Abstract
Introduction
We evaluated the efficacy and safety of ultra-rapid lispro (URLi) in comparison to lispro in a subgroup analysis of Japanese adults with type 1 diabetes mellitus from the phase 3 PRONTO-T1D trial.
Methods
After an 8-week lead-in to optimize basal insulin treatment, patients were randomized to 52-week double-blind mealtime URLi or lispro, or 26-week open-label postmeal URLi. The primary endpoint was change in hemoglobin A1c (HbA1c) from baseline (week 0) to week 26 between mealtime URLi and lispro. The multiplicity adjusted objectives were 1- and 2-h postprandial glucose (PPG) excursions after a meal test between mealtime URLi and lispro, and change in HbA1c from baseline to week 26 between postmeal URLi and mealtime lispro.
Results
This manuscript presents pre-specified exploratory analyses of 26-week data from Japanese patients randomized to double-blind URLi (n = 62) or lispro (n = 59), or open-label URLi (n = 46). Mean baseline HbA1c levels were 7.52% for mealtime URLi, 7.44% for lispro, and 7.51% for postmeal URLi at randomization. At week 26, the least squares mean (LSM) difference compared to lispro was 0.04% (95% confidence interval [CI] − 0.14 to 0.22) for mealtime URLi, and 0.16% (95% CI − 0.04 to 0.35) for postmeal URLi. In comparison to lispro, mealtime URLi resulted in statistically significantly lower 1- and 2-h PPG excursions during the mixed-meal tolerance test. LSM differences were − 40.5 mg/dL, 95% CI − 59.5 to 21.4 (− 2.25 mmol/L, 95% CI − 3.3 to − 1.2) for 1-h PPG excursions and − 51.7 mg/dL, 95% CI − 81.7 to − 21.8 (− 2.87 mmol/L, 95% CI − 4.5 to − 1.2) for 2-h PPG excursions at week 26. There were no significant treatment differences in rates of severe/overall hypoglycemia, or incidence of treatment-emergent adverse events.
Conclusions
Mealtime and postmeal URLi provide effective and comparable glycemic control in Japanese patients. Mealtime URLi demonstrated more effective PPG control compared to lispro.
Trial Registration
ClinicalTrials.gov, NCT03214367.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
For patients with type 1 diabetes mellitus (T1DM), reaching and maintaining glycemic goals (including postprandial glucose) remains challenging. |
In Japan, there is a medical need for the development and approval of faster rapid-acting insulins, including those which can be administered postmeal. |
We evaluated the efficacy and safety of ultra-rapid lispro (URLi) in comparison to Humalog® (lispro) in Japanese patients with T1DM in a phase 3, randomized, subpopulation analysis. |
What was learned from the study? |
URLi and lispro provide effective and comparable overall glycemic control. |
URLi reduced postprandial glucose excursions in comparison to lispro in Japanese patients with T1DM and is well tolerated. |
Introduction
For people with diabetes, adequate blood glucose control is important for preventing diabetic complications and cardiovascular disease and for reducing the medical and public health burden [1]. Hemoglobin A1c (HbA1c) is the current standard for measuring overall glucose exposure and is the primary variable used in registration studies (US Food and Drug Administration, European Medicines Agency, and Ministry of Health Labour and Welfare guidelines), providing a combined measurement of both fasting and postprandial glycemic control [2, 3]. For patients with type 1 diabetes mellitus (T1DM), achieving and maintaining glycemic goals remains challenging, with many patients experiencing difficulties [4, 5] reaching the target HbA1c of less than 7% [4]. In Japan, the mean HbA1c of patients with T1DM is 7.8% [6].
Control of both fasting and postprandial hyperglycemia is essential to reach HbA1c goals. It has been demonstrated that postprandial hyperglycemia contributes to overall glucose control as assessed by HbA1c, and increases the risk of micro- and macrovascular complications in patients with diabetes [7,8,9,10,11], although the association of postprandial glucose (PPG) with complications in T1DM is much less supported by evidence compared with type 2 diabetes mellitus [12]. Control of postprandial hyperglycemia continues to be a challenge [13] in the overall treatment of patients with T1DM, with increased PPG levels significantly associated with increased healthcare resource utilization [14].
