Abstract
Individually, Nigella sativa (NS) and physical training interventions have been shown to be effective preventive and therapeutic strategies for diabetes mellitus. However, the effect of these in combination on bioindicators of diabetes has not yet been evaluated; there is little information available in the literature. A systematic review was therefore performed to assess any mutually potentiating impacts of NS and physical training interventions in diabetic subjects. A search was performed on this topic in the PubMed, CINAHL, Google Scholar and Web of Science databases for randomised, quasi-randomised or non-randomised controlled trials, studies with factorial or single-cohort pre-post designs, case series as well as case reports. The search terms encompassed various combinations of the following: “exercise”, “training”, “physical activity”, “NS”, “treadmill”, “swimming”, “Thymoquinone”, “Nigellone”, “caraway oil” and “black seeds”. Two reviewers screened the abstracts of 202 identified publications according to predetermined inclusion criteria—i.e. papers published from 2009 onwards in the English language, studies on human or animal subjects, and the assessment of diabetic bioindicators following the combined administration of NS and exercise regimens in comparison with just one of these interventions or against controls. Despite the rich data available regarding the effect of both interventions separately, two human studies and two animal studies were ultimately included in the review. However, the benefit of combined administration of NS and exercise regimens on glycemic and lipidemic control was much more obvious compared to exercise alone. In conclusion, these findings suggested that combined administration of NS and exercise regimens could be used as an effective adjuvant for oral antidiabetic drugs in diabetes control.
Similar content being viewed by others
Diabetes mellitus is the most common non-communicable disease worldwide. |
Diabetes exerts a heavy economic burden on society. This burden is related to health system costs incurred by society in managing the disease, indirect costs resulting from productivity losses due to patient disability and premature mortality, time spent by family members accompanying patients when seeking care, and intangible costs (psychological pain to the family and loved ones). |
This review highlights an integrated patient-centered approach using natural and physical intervention with the traditional approach to control diabetes and/or its complications to achieve a better outcome. |
The study tries to find any significant effect of combined Nigella sativa (NS) and exercise on the diabetic profile. |
The review includes a description of the clinical and pharmaceutical merits of a combined management strategy in diabetes, which encompasses both physical activity and NS. |
The study found a significant effect of Nigella sativa supplement and training on the diabetic profile. |
Introduction
Diabetes mellitus (DM) is the most common chronic non-communicable metabolic syndrome that has been defined as a disease of the twenty-first century [1]. Although there are several different forms of DM, type II DM (T2DM) is the most common type, as it accounts for 90% of DM cases [2].
Lifestyle has been proposed to play a big role in the development of T2DM, which might be worsened by the lack of awareness of the disease [3].
Since more than 80% of T2DM patients are overweight, physical inactivity was considered to be one of the most dangerous pandemics of the twenty-first century before the advent of the COVID-19 pandemic [4, 5].
Even more, the alarming rising rates of obesity and physical inactivity as well as environmental factors are expected to duplicate the prevalence of T2DM in the next 20 years [6, 7].
T2DM is a very complex condition that can lead to metabolic, biomechanical, physiological, or psychosocial impairment [8]. Hyperglycaemia is thought to persistently trigger long-term complications of DM such as retinopathy, nephropathy, osteopathy, neuropathy, otopathy, cerebrovascular disease and peripheral artery disease as well as other cognitive impairments and dementia [9,10,11,12,13,14].
Over the last few years, many pharmacological agents have been used for the treatment of T2DM, and oral anti-hyperglycaemic therapies aim to be effective in ameliorating hyperglycaemic conditions [15].
However, several of these synthetic drugs do not always provide satisfactory results and have serious side effects such as weight gain, hypoglycaemia, gastrointestinal problems, impaired bone micro-architecture, biochemical vitamin B12 deficiency, and urinary tract infections, predominantly in females [16]. In addition, some of these medications can increase the incidence of myocardial infarction, congestive heart failure, and peripheral oedema, and they are associated with an increased risk of carcinogenesis and even death after long-term use [17]. Thus, managing DM without any side effects is a challenge. Herbal plants and lifestyle interventions, including exercise, have been proven to be important in this respect [18,19,20,21,22]. Therefore, it is necessary for researchers to investigate interventions that lead to the use of comprehensive treatment approaches by T2DM patients.
Exercise directly impacts the cardiorespiratory system and enhances its performance [23]. It also promotes intracellular signalling pathways that facilitate mitochondrial biogenesis and hypertrophy of skeletal muscle cells [24]. The benefits of adding exercise to routine clinical management encompass life quality enhancement as well as improvements in the areas of all-cause mortality, neurocognitive health, metabolic health, musculoskeletal health, cardiovascular health and cancer [25].
