysfunction) and organ support therapies (including renal replacement therapy and ECMO). Perhaps less expected was the range of methods reported in the studies.
A wide range of methods were utilized to measure vancomycin concentrations. Although it is likely that most of these methods give equivalent results, that has not always been confirmed, and there are concerns that some methodologies may cross-react with other medications used in the PICU, with resultant inaccuracies [34]. Ideally, the measurement methods used should be both standardized and quality controlled.
The reviewed studies used a wide range of dosing strategies and blood sampling times, which may have contributed somewhat to the wide variations in the pharmacokinetics of vancomycin observed in the studies. Given the wide range of clinical conditions and indications for vancomycin, it is likely that this will persist. However, there should be congruence between the therapeutic strategies and the monitoring processes used. As an example, very different sampling patterns could be used to reasonably accurately calculate AUC24 if intermittent or continuous infusions of vancomycin are administered.
Some studies [3, 22, 23, 26,27,28,29,30] emphasized that blood samples were obtained at steady state; however, the authors’ definitions of the steady state varied. The steady-state concentration of a drug refers to a dynamic equilibrium where the drug concentrations remain consistently within therapeutic limits for extended and potentially indefinite periods of time [35]. This must rely on consistent and appropriate dosing, but it is equally dependent on the stability of the patient physiology. Stability is often difficult to establish in critically ill children, particularly during acute phases of the illness, because of the different disease processes and medical interventions that are employed [13]. For example, changes such as capillary leaks, fluid shifts, altered renal clearance (both increased and decreased), changes in protein concentration and drug binding and changed hepatic function may lead to dramatic changes in vancomycin serum concentrations at standard doses [36]. Therapeutic interventions such as renal replacement therapy, extracorporeal membrane oxygenation, fluid administration, blood product administration, use of vasoactive agents and administration of multiple drugs may all contribute to changes in pharmacodynamics. In the PICU population, it cannot be assumed that patients will achieve steady state after a set number of doses. It would seem sensible to assume that patients’ pharmacokinetic and pharmacodynamic parameters will continue to change while there are substantial changes in parameters such as fluid balance, protein levels, pH and organ function (particularly renal and, to a lesser extent, hepatic function). Thus, achievement of optimal therapeutic vancomycin levels may depend on ongoing testing, ongoing dose adjustment, and clinical responses to therapy.
There was a lack of standardization of the timing of specimen collection. Peak and trough specimens were collected at variable times in the reviewed studies, with the timing of trough specimens ranging from < 30 min to > 2 h before the next dose, and peak specimens being collected between 30 and 60 min after infusion. It is difficult to predict the impact of these timing changes, but if the half-life is < 6 h, a difference of up to 2 h may lead to a substantial difference in values (and the related calculations). To add to the challenge, a variety of techniques were utilized to calculate pharmacokinetic and pharmacodynamic values in the different studies. Some studies incorporated enough specimens to directly calculate parameters such as Vd, CL and area under the concentration–time curve, but other studies estimated some of these values from trough levels (with or without peak levels), thus inevitably incorporating assumptions about values and compartmental dynamics. Finally, a wide variety of compartmental models were utilized to calculate pharmacokinetic and pharmacodynamic values. Clearly, compartmental dynamics may be profoundly affected by pathophysiological features present in critically ill children, and so these models must be utilized with care.
As expected, in all studies there was substantial variability in pharmacokinetic parameters, including trough, peak, CL, Vd, t1/2 and AUC24, both between groups of patients and between individual patients. These parameters also varied over time, although this might be relatively unpredictable.
The common feature of a high Vd is in keeping with previous reviews. Mula and Pooboni [37] reported that Vd was higher in children than in adults [37]. Critically ill children typically have higher Vd values than non-critically ill children [8, 36], and this is usually related to fluid resuscitation and positive fluid balances. It is important to note that the Vd is related to the fluid balances incurred by these patients (both prior to [31] and after [30] initiation of therapy).