Current rapid-acting insulins were designed to control PPG excursions [15,16,17,18]. Although these insulins have a more rapid onset of action compared to human insulins, they are not able to match the speed of physiological insulin secretion, leaving many patients unable to achieve optimal glycemic control [19]. Newly developed ultra-rapid-acting insulin formulations, such as ultra-rapid lispro (URLi), were designed to more closely match the physiological insulin secretory response to meals, characteristic of individuals without diabetes. Lispro (Humalog®) is a commercially available, rapid-acting human insulin analogue indicated to improve glycemic control in patients with diabetes mellitus [20]. URLi (LY900014) is a newly developed insulin lispro formulation utilizing two enabling excipients, treprostinil and citrate, with independent mechanisms to accelerate the absorption of insulin lispro. Treprostinil is used in microdose quantities in the formulation of URLi, but is not detectable in clinical doses in the systemic circulation, nor associated with systemic effects. URLi was formulated to more closely match the physiological prandial insulin secretion pattern, and is expected to more effectively control PPG excursions in comparison to lispro.
PRONTO-T1D was a recent, prospective, randomized, double-blind, phase 3 trial in adults with T1DM [21]. PRONTO-T1D compared double-blind mealtime URLi to lispro with an open-label postprandial URLi treatment group in combination with insulin glargine or insulin degludec. This pivotal study demonstrated that URLi was non-inferior to lispro for change in HbA1c following 26 weeks of treatment [21]. In a recent survey, Japanese patients with diabetes reported moderate to severe burden in multiple aspects of their lives associated with current mealtime insulin timing [22]. Specifically, 8.9% of patients reported that their normal mealtime insulin injection timing was after the start of a meal in the past month. Also, the average percentage of time that patients inject after the start of a meal in the past month was 11.0% [22]. The main reasons for dosing during or after a meal were lifestyle-related, such as eating or working outside of home, or not being able to find a suitable place to inject [22,23]. Unlike the USA and Europe, there are limited rapid-acting insulins approved for postmeal administration in Japan, and none were approved at the time this study commenced. At the time this manuscript was submitted, fast-acting insulin aspart (Fiasp®) was the only rapid-acting insulin approved for postmeal use in Japan (approved September 2019). Hence, there has been a medical need for the development and approval of both postprandial and mealtime products. We describe a Japanese subpopulation analysis of the PRONTO-T1D trial. The aim of this report is to evaluate the efficacy and safety of URLi, administered at mealtime and postmeal, in comparison to mealtime lispro in Japanese patients with T1DM.
Methods
Study Participants
The study design has been previously described [20]. Briefly, adults at least 18 years old with a clinical diagnosis of T1DM (based on World Health Organization [WHO] classification) [24] for at least 1 year prior to screening were eligible for participation. Participants must have been treated with rapid-acting insulin for at least 90 days and basal insulin for at least 30 days prior to screening, with an HbA1c between 7.0 and 9.5% (53–80.32 mmol/mol).
Exclusion criteria included hypoglycemia unawareness as judged by an investigator, more than one instance of severe hypoglycemia requiring assistance, or hyperglycemic hyperosmolar state within 6 months prior to screening.
The study was conducted in accordance with the International Council for Harmonization Guidelines for Good Clinical Practice including the Declaration of Helsinki and its later amendments and the Council for International Organisation of Medical Sciences International Ethical Guidelines. An ethical review board at each site (Kobori Clinic, H.E.C. Science Clinic, Jinnouchi Hospital, Heiwadai Hospital, The Institute for Adult Diseases, Naka Memorial Clinic, Tomonaga Clinic, Nihonbashi Sakura Clinic, AMC Nishi-umeda Clinic, Manda Hospital, Fukuoka Red Cross Hospital, Shin Akasaka Clinic, Shinagawa East One Medical Clinic, Sasebo Chuo Hospital, Okuguchi Clinic of Internal Medicine, Sapporo Skin Clinic, Takatsuki Red Cross Hospital, Osaka City University Medical School Hospital, Goshogatani Home Clinic, Tokyo Women's Medical University Hospital) approved all procedures, and all subjects provided written, informed consent.
Study Design
PRONTO-T1D was a phase 3, prospective, randomized, double-blind, outpatient, multinational, multicenter, three treatment group, parallel, active-controlled study, and was conducted in patients with T1DM. In two of the treatment groups, URLi and lispro were administered immediately (0–2 min) prior to each meal in a double-blind manner. A third open-label treatment group was administered URLi at 20 min after the start of the meal (postmeal). The study included a 1-week screening period and an 8-week lead-in period prior to randomization. This was followed by 52 weeks of treatment and a 4-week safety follow-up period. For patients in the open-label treatment group (postmeal URLi), the treatment period ended after 26 weeks, except for Japanese patients where a 52-week treatment period was used. We report the results of the first 26-week treatment period (the pre-specified primary endpoint) in a subpopulation analysis of Japanese patients from the PRONTO-T1D trial. The study design is outlined in Fig. 1.
