Abstract
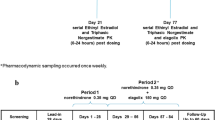
Pharmacokinetic (PK) interactions between the cytochrome P450 3A4 (CYP3A4) pathway and transdermally administered ethinyl estradiol (EE) and gestodene (GSD) were investigated. This paper reports the findings of three open-label, intra-individual, one-way crossover, Phase I trials. In two studies, women used a novel contraceptive patch for 3 weeks during two 4-week study periods; in the second period, the CYP3A4 inhibitors erythromycin (Study 1) or ketoconazole (Study 2) were administered concurrently. In a third study, women received single doses of the CYP3A4 model substrate midazolam, alone and after 3 weeks of concurrent patch application. In each period, the EE/GSD patch (delivering low EE and GSD doses resulting in the same systemic exposure as a combined oral contraceptive containing 0.02 mg EE and 0.06 mg GSD) was applied once weekly for 3 weeks, with one patch-free week. Erythromycin, ketoconazole, and midazolam were administered orally. Main outcome measures were area under the curves (AUCs) and maximum plasma concentration (C max) of EE, and total and unbound GSD (Studies 1 and 2). AUC and C max of midazolam (Study 3). Co-administration of CYP3A4 inhibitors did not affect EE metabolism, and had only weak effects on the PK of total and unbound GSD. The patch had no clinically relevant effect on metabolism of the CYP3A4 substrate midazolam.



Similar content being viewed by others
Notes
This patch was approved in various countries outside of the EU after this date.
References
Abbas R, Hug BA, Leister C, Burns J, Sonnichsen D (2011) Pharmacokinetics of oral neratinib during co-administration of ketoconazole in healthy subjects. Br J Clin Pharmacol 71:522–527. doi:10.1111/j.1365-2125.2010.03845.x
Aubeny E, Buhler M, Colau JC, Vicaut E, Zadikian M, Childs M (2004) The Coraliance study: non-compliant behavior. Results after a 6-month follow-up of patients on oral contraceptives. Eur J Contracept Reprod Health Care 9:267–277
Baerwald AR, Pierson RA (2004) Ovarian follicular development during the use of oral contraception: a review. J Obstet Gynaecol Can 26:19–24
Barbosa IC, Filho CI, Faggion D Jr, Baracat EC (2006) Prospective, open-label, noncomparative study to assess cycle control, safety and acceptability of a new oral contraceptive containing gestodene 60 microg and ethinylestradiol 15 microg (Minesse). Contraception 73:30–33. doi:10.1016/j.contraception.2005.06.057
Burkman RT (2007) Transdermal hormonal contraception: benefits and risks. Am J Obstet Gynecol 197(134):e131–e136. doi:10.1016/j.ajog.2007.04.027
Coelingh Bennink HJ (2004) Are all estrogens the same? Maturitas 47:269–275. doi:10.1016/j.maturitas.2003.11.009
Dresser GK, Spence JD, Bailey DG (2000) Pharmacokinetic-pharmacodynamic consequences and clinical relevance of cytochrome P450 3A4 inhibition. Clin Pharmacokinet 38:41–57
FDA (2006) Guidance for industry: drug interaction studies – study design, data analysis, and implications for dosing and labeling (draft guidance). http://www.fda.gov/OHRMS/DOCKETS/98fr/06d-0344-gdl0001.pdf. Accessed 7 Feb 2013
Goldzieher JW, Brody SA (1990) Pharmacokinetics of ethinyl estradiol and mestranol. Am J Obstet Gynecol 163:2114–2119
Guengerich FP (1990) Mechanism-based inactivation of human liver microsomal cytochrome P-450 IIIA4 by gestodene. Chem Res Toxicol 3:363–371