Nigella sativa (NS) is an annual plant belonging to the Ranunculaceae family. NS is rich in many important nutrients, vitamins, minerals, and chemical compounds that have potential therapeutic effects [26]. The seeds are mainly composed of protein, fat, and carbohydrates. The seeds contain fixed oil, alkaloids, saponins, and essential (volatile) oil. The main ingredient of NS is thymoquinone, and it is considered an active compound in several studies [27].
Many experimental and clinical studies have shown reproducible preferable physiological as well as pharmacological effects of NS, such as antioxidant, antimicrobial, anti-inflammatory, immunomodulatory, hypoglycemic and hypolipidemic effects as well as an enhancement of the cardiovascular profile [28,29,30,31].
The promising biological properties of NS and/or its active ingredients make NS a strong natural candidate for controlling DM and/or its related complications, and this effect can be magnified significantly and strengthened if combined with another non-pharmacological intervention.
Although data on the advantages of physical activity and NS in a range of pathologies and their risk factors are well documented when each therapy is applied individually, whether their actions are mutually potentiating has not yet been investigated in detail. Only a few studies have explored the use of both NS and physical activity. Therefore, the objective of the current review was to identify all studies that have examined the combined effect of NS and physical exercise used in combination on diabetic profiles.
Methods
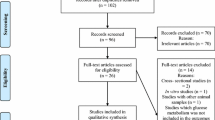
The two investigators formulated a search strategy with the use of search words identified from existing publications. The search was conducted on December 27, 2021, and included the PubMed, CINAHL, Google Scholar and Web of Science databases; it did not incorporate any filters for study design. Box A lists the inclusion and exclusion parameters applied to the screening process for the extracted citations (Table 1). The chosen titles and abstracts were acquired via the library system at Imam Abdulrahman bin Faisal University, through an online search or by communicating with the authors by email or through ResearchGate. Obtained full texts underwent additional screening in order to establish their aptness for inclusion in the review; data relevant to the review objectives were then retrieved. The systematic review followed the PRISMA guidelines (Fig. 1). This review is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Results
A total of 202 titles were retrieved from the electronic search, of which 104 were duplicates. The remaining underwent screening of the title and abstracts, and publications that failed to meet the inclusion criteria were discarded. Dosing regimens of NS, together with four different physical activity regimens, were studied in relation to diabetic profiles within the selected papers. All studies showed variation in the NS dose, type of exercise, duration, and sample size. Interestingly, combinations of both modalities showed significant synergistic effects.
Two papers were human studies, and their findings showed significant improvements in the metabolic and liver function profiles as well as uric acid and inflammatory markers, as presented in Table 2. The other two studies were animal studies, with the combined intervention modalities appearing to be more effective than exercise or NS supplement alone (Table 3).
Human Studies
The only two human studies included had a randomised controlled trial design and were published in 2011 and 2021. The total population of the two human studies was 106 subjects; 51 and 35 were included in the NS and control cohorts, respectively. Both studies recruited individuals suffering from diabetes, although the control group in the study by El-Shamy et al. [32] comprised healthy subjects.
As indicated by Table 2, the formulations and dosing regimens for NS in the two human studies were not identical. Similarly, the length of the intervention differed, with 8 weeks being implemented by Jangjo-Borazjani et al. [33] and 24 weeks by El-Shamy et al. [32].
The reason that El-Shamy et al. [32] selected a longer intervention period may have been the application of NS extract as hot tea, which presumably gave rise to a lower serum concentration than when given in tablet form.
The exercise protocols used in these two papers were also non-comparable. Jangjo-Borazjani et al. [33] were unable to conclude that any positive outcomes occurred with the use of NS and exercise together, whereas El-Shamy et al. [32] was clear about the beneficial effects of the combination strategy.
Animal Studies
The publication dates of the two animal studies were 2018 and 2019, and they included sample sizes of between 32 and 35 animals. Swimming regimes [34] and treadmill running [35] were utilised as physical activity programs alongside NS. In both animal studies, 8- to 11-week-old male rats weighing between 208 and 244 g were included. However, the characteristics of the rodents, the physical activity regimens and the type of NS used were dissimilar between these two papers.
The length of the intervention was 4 weeks in one study [34] while it was 8 weeks in the second [35]. The treadmill used in the latter animal study had an electric grid at the back of the belt in order to persuade the rats to keep running, and the longest exercise interval was 30 min. In that study, the intensity of the session was heightened over 14 days to a peak of 25 m/min for 5 days [35].