Study design. Following a 1-week screening period and an 8-week lead-in period, Japanese patients were randomized in a 4:4:3 ratio to receive mealtime URLi, mealtime lispro, or postmeal URLi, in combination with insulin degludec or glargine. All patients underwent a 4-h MMTT at baseline (week 0, visit 8) and at the end of the primary treatment period (week 26, visit 18). MMTT mixed-meal tolerance test, N number of patients, URLi ultra-rapid lispro
Study Interventions and Randomization
Patients treated with insulin aspart or insulin glulisine as their pre-study prandial insulin were switched to lispro (unit for unit) at the start of the lead-in period. Prandial insulin doses were not changed during the lead-in period except for safety reasons or to facilitate basal insulin optimization.
Patients were randomized in a 4:4:3 ratio to one of the following treatment groups: double-blind URLi 100 U/mL at mealtime, double-blind insulin lispro 100 U/mL at mealtime, or open-label URLi 100 U/mL administered at 20 min postmeal.
Following randomization, the prandial insulin dose was adjusted as necessary during the initial 12 weeks in a treat-to-target manner in order to meet target self-monitored blood glucose (SMBG) levels. Prandial insulin dose was determined for each participant using either the pattern adjustment method or the carbohydrate counting method. The correction factor was implemented with either prandial insulin dosing plan. For patients using the carbohydrate counting method, prandial insulin dosing was calculated on the basis of the estimated carbohydrate content of the meal as units of insulin per grams carbohydrate. The insulin-to-carbohydrate ratio (ICR) and correction factor were assessed at least weekly in order to meet target SMBG levels. When the pattern adjustment plan was used, patients were prescribed a fixed dose or dose range of insulin for each meal, with the fixed insulin dose individualized for each meal, and the prandial insulin dose and the correction factor were assessed and adjusted at least weekly. During the maintenance period (weeks 12–26 of treatment), adjustments to prandial and basal insulin doses were only performed to maintain glycemic control, or for safety reasons such as hypoglycemia or unacceptable hyperglycemia.
Standardized Meal Test
All patients underwent a 4-h mixed-meal tolerance test (MMTT) at baseline and at the end of the 26-week treatment period. The MMTT meal consisted of a liquid nutrition shake with approximately 700 kcal including 100 g of carbohydrate, and was required to be consumed within 15 min.
Self-Monitored Blood Glucose
All study participants were instructed to perform three sets of 10-point SMBG profiles prior to five visits (baseline and weeks 4, 8, 12, and 26). Participants were also instructed to measure a minimum of four SMBG readings daily (morning, midday, and evening premeal, and pre-bedtime), with additional SMBG readings as needed for glucose self-management.
Study Endpoints
The primary endpoint was the change in HbA1c from baseline to week 26. Key secondary endpoints were 1- and 2-h PPG excursions during the MMTT. Other secondary endpoints included the percentage of patients who achieved the target HbA1c, 10-point SMBG values, and safety measurements.
Statistical Analysis
The analyses have already been described for the overall population [21]. These methods were applied to the Japanese population in a pre-specified subset analysis, although statistical power was not taken into consideration, and p values are provided as a reference. Briefly, analyses for efficacy parameters and hypoglycemia were conducted utilizing data collected during the treatment period prior to discontinuation of study insulin. Analyses for other safety parameters were conducted utilizing all available data.
Hypoglycemia, an episode requiring assistance due to neurological impairment as confirmed by the investigator, was summarized by rate and incidence. For other categories of hypoglycemia, rate and incidence of events were analyzed using a negative binomial regression model and a logistic regression model respectively, using both no greater than 70 mg/dL (3.9 mmol/L) and less than 54 mg/dL (3.0 mmol/L) glucose thresholds.
Results
Study Participants in Japanese Subpopulation
The Japanese population comprised 167 of the 1222 randomized patients in the overall population. Of the 167 Japanese patients, 59 were randomized to lispro, 62 were randomized to mealtime URLi, and 46 were randomized to postmeal URLi. Three Japanese patients discontinued study treatment: two patients in the mealtime URLi treatment arm due to patient withdrawal, and one patient from the postmeal URLi treatment arm due to pregnancy. Baseline patient characteristics are outlined in Table 1. Overall, baseline characteristics were similar between treatment groups.
