Abstract
Background and Objective
The effectiveness of direct-acting antivirals (DAAs) is not well established in end-stage renal disease (ESRD) patients. Assessment of the plasma concentrations may support understanding of their therapeutic outcomes in this population. The aim of this study is to develop a direct, yet matrix-effect tolerant, analytical method for determining DAAs in the plasma of ESRD patients while maintaining a moderate cost per sample and with an improved analyte extraction recovery.
Methods
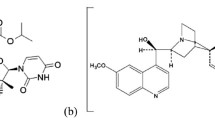
In this study, a liquid chromatography–tandem mass spectrometric (LC–MS/MS) method was developed for the analysis of ombitasvir (OMB), paritaprevir (PRT) and ritonavir (RIT) in plasma. Sample preparation was performed using the liquid–liquid extraction (LLE) method. Isocratic separation was performed using a mixture of methanol and 10 mM ammonium acetate (79:21, v/v) followed by MS/MS detection. The method was validated and applied to determine DAAs in the plasma of ESRD patients (n = 7).
Results
The developed method was linear (r2 > 0.995), accurate (89.4 ± 7.8 to 108.3 ± 3.0) and precise (% CV 0.9–15.0) and showed improved recovery (> 80) over previously published ones in the range 5–250, 30–1,500, 20–1,000 ng/mL for OMB, PRT and RIT, respectively. Relative matrix effect was absent, and the method accurately determined the three DAAs in real-life samples (n = 7).
Conclusions
An efficient analytical method for the determination of DAAs is presented. The method overcomes the potential analytical response fluctuation in ESRD. The developed method show improved extraction recoveries and is suitable for routine application in developing economies where hepatitis C virus is most prevalent.



Similar content being viewed by others
References
Kouyoumjian SP, Chemaitelly H, Abu-Raddad LJ. Characterizing hepatitis C virus epidemiology in Egypt: systematic reviews, meta-analyses, and meta-regressions. Sci Rep. 2018;8(1):1661. https://doi.org/10.1038/s41598-017-17936-4.
Soriano V, Vispo E, Poveda E, Labarga P, Martin-Carbonero L, Fernandez-Montero JV, et al. Directly acting antivirals against hepatitis C virus. J Antimicrob Chemother. 2011;66(8):1673–86.
Pearlman BL, Traub N. Sustained virologic response to antiviral therapy for chronic hepatitis C virus infection: a cure and so much more. Clin Infect Dis. 2011;52(7):889–900. https://doi.org/10.1093/cid/cir076.
Fried MW, Hadziyannis SJ, editors. Treatment of chronic hepatitis C infection with peginterferons plus ribavirin. Seminars in liver disease. New York: Thieme Medical Publishers, Inc.; 2004.
Foster GR, Irving WL, Cheung MCM, Walker AJ, Hudson BE, Verma S, et al. Impact of direct acting antiviral therapy in patients with chronic hepatitis C and decompensated cirrhosis. J Hepatol. 2016;64(6):1224–31. https://doi.org/10.1016/j.jhep.2016.01.029.
Schinazi R, Halfon P, Marcellin P, Asselah T. HCV direct-acting antiviral agents: the best interferon-free combinations. Liver Int. 2014;34(s1):69–78. https://doi.org/10.1111/liv.12423.
Gentile I, Buonomo AR, Borgia G. Ombitasvir: a potent pan-genotypic inhibitor of NS5A for the treatment of hepatitis C virus infection. Routledge: Taylor & Francis; 2014.
Deeks ED. Ombitasvir/paritaprevir/ritonavir plus dasabuvir: a review in chronic HCV genotype 1 infection. Drugs. 2015;75(9):1027–38.
Hézode C, Asselah T, Reddy KR, Hassanein T, Berenguer M, Fleischer-Stepniewska K, et al. Ombitasvir plus paritaprevir plus ritonavir with or without ribavirin in treatment-naive and treatment-experienced patients with genotype 4 chronic hepatitis C virus infection (PEARL-I): a randomised, open-label trial. Lancet. 2015;385(9986):2502–9.
Pockros PJ, Reddy KR, Mantry PS, Cohen E, Bennett M, Sulkowski MS, et al. Efficacy of direct-acting antiviral combination for patients with hepatitis C virus genotype 1 infection and severe renal impairment or end-stage renal disease. Gastroenterology. 2016;150(7):1590–8. https://doi.org/10.1053/j.gastro.2016.02.078.
Smolders EJ, de Kanter CTMM, van Hoek B, Arends JE, Drenth JPH, Burger DM. Pharmacokinetics, efficacy, and safety of hepatitis C virus drugs in patients with liver and/or renal impairment. Drug Saf. 2016;39(7):589–611. https://doi.org/10.1007/s40264-016-0420-2.