For the rats which undertook physical exercise in the form of swimming, no force was applied, and 30 min of swimming were performed in 30-cm-deep water at a temperature of 35 °C/week for 4 days [34].
Discussion
Despite the low number of studies and the variation in the study design that evaluates the combined effects of NS with exercise on the diabetic parameters, the overall findings were encouraging and showed a significant positive beneficial impact on glycemic, lipidemic, and other metabolic-related parameters.
The absorption of glucose by the skeletal musculature during physical activity requires three stages. Firstly, the glucose is transported via the circulation to the muscle; secondly, it passes across muscle cell membranes; and finally, it undergoes intracellular phosphorylation [36]. Physical training recruits an alternative messenger pathway, potentially arising from a rise in calcium titres following nerve cell activation or as a consequence of cellular energy substrate adaptations, such as the conversion of ADP to ATP. The latter occurs following the rise in ATP utilisation via ATPase enzyme activity or the synthesis of reactive oxygen and nitrogen species [37]. Insulin and physical activities have an additive effect that substantiates the theory that they act via different pathways.
Baseline and glucose-induced insulin titres become diminished following routine physical activity sessions owing to a fall in insulin release [38]. The presence of a minimum of two intracellular processes responsible for the decline in insulin secretion has been suggested by the identification of proinsulin and glucokinase mRNA downregulation—i.e. a decrease in insulin manufacture and a fall in cellular sensitivity to glucose, respectively [39].
Whilst exercise is being carried out, insulin-dependent pathways facilitate the uptake of glucose into the exerting skeletal musculature [40].
The effects of the activity are ongoing for between 2 and 72 h; the observed fall in serum glucose levels is correlated with the duration of the physical activity and its vigour [41]. The activity of the pancreatic beta cells is promoted by habitual exercise sessions, together with enhancements in insulin sensitivity [42] and the performance of the vasculature [43]. These are all factors which improve the metabolic profile and well-being of individuals with diabetes and diminish their risk of complications.
The way in which physical exercise lowers serum glucose and lipid titres is likely to be a result of a range of factors. One factor is the form of activity undertaken—e.g. aerobic or resistance, or a combination, or yoga or Tai Chi, amongst others. Exercise parameters—e.g. degree of effort, time, how often (i.e. regular versus bout, and post-exercise routines)—are also determinants, together with whether the activity is performed on dry land or in water, and the climatic conditions. The general status of the individual, such as age, gender, body mass index (BMI) and the degree of pathological progression—i.e. early, middle or late stage—are also influential.
Most of the studies included in this review followed the recommendations relating to aerobic exercise [44].
A number of researchers have investigated the way in which physical activity enhances the lipid profile—e.g. leading to elevated HDL titres and a decline in LDL and TG concentrations. One mechanism which has been demonstrated is the heightened resistance of LDL to oxidation [45]. In individuals with diabetes, rising levels of LDL and a fall in HDL comprise risk factors for cardiovascular pathologies.
There is a propensity for LDL to accrue within the walls of the vasculature, thus precipitating cardiovascular insults [46]. In contrast, arterial lipid accretion is mitigated against by HDL-C, as these lipids promote hepatic cholesterol uptake [47]. The way in which physical activity can enhance lipid metabolism may arise as a result of alterations in the activities of lipase enzymes—e.g. lipoprotein lipase and hormone-sensitive lipase (HSL) [45]. The latter, following phosphorylation and activation, is implicated in TG lipolysis within fatty tissue, a pathway which results from a cascade of intracellular signalling processes [45]. HSL phosphorylation causes the enzyme to relocate within fatty tissue cells from the cytosol to the surfaces of intracellular lipid droplets, where it catalyses lipolysis [45, 48]. A precursor event to this is the phosphorylation of proteins—i.e. perilipins contained within the fat droplets [49]. The breakdown of lipids is inhibited by non-phospholipid perilipins, which separate HSL and intracellular lipids [49]. The concentration of fat droplets, mediated by phosphorylated perilipins, may allow HSL to come into contact with TG within the cell [49].
NS and its active components have been found to have potent antioxidant properties which potentiate their glucose-lowering action [50]. In individuals with diabetes, these characteristics may promote the maintenance of pancreatic islet cells and the cellular pathways which contribute to normoglycaemia. β-Cell preservation has been documented on histopathological assessment in many animal models of diabetes in which NS or its extracts have been administered, an observation associated with raised insulin titres [51].