Efficacy
HbA1c
The primary study endpoint was the change in HbA1c from baseline to week 26. For the overall population, non-inferiority of both mealtime and postmeal URLi to lispro was confirmed by the change from baseline to week 26 in HbA1c [21] (the upper 95% confidence interval [CI] of LSM difference was lower than 0.4%, defined as the pre-specified non-inferiority criteria for the overall population).
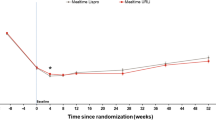
In Japanese patients, the mean HbA1c at study entry was 7.98% (63.8 mmol/mol) for mealtime URLi, 7.91% (62.9 mmol/mol) for lispro, and 7.95% (63.4 mmol/mol) for postmeal URLi. Mean HbA1c improved during the lead-in period to a baseline value of 7.52% (58.7 mmol/mol) for mealtime URLi, 7.44% (57.8 mmol/mol) for lispro, and 7.51% (58.6 mmol/mol) for postmeal URLi. The LSM change from baseline to week 26 for each treatment was − 0.09% (− 1.0 mmol/mol) for mealtime URLi, − 0.13% (− 1.4 mmol/mol) for lispro, and 0.02% (0.3 mmol/mol) postmeal URLi. At week 26, the LSM difference between mealtime URLi and lispro was 0.04%, 95% CI − 0.14 to 0.22 (0.4 mmol/mol, 95% CI − 1.6 to 2.4), and 0.16%, 95% CI − 0.04 to 0.35 (1.7 mmol/mol, 95% CI − 0.4 to 3.8) between postmeal URLi and lispro. HbA1c levels at baseline and at weeks 4, 8, 12, and 26 are shown in Fig. 2.
HbA1c levels during the lead-in period and from baseline to week 26 (% and mmol/mol) in Japanese patients for mealtime URLi, lispro, and postmeal URLi. Data are mean at study entry and LSM ± SE at all other timepoints. CI confidence interval, HbA1c hemoglobin A1c, LSM least squares mean, SE standard error, URLi ultra-rapid lispro
The number of patients achieving the HbA1c target of less than 7% at week 26 was 12/60 (20.0%) for mealtime URLi, 16/59 (27.1%) for lispro, and 6/45 (13.3%) for postmeal URLi (odds ratio [OR] mealtime URLi vs. lispro 0.66, 95% CI 0.23 to 1.88; postmeal URLi vs. lispro 0.43, 95% CI 0.14 to 1.33). The number of patients achieving the HbA1c target of no greater than 6.5% at week 26 was 5/60 (8.3%) for mealtime URLi, 8/59 (13.6%) for lispro, and 1/45 (2.2%) for postmeal URLi (OR mealtime URLi vs. lispro 0.50, 95% CI 0.14 to 1.82; postmeal URLi vs. lispro 0.17, 95% CI 0.02 to 1.33).
Meal Test/Postprandial Glucose Excursions
In Japanese patients, treatment with mealtime URLi resulted in statistically significantly lower 1- and 2-h PPG excursions during the MMTT at week 26 in comparison to lispro. LSM differences were − 40.5 mg/dL, 95% CI − 59.5 to − 21.4 (− 2.25 mmol/L, 95% CI − 3.3 to − 1.2) for 1-h PPG excursions and − 51.7 mg/dL, 95% CI − 81.7 to − 21.8 (− 2.87 mmol/L, 95% CI − 4.5 to − 1.2) for 2-h PPG excursions. A statistically significant reduction in PPG excursions was also evident at other timepoints, ranging from 30 min to 4 h (Fig. 3).
Postprandial glycemic excursions during MMTT at week 26 in Japanese patients. PPG excursions (mg/dL) following mealtime URLi, lispro, or postmeal URLi treatment at timepoints ranging from 15 min to 4 h during an MMTT. Data are LSM ± SE. LSM least squares mean, MMTT mixed-meal tolerance test, PPG postprandial glucose, SE standard error, URLi ultra-rapid lispro
In Japanese patients, there were no statistically significant differences in PPG excursions during the MMTT between postmeal URLi and lispro at 1 h (LSM differences of 11.1 mg/dL, 95% CI − 9.4 to 31.6 [0.61 mmol/L, 95% CI − 0.5 to 1.8]) and 2 h (LSM differences of − 7.4 mg/dL, 95% CI − 39.9 to 25.1 [− 0.41 mmol/L, 95% CI − 2.2 to 1.4]) at week 26. Postmeal URLi resulted in statistically greater PPG excursions in comparison to lispro at 30 min (LSM differences of 23.1 mg/dL, 95% CI 10.4 to 35.7 [1.28 mmol/L, 95% CI 0.6 to 2.0]), but showed significantly reduced PPG excursions at 3 h (LSM difference of − 39.6 mg/dL, 95% CI − 74.7 to − 4.4 [− 2.20 mmol/L, 95% CI − 4.2 to − 0.2]). The mean insulin dose during the MMTT was similar in all treatment groups: mealtime URLi 10.68 U/mL (0.16 U/kg), lispro 9.22 U/mL (0.14 U/kg), and postmeal URLi 10.29 U/mL (0.16 U/kg). PPG excursions during the MMTT at week 26 for all treatment groups is shown in Fig. 3.