Talavera Pons S, Boyer A, Lamblin G, Chennell P, Châtenet F-T, Nicolas C, et al. Managing drug–drug interactions with new direct-acting antiviral agents in chronic hepatitis C. Br J Clin Pharmacol. 2017;83(2):269–93. https://doi.org/10.1111/bcp.13095.
Khatri A, Menon RM, Marbury TC, Lawitz EJ, Podsadecki TJ, Mullally VM, et al. Pharmacokinetics and safety of co-administered paritaprevir plus ritonavir, ombitasvir, and dasabuvir in hepatic impairment. J Hepatol. 2015;63(4):805–12.
Kiser JJ, Burton JR Jr, Everson GT. Drug–drug interactions during antiviral therapy for chronic hepatitis C. Nat Rev Gastroenterol Hepatol. 2013;10(10):596.
Haubrich RH, Little SJ, Currier JS, Forthal DN, Kemper CA, Beall GN, et al. The value of patient-reported adherence to antiretroviral therapy in predicting virologic and immunologic response. AIDS. 1999;13(9):1099–107.
Forte F, Portier H, Piroth L, Buisson M, Duong M, Grappin M, et al. Value of patient self-report and plasma human immunodeficiency virus protease inhibitor level as markers of adherence to antiretroviral therapy: relationship to virologic response. Clin Infect Dis. 2001;33(3):386–92. https://doi.org/10.1086/321876.
Durant J, Clevenbergh P, Garraffo R, Halfon P, Icard S, Del Giudice P, et al. Importance of protease inhibitor plasma levels in HIV-infected patients treated with genotypic-guided therapy: pharmacological data from the Viradapt Study. AIDS. 2000;14(10):1333–9.
Gross R, Bilker WB, Friedman HM, Strom BL. Effect of adherence to newly initiated antiretroviral therapy on plasma viral load. AIDS. 2001;15(16):2109–17.
Levey AS, Coresh J, Bolton K, Culleton B, Harvey KS, Ikizler TA, et al. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002;39(2 Suppl 1):S1–266.
Ariaudo A, Favata F, De Nicolò A, Simiele M, Paglietti L, Boglione L, et al. A UHPLC–MS/MS method for the quantification of direct antiviral agents simeprevir, daclatasvir, ledipasvir, sofosbuvir/GS-331007, dasabuvir, ombitasvir and paritaprevir, together with ritonavir, in human plasma. J Pharm Biomed Anal. 2016;125:369–75. https://doi.org/10.1016/j.jpba.2016.04.031.
Burger D, Hugen P, Reiss P, Gyssens I, Schneider M, Kroon F, et al. Therapeutic drug monitoring of nelfinavir and indinavir in treatment-naive HIV-1-infected individuals. AIDS. 2003;17(8):1157–65.
Stemer G, Lemmens-Gruber R. Clinical pharmacy activities in chronic kidney disease and end-stage renal disease patients: a systematic literature review. BMC Nephrol. 2011;12(1):35.
Dhondup T, Qian Q. Electrolyte and acid-base disorders in chronic kidney disease and end-stage kidney failure. Blood Purif. 2017;43(1–3):179–88.
Kim H-Y, Baylis C, Verlander JW, Han K-H, Reungjui S, Handlogten ME, et al. Effect of reduced renal mass on renal ammonia transporter family, Rh C glycoprotein and Rh B glycoprotein, expression. Am J Physiol Renal Physiol. 2007;293(4):F1238–47.
Piperi C, Kalofoutis C, Tzivras M, Troupis T, Skenderis A, Kalofoutis A. Effects of hemodialysis on serum lipids and phospholipids of end-stage renal failure patients. Mol Cell Biochem. 2004;265(1):57–61. https://doi.org/10.1023/b:mcbi.0000044315.74038.78.
Honda H, Qureshi AR, Heimbürger O, Barany P, Wang K, Pecoits-Filho R, et al. Serum albumin, c-reactive protein, interleukin 6, and fetuin a as predictors of malnutrition, cardiovascular disease, and mortality in patients with ESRD. Am J Kidney Dis. 2006;47(1):139–48. https://doi.org/10.1053/j.ajkd.2005.09.014.
George R, Haywood A, Khan S, Radovanovic M, Simmonds J, Norris R. Enhancement and suppression of ionization in drug analysis using HPLC–MS/MS in support of therapeutic drug monitoring: a review of current knowledge of its minimization and assessment. Ther Drug Monit. 2018;40(1):1–8. https://doi.org/10.1097/ftd.0000000000000471.
Hewavitharana AK, Tan SK, Shaw PN. Strategies for the detection and elimination of matrix effects in quantitative LC–MS analysis. LCGC N Am. 2014;32(1):54–64.
Kratochwil NA, Huber W, Müller F, Kansy M, Gerber PR. Predicting plasma protein binding of drugs: a new approach. Biochem Pharmacol. 2002;64(9):1355–74.