In a hamster model of diabetes induced by streptozotocin (STZ), NS oil led to a reduction in glucose synthesis in the liver from antecedent gluconeogenic compounds—e.g. alanine, glycerol and lactate [52, 53]. Diminished gluconeogenesis was a potential mechanism cited in order to explain the glucose-lowering effect of NS oil in diabetes [53]. Meddah et al. postulated that glucose absorption was attenuated following the immediate in vitro suppression of electrogenic glucose absorption in intestinal cells by NS derivatives [54].
A notable enhancement of glucose uptake equivalent to that of insulin was seen in cultured cells from skeletal muscle lineages and fatty tissue cells after 18 h of administration of an ethanol extract of NS [55].
A flavonoid-like influence is a further suggested mechanism of action—e.g. in in vitro cultures, mirstin affects lipogenesis and the passage of glucose across adipose tissue cell membranes [56]. This compound is likely to exert a specific effect on glucose absorption activation via membrane lipid layer fluidity or alteration in glucose transport; it is, therefore, advantageous to individuals with diabetes, leading to a fall in serum glucose levels and weight reduction, which can be ascribed to the flavonoids contained in NS [57].
Reported evidence showed that NS supplementation diminishes serum glucose titres through a number of mechanisms involving changes in sensitivity to insulin, inhibition of intestinal amylase, suppression of electrogenic adsorption of glucose within the intestine, stimulation of the AMP-activated adenosine monophosphate-activated protein kinase (AMPK) pathway and muscular glucose transporter 4 amplification as a result of elevated acetyl-CoA carboxylase phosphorylation [57, 58].
Downregulation of the expression of glucose-6-phosphatase and fructose 1,6-bisphosphatase, which function as gluconeogenic enzymes, and a reduction in the manufacture of glucose by the liver can occur following the administration of thymoquinone [59]. This compound also leads to the activation of the AMPK pathway in skeletal muscle and the liver, and the suppression of gluconeogenesis [60]. Research relating to gene expression has demonstrated that NS operates as a peroxisome proliferator-activated receptor gamma (PPAR-gamma) gene agonist and can amplify its activity [61]. Studies evaluating glucose absorption have indicated that the sodium-glucose cotransporter is suppressed by NS, a phenomenon that may be mediated via the inhibitory actions of its polyphenol constituents [54].
The way in which NS influences lipid metabolism is not yet clear. Nevertheless, the modes of action proposed in earlier studies could be implicated as both primary and secondary mechanisms. It has been postulated that NS reduces cholesterol titres via the promotion of cholesterol elimination via the bile duct, through the suppression of its manufacture within liver cells, or by a combination of the two [62]. Additionally, NS may inhibit the absorption of cholesterol within the small bowel [62].
A further potential way in which NS optimises serum lipids is via its unsaturated fatty acid components, which include palmitoleic, oleic and linoleic acids in proportions of 10%, 20% and 50–60%, respectively [63]. Additionally, polyunsaturated fatty acids, together with phytosterols (e.g. beta-sitosterol), may contribute to the cholesterol-reducing properties of NS [64].
The abnormal lipid profile frequently linked with T2DM could also be improved through the reduction in IR seen as a result of the antioxidant properties of NS [65].
The principal mechanism underlying the generation of increased IR is now considered to be the presence of oxygen species (OS) and reactive oxygen species [66]. Thus, the antioxidant characteristics of NS have the potential to suppress the peroxidation of lipids and to enhance the activity of enzymes which contribute to the breakdown of lipids [65]. A number of studies have documented the glucose-lowering properties of NS and its efficacy in enhancing lipid profiles, and have associated these effects with its antioxidant traits [67, 68]. These natural products may exert a prophylactic influence with respect to lipid abnormalities via a reduction in the activity of HMG-CoA reductase within the liver, the amplification of esterase activities, and their governing actions on genes that regulate the breakdown of cholesterol and antioxidant pathways [69]. Furthermore, the promotion of a normoglycaemic status and more favourable lipid ratios are linked with weight reduction, the latter potentially occurring as a result of an impact of NS on appetite suppression and, consequently, on energy intake, which in turn enhances serum glucose and lipid parameters.
Conclusion
The critique of the small number of studies included in the review pertaining to the combined use of NS and physical activity (aerobic or resistance) in subjects with T2DM may be a valuable addition to conventional diabetic therapy. The mechanism of the effect of NS supplement and training on glucose breakdown is due to insulin-like muscle contractions that simultaneously send a large amount of glucose with the components of NS into the cell. In addition, muscle contraction potential increases transmembrane glucose permeability and increases the amount of GLUT4 in trained muscles stimulated with NS components, which improves insulin function.