There is limited information on protein binding in this review, but previous authors have shown that protein binding is generally lower in younger children (possibly due to lower albumen levels, but also due to low IgA levels [38, 39]) with higher free vancomycin levels. This is an area that will require further research in the future, particularly as protein levels may change dramatically in critically ill children due to their illness, and therapies may include plasma products and albumin infusions. While Sridharan et al. [21] stated that the attainment of AUC24 > 400 μg·h/mL was not affected by vancomycin binding, it is difficult to know what the implications of protein binding would be for AUC24/MIC.
Vancomycin clearance ranged from very low to very high and showed a remarkable range in the studies reported. In one study, CL after the first dose was similar to that after two to three doses of vancomycin [21], but this was in a group of patients with no renal dysfunction. The CL may have changed dramatically in patients with evolving renal or multiorgan dysfunction. Concomitant administration of a nephrotoxic medication led to reduced clearance with increased plasma concentrations of vancomycin [25]. Although there are associations between high levels of vancomycin and the use of nephrotoxic drugs such as furosemide in critically ill children [4, 40], more studies are required to understand the nature of the relationships between vancomycin, AKI and other agents. Clearly, increased vancomycin CL occurs in some patients and may be associated with substantially sub-therapeutic levels of vancomycin in patients with augmented renal excretion [23, 41]. Intriguingly, hypothermia is also related to decreased vancomycin clearance [20], and this will need to be taken into account in future studies with hypothermic patients.
The half-life of vancomycin in the reviewed studies ranged from 2 to 24 h. The presence of AKI, even with RRT, was associated with a prolonged half-life (as expected).
AUC24 was calculated in many of the studies, and this has come under increased focus, as adult studies suggest that AUC24 > 400 μg·h/mL is associated with improved outcomes. This was not confirmed by Kloprogge et al. in a paediatric study [36]. It is not clear to what extent this may be driven by issues such as variable penetration of vancomycin into tissues such as the cerebrospinal fluid [42] or the lung [43], or issues such as relative amounts of protein binding [44].
Unfortunately, there are concerns about the assumptions made in the calculation of AUC24 in the paediatric setting, particularly when relying only on trough levels and because there is little information about the appropriate compartmental models to use.
4.2 Implications for Current Practice
-
i.
This review of vancomycin pharmacokinetics studies in children has shown wide variability in CL and Vd as a result of maturation and pathophysiological changes
-
ii.
This review of vancomycin pharmacokinetics and pharmacodynamics has shown that there is even higher variability in the PICU context, even when excluding patients with AKI, and has highlighted the need for much more detailed studies.
5 Conclusion
Much more detailed studies with higher numbers of patients and samples will be required in order to understand the relationships between vancomycin therapy, pharmacokinetic/pharmacodynamic studies and modelling, and patient outcomes (death and adverse outcomes such as AKI). Those studies will require a much more standardized approach to methodology in the sampling, measurement, modelling and integration of relevant critical care elements. In the interim, it would seem reasonable to continue with therapeutic drug monitoring on a frequent basis until at least there is assurance that the patients have indeed stabilized and reached a steady state.
6 Recommendations for Future Studies
-
i.
Studying the pharmacokinetics of vancomycin while considering age-related changes in renal function will be informative but may be more appropriately done outside of the PICU
-
ii.
There is a need for standardized studies of vancomycin pharmacokinetics and pharmacodynamics in the PICU context
-
iii.
The potential effect of drug–plasma protein binding in critical illness in children should be evaluated
-
iv.
The effect of drug−drug interactions of nephrotoxic drugs in the PICU needs further evaluation, but this will be challenging given the context
-
v.
The effect of intermittent vs. continuous vancomycin infusion on the plasma concentration in critically ill children needs to be evaluated in more detail, although continuous infusions seem appropriate
-
vi.
Estimating the pharmacokinetics of vancomycin in sub-populations of critically ill children defined by their disease state may be helpful to understand the effects of particular disease states and disease progression on vancomycin pharmacokinetics in the patient sub-groups, but this will require both high numbers of patients and drug sampling
-
vii.