Ocque AJ, Hagler CE, DiFrancesco R, Morse GD, Talal AH. Ultra-performance liquid chromatography tandem mass spectrometry for determination of direct acting antiviral drugs in human liver fine needle aspirates. J Chromatogr B. 2017;1052:103–9. https://doi.org/10.1016/j.jchromb.2017.03.020.
Ocque AJ, Hagler CE, Difrancesco R, Woolwine-Cunningham Y, Bednasz CJ, Morse GD, et al. Development and validation of a UPLC–MS/MS method for the simultaneous determination of paritaprevir and ritonavir in rat liver. Bioanalysis. 2016;8(13):1353–63. https://doi.org/10.4155/bio-2016-0040.
Food, Administration D. Bioanalytical method validation. Guidance for industry. 2018. Available from: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/bioanalytical-method-validation-guidance-industry. Accessed 29 Oct 2019.
Matuszewski B, Constanzer M, Chavez-Eng C. Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC−MS/MS. Anal Chem. 2003;75(13):3019–30.
Viekira P. Highlights of prescribing information. North Chicago: AbbVie Inc.; 2014.
Feld JJ. Direct-acting antivirals for hepatitis C virus (HCV): the progress continues. Curr Drug Targets. 2017;18(7):851–62.
Pawlotsky JM. Treatment failure and resistance with direct-acting antiviral drugs against hepatitis C virus. Hepatology. 2011;53(5):1742–51.
Farouk F, Nabhan S, Niessen WM, Azzazy HM. LC–MS/MS assay for assessing medical adherence in patients under warfarin maintenance therapy. Microchem J. 2018;141:135–40.
Shen J, Serby M, Surber B, Lee AJ, Ma J, Badri P, et al. Metabolism and disposition of pan-genotypic inhibitor of HCV NS5A ombitasvir in humans. Drug Metab Dispos. 2016;8:1148–57.
Schellinger AP, Carr PW. Isocratic and gradient elution chromatography: a comparison in terms of speed, retention reproducibility and quantitation. J Chromatogr A. 2006;1109(2):253–66.
Velenosi TJ, Urquhart BL. Pharmacokinetic considerations in chronic kidney disease and patients requiring dialysis. Exp Opin Drug Metab Toxicol. 2014;10(8):1131–43. https://doi.org/10.1517/17425255.2014.931371.
Mason NA. Polypharmacy and medication-related complications in the chronic kidney disease patient. Curr Opin Nephrol Hypertens. 2011;20(5):492–7.
Ballew SH, Chen Y, Daya NR, Godino JG, Windham BG, McAdams-DeMarco M, et al. Frailty, kidney function, and polypharmacy: the atherosclerosis risk in communities (ARIC) study. Am J Kidney Dis. 2017;69(2):228–36.
Zurek G. LC–MS/MS as versatile tool in therapeutic drug monitoring. Pharmacopsychiatry. 2018;51(03):19.
Williams L, Lodder H, Jones R, Jordan S, Calverley R, Cleeve M et al. Phospholipid removal: a comparison between traditional liquid–liquid extraction (LLE) and supported liquid extraction (SLE) using LC-MS/MS analysis. 2009. Available from: http://data.biotage.co.jp/pdf/poster/10205_asms_2009_lle_sle+_phospolipids_2_.pdf. Accessed 29 Oct 2019.
Guiard-Schmid J-B, Poirier J-M, Meynard J-L, Bonnard P, Gbadoe AH, Amiel C, et al. High variability of plasma drug concentrations in dual protease inhibitor regimens. Antimicrob Agents Chemother. 2003;47(3):986–90. https://doi.org/10.1128/AAC.47.3.986-990.2003.
Menon RM, Klein CE, Podsadecki TJ, Chiu YL, Dutta S, Awni WM. Pharmacokinetics and tolerability of paritaprevir, a direct acting antiviral agent for hepatitis C virus treatment, with and without ritonavir in healthy volunteers. Br J Clin Pharmacol. 2016;81(5):929–40. https://doi.org/10.1111/bcp.12873.
Acknowledgements
The authors thank Professor Wilfried Niessen (hyphen MassSpec, Margrietstraat 34, 2215 HJ Voorhout, the Netherlands) for his appreciated efforts in revising this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study did not receive any funding.
Conflict of interest
Authors confirm no conflict of interest.
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Study was approved by the ethics committee of Cairo University; Faculty of Medicine; 2016.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Farouk, F., Wahba, D., Mogawer, S. et al. Development and Validation of a New LC–MS/MS Analytical Method for Direct-Acting Antivirals and Its Application in End-Stage Renal Disease Patients. Eur J Drug Metab Pharmacokinet 45, 89–99 (2020). https://doi.org/10.1007/s13318-019-00584-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13318-019-00584-6