Limitations
Some of the limitations of this systematic review are notable and must be considered. The present review included studies that utilised different durations and doses of NS as well as exercise protocols. Besides that, only a small number of relevant studies have been included, and these studies have research design limitations and limited phytochemical characterisation of the NS preparations used. Thus, the present findings on the efficacies of supplementation of NS in combination with exercise should be interpreted with caution.
References
Akhter S. Low to no cost remedies for the management of diabetes mellitus; global health concern. J Diabetes Metab Disord. 2021;20:951–62.
Kahn BB. Type 2 diabetes: when insulin secretion fails to compensate for insulin resistance. Cell. 1998;92:593–6.
Al-Hariri MT, Alhefnawi MAM, Istanbouli MJ, Algehlani WAG. Glycemic control X invisible risk factors among diabetic patients in Eastern Province. GJHS. 2019;11:38.
AlKhafaji D, Al Argan R, Albahrani S, Al Elq A, Albaker W, Al-Hariri M, et al. The impact of obesity on hospitalized patients with COVID-19 infection in the Eastern Province of Saudi Arabia. J Med Life. 2022;15:425–32.
Al-Hariri MT, Al-Hattami A. Extracurricular activities amongst health colleges students at the Imam Abdulrahman Bin Faisal University. GJHS. 2018;10:105.
Al-Hariri M. Life style activities and feeling state responses in community sample of diabetic patients in Eastern Providence Saudi Arabia. J Diabetes Metab Disord. 2019;18:301–5.
Sidawi B, Al-Hariri MT. The importance of healthy and sustainable built environments for vulnerable groups of population. Built Environ J (BEJ). 2015;12:24–32.
Albaker WI, Al-Hariri MT, Al Elq AH, Alomair NA, Alamoudi AS, Voutchkov N, Beneficial effects of adding magnesium to desalinated drinking water on metabolic and insulin resistance parameters among patients with type 2 diabetes mellitus: a randomized controlled clinical trial. npj Clean Water. https://www.nature.com/articles/s41545-022-00207-9. Cited 23 Nov 2022.
Stratton IM, Adler AI, Neil HAW, Matthews DR, Manley SE, Cull CA, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ Brit Med J. 2000;321:405–12.
Al-Hariri M. The pathophysiological mechanisms of diabetic otopathy. J Taibah Univ Med Sci. 2016;11:401–3.
Al-Hariri MT, Al-Enazi AS, Alshammari DM, Bahamdan AS, Al-Khtani SM, Al-Abdulwahab AA. Descriptive study on the knowledge, attitudes and practices regarding the diabetic foot. J Taibah Univ Med Sci. 2017;12:492–6.
Al-Hariri M, Eldin TG, Abu-Hozaifa B, Elnour A. Glycemic control and anti-osteopathic effect of propolis in diabetic rats. Diabetes Metab Syndr Obes. 2011;4:377–84.
Simó R, Ciudin A, Simó-Servat O, Hernández C. Cognitive impairment and dementia: a new emerging complication of type 2 diabetes—the diabetologist’s perspective. Acta Diabetol. 2017;54:417–24.
Al-Hariri M. Sweet bones: the pathogenesis of bone alteration in diabetes. J Diabetes Res. 2016;2016:6969040.
Satin LS, Soleimanpour SA, Walker EM. New aspects of diabetes research and therapeutic development. Pharmacol Rev. 2021;73:1001–15.
Saad B, Kmail A, Haq SZH. Anti-diabesity Middle Eastern medicinal plants and their action mechanisms. Evid Based Complement Alternat Med. 2022;2022:2276094.
Panicker GK, Karnad DR, Salvi V, Kothari S. Cardiovascular risk of oral antidiabetic drugs: current evidence and regulatory requirements for new drugs. J Assoc Physicians India. 2012;60:56–61.
Al-Hariri M, Eldin TG, Al-Harbi M, Hashim T, Ahmad R. Effect of propolis administration on the endocrine functions and histopathology of pancreas in streptozotocin-induced diabetic rats. Adv Sci Eng Med. 2019;11:1155–60.
Alshahri BK, Bamashmoos M, Alnaimi MI, Alsayil S, Basager S, Al-Hariri MT, et al. Assessment of self-management care and glycated hemoglobin levels among type 2 diabetes mellitus patients: a cross-sectional study from the Kingdom of Saudi Arabia. Cureus. 2020;12. https://www.cureus.com/articles/44913-assessment-of-self-management-care-and-glycated-hemoglobin-levels-among-type-2-diabetes-mellitus-patients-a-cross-sectional-study-from-the-kingdom-of-saudi-arabia. Cited 23 Nov 2022.