Prospective studies with sample sizes large enough to establish the effects of covariates on vancomycin pharmacokinetics and to make dosing recommendations are necessary.
References
Levine DP. Vancomycin: a history. Clin Infect Dis. 2006;42(Suppl 1):5–12.
Bruniera FR, et al. The use of vancomycin with its therapeutic and adverse effects: a review. Eur Rev Med Pharmacol Sci. 2015;19(4):694–700.
Mali NB, et al. Steady-state pharmacokinetics of vancomycin in children admitted to pediatric intensive care unit of a tertiary referral center. Indian J Crit Care Med. 2019;23(11):497–502. https://doi.org/10.5005/jp-journals-10071-23275.
Bonazza S, Bresee LC, Kraft T, Catherine Ross B, Dersch-Mills D. Frequency of and risk factors for acute kidney injury associated with vancomycin use in the pediatric intensive care unit. J Pediatr Pharmacol Ther. 2016;21(6):486–93. https://doi.org/10.5863/1551-6776-21.6.486.
Liu C, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):18–55. https://doi.org/10.1093/cid/ciq146.
Drennan PG, Begg EJ, Gardiner SJ, Kirkpatrick CMJ, Chambers ST. The dosing and monitoring of vancomycin: what is the best way forward? Int J Antimicrob Agents. 2019;53(4):401–7. https://doi.org/10.1016/J.IJANTIMICAG.2018.12.014.
Elyasi S, Khalili H, Dashti-Khavidaki S, Mohammadpour A. Vancomycin-induced nephrotoxicity: mechanism, incidence, risk factors and special populations. A literature review. Eur J Clin Pharmacol. 2012. https://doi.org/10.1007/s00228-012-1259-9.
Kishk OA, Lardieri AB, Heil EL, Morgan JA. Vancomycin AUC/MIC and corresponding troughs in a pediatric population. J Pediatr Pharmacol Ther. 2017;22(1):41–7. https://doi.org/10.5863/1551-6776-22.1.41.
Poston-Blahnik A, Moenster R. Association between vancomycin area under the curve and nephrotoxicity: a single center, retrospective cohort study in a veteran population. Open Forum Infect Dis. 2021;8(5):1–6. https://doi.org/10.1093/ofid/ofab094.
Tsutsuura M, et al. The monitoring of vancomycin: a systematic review and meta-analyses of area under the concentration-time curve-guided dosing and trough-guided dosing. BMC Infect Dis. 2021;21(1):1–15. https://doi.org/10.1186/s12879-021-05858-6.
Tongsai S, Koomanachai P. The safety and efficacy of high versus low vancomycin trough levels in the treatment of patients with infections caused by methicillin-resistant Staphylococcus aureus: a meta-analysis. BMC Res Notes. 2016;9(1):1–16. https://doi.org/10.1186/s13104-016-2252-7.
Smits A, De Cock PAJG. Antibiotic PK/PD research in critically ill neonates and children: how do we proceed? Int J Pharmacokinet. 2018;3(1):1–3. https://doi.org/10.4155/ipk-2017-0019.
Thakkar N, Salerno S, Hornik CP, Gonzalez D. Clinical pharmacology studies in critically Ill children. Pharm Res. 2017;34(1):7–24. https://doi.org/10.1007/s11095-016-2033-y.
Marsot A. Pharmacokinetic variability in pediatrics and intensive care: toward a personalized dosing approach. J Pharm Pharm Sci. 2019;21(1):354–62. https://doi.org/10.18433/JPPS30082.
Wu G, Furlanut M. Prediction of serum vancomycin concentrations using one-, two- and three-compartment models with implemented population pharmacokinetic parameters and with the Bayesian method. J Pharm Pharmacol. 1998;50(8):851–6. https://doi.org/10.1111/j.2042-7158.1998.tb03999.x.
Mahmood I. Dosing in children: a critical review of the pharmacokinetic allometric scaling and modelling approaches in paediatric drug development and clinical settings. Clin Pharmacokinet. 2014;53(4):327–46. https://doi.org/10.1007/s40262-014-0134-5.