Heger-Mahn D, Warlimont C, Faustmann T, Gerlinger C, Klipping C (2004) Combined ethinylestradiol/gestodene contraceptive patch: two-center, open-label study of ovulation inhibition, acceptability and safety over two cycles in female volunteers. Eur J Contracept Reprod Health Care 9:173–181
Janssen-Cilag (2012) Evra® transdermal patch. Summary of product characteristics
Jung-Hoffmann C, Kuhl H (1990) Intra- and interindividual variations in contraceptive steroid levels during 12 treatment cycles: no relation to irregular bleedings. Contraception 42:423–438
Kuhl H (2005) Pharmacology of estrogens and progestogens: influence of different routes of administration. Climacteric 8(Suppl 1):3–63. doi:10.1080/13697130500148875
Laine K, Yasar U, Widen J, Tybring G (2003) A screening study on the liability of eight different female sex steroids to inhibit CYP2C9, 2C19 and 3A4 activities in human liver microsomes. Pharmacol Toxicol 93:77–81. doi:10.1034/j.1600-0773.2003.t01-1-930202_93_2.x
Nassr N, Lahu G, von Richter O, Reutter F, Knoerzer D, Zech K, Erb KA, Schug B, Blume H, Hermann R (2007) Lack of a pharmacokinetic interaction between steady-state roflumilast and single-dose midazolam in healthy subjects. Br J Clin Pharmacol 63:365–370. doi:10.1111/j.1365-2125.2006.02762.x
Olkkola KT, Aranko K, Luurila H, Hiller A, Saarnivaara L, Himberg JJ, Neuvonen PJ (1993) A potentially hazardous interaction between erythromycin and midazolam. Clin Pharmacol Ther 53:298–305
Palovaara S, Kivisto KT, Tapanainen P, Manninen P, Neuvonen PJ, Laine K (2000) Effect of an oral contraceptive preparation containing ethinylestradiol and gestodene on CYP3A4 activity as measured by midazolam 1′-hydroxylation. Br J Clin Pharmacol 50:333–337. doi:10.1046/j.1365-2125.2000.00271.x
Sinofsky FE, Pasquale SA (1998) The effect of fluconazole on circulating ethinyl estradiol levels in women taking oral contraceptives. Am J Obstet Gynecol 178:300–304
Stanczyk FZ (2002) Pharmacokinetics and potency of progestins used for hormone replacement therapy and contraception. Rev Endocr Metab Disord 3:211–224
Thummel KE, Wilkinson GR (1998) In vitro and in vivo drug interactions involving human CYP3A. Annu Rev Pharmacol Toxicol 38:389–430. doi:10.1146/annurev.pharmtox.38.1.389
UN Department of Economic and Social Affairs Population Division (2011) World contraceptive use. http://www.un.org/esa/population/publications/contraceptive2011/contraceptive2011.htm. Accessed 5 Dec 2012
Wilde MI, Balfour JA (1995) Gestodene. A review of its pharmacology, efficacy and tolerability in combined contraceptive preparations. Drugs 50:364–395
Zhou S, Yung Chan S, Cher Goh B, Chan E, Duan W, Huang M, McLeod HL (2005) Mechanism-based inhibition of cytochrome P450 3A4 by therapeutic drugs. Clin Pharmacokinet 44:279–304
Acknowledgments
Dr Winkler is a former employee of Bayer Technology Services GmbH, and Dr Ludwig, Dr Rohde, and Dr Zurth are employees of Bayer Pharma AG. Mark Goldammer is a former employee of PHC Pharma Consult, affiliated to Bayer Pharma AG. Financial support for this study was provided by Bayer Pharma AG, Berlin, Germany. Editorial assistance was provided by Ogilvy 4D, Oxford, UK, funded by Bayer Pharma AG.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Winkler, J., Goldammer, M., Ludwig, M. et al. Pharmacokinetic drug–drug interaction between ethinyl estradiol and gestodene, administered as a transdermal fertility control patch, and two CYP3A4 inhibitors and a CYP3A4 substrate. Eur J Drug Metab Pharmacokinet 40, 389–399 (2015). https://doi.org/10.1007/s13318-014-0215-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13318-014-0215-8