Albaker WI, Al-Hariri MT, Al Elq AH, Alomair NA, Alamoudi AS, Voutchkov N, et al. Beneficial effects of adding magnesium to desalinated drinking water on metabolic and insulin resistance parameters among patients with type 2 diabetes mellitus: a randomized controlled clinical trial. NPJ Clean Water. 2022;5:63.
Mohamed AA, Jan Y-K. Effect of adding proprioceptive exercise to balance training in older adults with diabetes: a systematic review. Curr Diabetes Rev. 2020;16:327–39.
Al-Hariri M. Sweet treats sweet: a review of antidiabetic properties of honey. GJHS. 2018;10:94.
Dereli EE, Yaliman A. Comparison of the effects of a physiotherapist-supervised exercise programme and a self-supervised exercise programme on quality of life in patients with Parkinson’s disease. Clin Rehabil. 2010;24:352–62.
Erlich AT, Tryon LD, Crilly MJ, Memme JM, Moosavi ZSM, Oliveira AN, et al. Function of specialized regulatory proteins and signaling pathways in exercise-induced muscle mitochondrial biogenesis. Integr Med Res. 2016;5:187–97.
Miko H-C, Zillmann N, Ring-Dimitriou S, Dorner TE, Titze S, Bauer R. Effects of physical activity on health. Gesundheitswesen. 2020;82:S184–95.
Al-Hariri MT, Yar T, Bamosa AO, El-Bahai MN. Effects of two-months Nigella sativa supplementation on cardiac hemodynamics and adrenergic responsiveness. J Pak Med Assoc. 2009;59:5.
Gholamnezhad Z, Havakhah S, Boskabady MH. Preclinical and clinical effects of Nigella sativa and its constituent, thymoquinone: a review. J Ethnopharmacol. 2016;190:372–86.
Yar T, El-Hariri M, El-Bahai MN, Bamosa AO. Effects of Nigella sativa supplementation for one month on cardiac reserve in rats. Indian J Physiol Pharmacol. 2008;52:141–8.
El-Bahai MN, Al-Hariri MT, Yar T, Bamosa AO. Cardiac inotropic and hypertrophic effects of Nigella sativa supplementation in rats. Int J Cardiol. 2009;131:e115-117.
Badar A, Kaatabi H, Bamosa A, Al-Elq A, Abou-Hozaifa B, Lebda F, et al. Effect of Nigella sativa supplementation over a one-year period on lipid levels, blood pressure and heart rate in type-2 diabetic patients receiving oral hypoglycemic agents: nonrandomized clinical trial. Ann Saudi Med. 2017;37:56–63.
Al-Khalifa KS, AlSheikh R, Al-Hariri MT, El-Sayyad H, Alqurashi MS, Ali S, et al. Evaluation of the antimicrobial effect of thymoquinone against different dental pathogens: an in vitro study. Molecules. 2021;26:6451.
El-Shamy KA, Mosa MMA, El-Nabarawy SK, El-Qattan GM. Effect of Nigella sativa tea in type 2-diabetic patients as regards glucose homeostasis, liver and kidney functions. J Appl Sci Res. 2011;7:2524–34.
Jangjo-Borazjani S, Dastgheib M, Kiyamarsi E, Jamshidi R, Rahmati-Ahmadabad S, Helalizadeh M, et al. Effects of resistance training and nigella sativa on type 2 diabetes: implications for metabolic markers, low-grade inflammation and liver enzyme production. Arch Physiol Biochem 2021;1–9.
Hosseini SA, Zar A, Dehghani Z. Lipid lowering effects of Nigella sativa and swimming training in streptozotocin induced diabetic rats. Ann Mil Health Sci Res. 16. https://brieflands.com/articles/amhsr-84153.html. Cited 11 Dec 2022.
Babaei Bonab S, Tofighi A. Effect of 8 weeks aerobic training and Nigella supplement on insulin resistance, lipid profile and plasma level of HbA1c in type 2 diabetic rats. J Adv Med Biomed Res. 2019;27:20–9.
Sylow L, Kleinert M, Richter EA, Jensen TE. Exercise-stimulated glucose uptake—regulation and implications for glycaemic control. Nat Rev Endocrinol. 2017;13:133–48.
Miotto PM, Holloway GP. Exercise-induced reductions in mitochondrial ADP sensitivity contribute to the induction of gene expression and mitochondrial biogenesis through enhanced mitochondrial H2O2 emission. Mitochondrion. 2019;46:116–22.