Hartman SJF, Brüggemann RJ, Orriëns L, Dia N, Schreuder MF, de Wildt SN. Pharmacokinetics and target attainment of antibiotics in critically ill children: a systematic review of current literature. Clin Pharmacokinet. 2020. https://doi.org/10.1007/s40262-019-00813-w.
Picot A, Hartwell J,Harris D, Mendes P, Clegg D, Takeda AJ. Appendix 2: The Preferred Reporting Items for Systematic Reviews and Meta-Analyses checklist. In: The effectiveness of interventions to treat severe acute malnutrition in young children: a systematic review. Health Technology Assessment no. 16.19. Southampton: NIHR Journals Library; 2012. Available at https://www.ncbi.nlm.nih.gov/books/NBK98563/.
Whiting P, et al. ROBIS: a new tool to assess risk of bias in systematic reviews was developed. J Clin Epidemiol. 2016;69:225–34. https://doi.org/10.1016/j.jclinepi.2015.06.005.
Zane NR, et al. A population pharmacokinetic analysis to study the effect of therapeutic hypothermia on vancomycin disposition in children resuscitated from cardiac arrest. Pediatr Crit Care Med. 2017;18(7):e290–7. https://doi.org/10.1097/PCC.0000000000001198.
Sridharan K, Al Daylami A, Ajjawi R, Al-Ajooz H, Veeramuthu S. Clinical pharmacokinetics of vancomycin in critically ill children. Eur J Drug Metab Pharmacokinet. 2019;44(6):807–16. https://doi.org/10.1007/s13318-019-00568-6.
Villena R, González CA, Nalegach ME, Vásquez A, Villareal M, Drago M. Monitoreo terapéutico de vancomicina intravenosa en una unidad de paciente crítico pediátrico. Rev Chil Infectol. 2014;31(3):249–53. https://doi.org/10.4067/S0716-10182014000300001.
Avedissian SN, et al. Augmented renal clearance using population-based pharmacokinetic modeling in critically ill pediatric patients. Pediatr Crit Care Med. 2017;18(9):e388–94. https://doi.org/10.1097/PCC.0000000000001228.
Acuña C, Morales J, Castillo C, Torres JP. Phamacokinetics of vancomycin in children hospitalized in a critical care unit. Rev Chil Infectología. 2013;30(6):585–90. https://doi.org/10.4067/S0716-10182013000600001.
Genuini M, et al. Achievement of therapeutic vancomycin exposure with continuous infusion in critically Ill children. Pediatr Crit Care Med. 2018;19(6):e263–9. https://doi.org/10.1097/PCC.0000000000001474.
Zylbersztajn BL, et al. Therapeutic drug monitoring of vancomycin in pediatric patients with extracorporeal membrane oxygenation support. J Pediatr Pharmacol Ther. 2018;23(4):305–10. https://doi.org/10.5863/1551-6776-23.4.305.
Seixas GTF, Araujo OR, Silva DCB, Arduini RG, Petrilli AS. Vancomycin therapeutic targets and nephrotoxicity in critically ill children with cancer. J Pediatr Hematol Oncol. 2016;38(2):e56–62. https://doi.org/10.1097/MPH.0000000000000470.
Silva DCB, Seixas GT, Araujo OR, Arduini RG, Carlesse FA, Petrilli AS. Vancomycin serum concentrations in pediatric oncologic/hematologic intensive care patients. Braz J Infect Dis. 16(4):361–365. https://doi.org/10.1016/j.bjid.2012.06.011
Gomez DS, et al. Individualised vancomycin doses for paediatric burn patients to achieve PK/PD targets. Burns. 2013;39(3):445–50. https://doi.org/10.1016/j.burns.2012.07.005.
Gous AGS, Dance MD, Lipman J, Luyt DK, Mathivha R, Scribante J. Changes in vancomycin pharmacokinetics in critically ill infants. Anaesth Intensive Care. 1995;23(6):678–82. https://doi.org/10.1177/0310057x9502300603.