Wahid A, Manek N, Nichols M, Kelly P, Foster C, Webster P, et al. Quantifying the association between physical activity and cardiovascular disease and diabetes: a systematic review and meta-analysis. J Am Heart Assoc. 2016;5: e002495.
Ayer A, Fazakerley DJ, James DE, Stocker R. The role of mitochondrial reactive oxygen species in insulin resistance. Free Radic Biol Med. 2022;179:339–62.
Pereira RM, Moura LP de, Muñoz VR, Silva ASR da, Gaspar RS, Ropelle ER, et al. Molecular mechanisms of glucose uptake in skeletal muscle at rest and in response to exercise. Motriz: Rev Educ Fis. 2017;23. http://www.scielo.br/j/motriz/a/xQwdfk3DWVGcxX4cxqkXT9s/abstract/?lang=en. Cited 23 Nov 2022.
Gaesser GA. Exercise for prevention and treatment of cardiovascular disease, type 2 diabetes, and metabolic syndrome. Curr Diab Rep. 2007;7:14–9.
Colberg SR, Sigal RJ, Yardley JE, Riddell MC, Dunstan DW, Dempsey PC, et al. Physical activity/exercise and diabetes: a position statement of the American Diabetes Association. Diabetes Care. 2016;39:2065–79.
Hayashino Y, Jackson JL, Fukumori N, Nakamura F, Fukuhara S. Effects of supervised exercise on lipid profiles and blood pressure control in people with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Diabetes Res Clin Pract. 2012;98:349–60.
Snowling NJ, Hopkins WG. Effects of different modes of exercise training on glucose control and risk factors for complications in type 2 diabetic patients: a meta-analysis. Diabetes Care. 2006;29:2518–27.
Delevatti R, Marson E, Kruel LF. Effect of aquatic exercise training on lipids profile and glycaemia: a systematic review. Rev Andal Med Deport. 2015;8:163–70.
Schlader Z, Charkoudian N. Review of advanced environmental exercise physiology, 2/E, by Cheung and Ainslie. Am J Physiol Regul Integr Comp Physiol. 2021;321:R139–40.
Bermon S, Castell LM, Calder PC, Bishop NC, Blomstrand E, Mooren FC, et al. Consensus statement immunonutrition and exercise. Exerc Immunol Rev. 2017;23:8–50.
Wang H, Hu L, Dalen K, Dorward H, Marcinkiewicz A, Russell D, et al. Activation of hormone-sensitive lipase requires two steps, protein phosphorylation and binding to the PAT-1 domain of lipid droplet coat proteins. J Biol Chem. 2009;284:32116–25.
Miyoshi H, Souza SC, Zhang H-H, Strissel KJ, Christoffolete MA, Kovsan J, et al. Perilipin promotes hormone-sensitive lipase-mediated adipocyte lipolysis via phosphorylation-dependent and -independent mechanisms. J Biol Chem. 2006;281:15837–44.
Ahmad A, Husain A, Mujeeb M, Khan SA, Najmi AK, Siddique NA, et al. A review on therapeutic potential of Nigella sativa: a miracle herb. Asian Pac J Trop Biomed. 2013;3:337–52.
Hamdan A, Haji Idrus R, Mokhtar MH. Effects of Nigella sativa on type-2 diabetes mellitus: a systematic review. Int J Environ Res Public Health. 2019;16:4911.
Fararh KM, Shimizu Y, Shiina T, Nikami H, Ghanem MM, Takewaki T. Thymoquinone reduces hepatic glucose production in diabetic hamsters. Res Vet Sci. 2005;79:219–23.
Fararh KM, Atoji Y, Shimizu Y, Shiina T, Nikami H, Takewaki T. Mechanisms of the hypoglycaemic and immunopotentiating effects of Nigella sativa L. oil in streptozotocin-induced diabetic hamsters. Res Vet Sci. 2004;77:123–9.
Meddah B, Ducroc R, El Abbes FM, Eto B, Mahraoui L, Benhaddou-Andaloussi A, et al. Nigella sativa inhibits intestinal glucose absorption and improves glucose tolerance in rats. J Ethnopharmacol. 2009;121:419–24.
Benhaddou-Andaloussi A, Martineau LC, Spoor D, Vuong T, Leduc C, Joly E et al. Antidiabetic activity of Nigella sativa seed extract in cultured pancreatic β-cells, skeletal muscle cells, and adipocytes. Pharmaceut Biol. 2008;46. https://doi.org/10.1080/13880200701734810. Cited 23 Nov 2022.