Giachetto GA, Telechea HM, Speranza N, Oyarzun M, Nanni L, Menchaca A. Vancomycin pharmacokinetic-pharmacodynamic parameters to optimize dosage administration in critically ill children. Pediatr Crit Care Med. 2011;12(6):250–4. https://doi.org/10.1097/PCC.0b013e3181fe4047.
Le J, et al. Pharmacodynamic characteristics of nephrotoxicity associated with vancomycin use in children. J Pediatric Infect Dis Soc. 2015;4(4):e109–16. https://doi.org/10.1093/jpids/piu110.
Rybak MJ, et al. Therapeutic monitoring of vancomycin for serious methicillin-resistant Staphylococcus aureus infections: a revised consensus guideline and review by the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the Society of Infectious Diseases Pharmacists. Am J Heal Pharm. 2020;77(11):835–63. https://doi.org/10.1093/ajhp/zxaa036.
Singer B, Stevens RW, Westley BP, Nicolau DP. Falsely elevated vancomycin-concentration values from enzyme immunoassay leading to treatment failure. Am J Heal Pharm. 2020;77(1):9–13. https://doi.org/10.1093/ajhp/zxz258.
Cascella M, Wadhwa RR. Steady state concentration. Treasure Island: StatPearls Publishing; 2021.
Kloprogge F, et al. Revising pediatric vancomycin dosing accounting for nephrotoxicity in a pharmacokinetic-pharmacodynamic model. Antimicrob Agents Chemother. 2019;63(5):1–12.
Mulla H, Pooboni S. Population pharmacokinetics of vancomycin in patients receiving extracorporeal membrane oxygenation. Br J Clin Pharmacol. 2005;60(3):265–75. https://doi.org/10.1111/j.1365-2125.2005.02432.x.
Smits A, et al. Factors impacting unbound vancomycin concentrations in neonates and young infants. Eur J Clin Microbiol Infect Dis. 2018;37(8):1503–10. https://doi.org/10.1007/s10096-018-3277-8.
Oyaert M, et al. Factors impacting unbound vancomycin concentrations in different patient populations. Antimicrob Agents Chemother. 2015;59(11):7073–9. https://doi.org/10.1128/AAC.01185-15.
Totapally BR, Machado J, Lee H, Paredes A, Raszynski A. Acute kidney injury during vancomycin therapy in critically ill children. Pharmacotherapy. 2013;33(6):598–602. https://doi.org/10.1002/phar.1259.
Baptista JP, Sousa E, Martins PJ, Pimentel JM. Augmented renal clearance in septic patients and implications for vancomycin optimisation. Int J Antimicrob Agents. 2012;39(5):420–3. https://doi.org/10.1016/J.IJANTIMICAG.2011.12.011.
Shokouhi S, Alavi Darazam IB. Determination of vancomycin trough level in serum and cerebrospinal fluid of patients with acute community-acquired meningitis: a prospective study. J Infect. 2014;69(5):424–9. https://doi.org/10.1016/j.jinf.2014.06.010.
Kollef MH. Limitations of vancomycin in the management of resistant staphylococcal infections. Clin Infect Dis. 2007;45:191–6. https://doi.org/10.1086/519470.
De Cock PAJG, et al. Impact of vancomycin protein binding on target attainment in critically ill children: back to the drawing board? J Antimicrob Chemother. 2017;72(3):801–4. https://doi.org/10.1093/jac/dkw495.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None received.
Availability of Data and Material
No data available.
Conflicts of Interest
The authors declare no conflict of interest.
Ethics Approval
Not applicable.
Author Contributions
OOA identified potential papers to review, wrote the first draft and extensively revised the manuscripts. PM extensively checked the included papers and revised the manuscript. AA extensively checked the included papers and revised the manuscript.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Akunne, O.O., Mugabo, P. & Argent, A.C. Pharmacokinetics of Vancomycin in Critically Ill Children: A Systematic Review. Eur J Drug Metab Pharmacokinet 47, 31–48 (2022). https://doi.org/10.1007/s13318-021-00730-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13318-021-00730-z