American Society of Plant Biologists. Medicinal plants: past, present and future. Plant Cell. 2013;25:tpc.112.tt0113.
Qidwai W, Hamza HB, Qureshi R, Gilani A. Effectiveness, safety, and tolerability of powdered Nigella sativa (kalonji) seed in capsules on serum lipid levels, blood sugar, blood pressure, and body weight in adults: results of a randomized, double-blind controlled trial. J Altern Complement Med. 2009;15:639–44.
Soleimani-Dodran M, Alipanah-Moghadam R, Jeddi F, Babaei M, Salimnejad R, Bahreini E. Effect of hydroalcoholic seed extract of Nigella sativa on hepatic and pancreatic factors of Nrf2 and FGF21 in the regulation of insulin transcription factors of MafA and PDX-1 in streptozotocin-treated diabetic rats. Nutr Metab. 2022;19:64.
Sankaranarayanan C, Pari L. Thymoquinone ameliorates chemical induced oxidative stress and β-cell damage in experimental hyperglycemic rats. Chem Biol Interact. 2011;190:148–54.
Ong KW, Hsu A, Tan BKH. Anti-diabetic and anti-lipidemic effects of chlorogenic acid are mediated by AMPK activation. Biochem Pharmacol. 2013;85:1341–51.
Abdallah HM, El Dine RS, Mohamed GA, Ibrahim SRM, Shehata IA, El-Halawany AM. Natural peroxisome proliferator-activated receptor γ (PPARγ) activators for diabetes. Altern Ther Health Med. 2020;26:28–44.
Sahebkar A, Beccuti G, Simental-Mendía LE, Nobili V, Bo S. Nigella sativa (black seed) effects on plasma lipid concentrations in humans: a systematic review and meta-analysis of randomized placebo-controlled trials. Pharmacol Res. 2016;106:37–50.
Mollazadeh H, Hosseinzadeh H. The protective effect of Nigella sativa against liver injury: a review. Iran J Basic Med Sci. 2014;17:958–66.
Sabzghabaee AM, Dianatkhah M, Sarrafzadegan N, Asgary S, Ghannadi A. Clinical evaluation of Nigella sativa seeds for the treatment of hyperlipidemia: a randomized, placebo controlled clinical trial. Med Arch. 2012;66:198.
Shen HH, Peterson SJ, Bellner L, Choudhary A, Levy L, Gancz L, et al. Cold-pressed Nigella sativa oil standardized to 3% thymoquinone potentiates omega-3 protection against obesity-induced oxidative stress, inflammation, and markers of insulin resistance accompanied with conversion of white to beige fat in mice. Antioxidants. 2020;9:489.
Ge X, Liu Z, Qi W, Shi X, Zhai Q. Chromium (VI) induces insulin resistance in 3T3-L1 adipocytes through elevated reactive oxygen species generation. Free Radical Res. 2008;42:554–63.
Mathur ML, Gaur J, Sharma R, Haldiya KR. Antidiabetic properties of a spice plant Nigella sativa. J Endocrinol Metab. 2011. http://www.jofem.org/index.php/jofem/article/view/15. Cited 25 Nov 2022.
Butt MS, Sultan MT. Nigella sativa: reduces the risk of various maladies. Crit Rev Food Sci Nutr. 2010;50:654–65.
Ali S, Hashim A, Shiekh A, Majid S, Rehman MU. Chapter 10—The cardioprotective effect of thymoquinone from Nigella sativa. In: Khan A, Rehman M, editors. Black seeds (Nigella Sativa). Amsterdam: Elsevier; 2022. p. 239–52. https://www.sciencedirect.com/science/article/pii/B9780128244623000068. Cited 25 Nov 2022.
Acknowledgements
Funding
The study and journal’s Rapid Service Fee was funded by the authors.
Authorship
Haider H. Al-Yammi and Mohammed T. Al-Hariri meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authors’ Contribution
Haider H. Al-Yammi and Mohammed T. Al-Hariri have contributed equally to this manuscript.
Disclosures
Haider H. Al-Yammi and Mohammed T. Al-Hariri have no conflicts of interest to declare.
Compliance with Ethics Guidelines
This review is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
The study data are available from the corresponding author upon reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Alyami, H.H., Al-Hariri, M.T. Synergistic Effects of Nigella sativa and Exercise on Diabetic Profiles: A Systematic Review. Diabetes Ther 14, 467–478 (2023). https://doi.org/10.1007/s13300-022-01362-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-022-01362-5