A significant reduction in the incremental area under the serum glucose concentration–time curve (iAUC), above the glucose level at the start of MMTT, was observed during the MMTT for mealtime URLi in comparison to lispro at all measured time intervals from 1 to 4 h (0–1 h, 0–2 h, 0–3 h, 0–4 h). The serum glucose iAUC at week 26 was not significantly different between postmeal URLi and lispro at any measured time interval.
Self-Monitored Blood Glucose
A time course of 10-point SMBG profiles at week 26 for all treatment groups is outlined in Fig. 4. At week 26, LSM differences for the morning postmeal glucose excursions at 1 h were − 13.9 mg/dL, 95% CI − 35.0 to 7.2 (− 0.77 mmol/L) for mealtime URLi and 11.2 mg/dL, 95% CI − 11.7 to 34.0 (0.62 mmol/L) for postmeal URLi compared to lispro. At 2 h, LSM differences were − 3.2 mg/dL, 95% CI − 25.1 to 18.6 (− 0.18 mmol/L) for mealtime URLi and 5.7 mg/dL, 95% CI − 18.6 to 30.0 (0.32 mmol/L) for postmeal URLi compared to lispro.
Time course of 10-point self-monitored blood glucose profile at week 26 in Japanese patients. SMBG profiles (mg/dL and mmol/L) following mealtime URLi, lispro, or postmeal URLi treatment at week 26 at the timepoints of morning, midday, evening, and bedtime. Data are LSM ± SE. LSM least squares mean, SE standard error, SMBG self-monitored blood glucose, URLi ultra-rapid lispro
At week 26, LSM differences for the midday postmeal glucose excursions at 1 h were − 23.6 mg/dL, 95% CI − 43.8 to − 3.4 (− 1.31 mmol/L) for mealtime URLi and 10.8 mg/dL, 95% CI − 11.3 to 32.9 (0.60 mmol/L) for postmeal URLi compared to lispro. At 2 h, LSM differences were − 20.4 mg/dL, 95% CI − 44.3 to 3.5 (− 1.13 mmol/L) for mealtime URLi and − 7.5 mg/dL, 95% CI − 33.6 to 18.5 (− 0.42 mmol/L) for postmeal URLi compared to lispro.
At week 26, LSM differences for the evening postmeal glucose excursions at 1 h were − 31.2 mg/dL, 95% CI − 53.7 to − 8.7 (− 1.73 mmol/L) for mealtime URLi and 8.9 mg/dL, 95% CI − 15.8 to 33.6 (0.50 mmol/L) for postmeal URLi compared to lispro. At 2 h, LSM differences were − 36.2 mg/dL, 95% CI − 63.6 to − 8.8 (− 2.01 mmol/L) for mealtime URLi, and − 18.7 mg/dL, 95% CI − 48.6 to 11.3 (− 1.04 mmol/L) for postmeal URLi compared to lispro.
Overall, daily average blood glucose levels in the 10-point SMBG profiles were similar between treatments, except for lower blood glucose levels for lispro compared to mealtime URLi at the midday and evening premeal timepoints; LSM differences of 19.3 mg/dL (1.07 mmol/L), p = 0.036 and 33.0 mg/dL (1.83 mmol/L), p = 0.003, respectively. In addition, postmeal URLi treatment resulted in significantly higher SMBG levels compared to lispro 1-h after the midday and evening meal; LSM differences of 27.9 mg/dL (1.55 mmol/L), p = 0.015 and 25.9 mg/dL (1.44 mmol/L), p = 0.036, respectively (Fig. 4).
Insulin Dose
The total daily basal insulin dose at baseline was 0.29 U/kg for mealtime URLi, 0.26 U/kg for postmeal URLi, and 0.27 U/kg for lispro. At week 26, daily basal insulin dose was 0.29 U/kg for mealtime URLi and 0.29 U/kg for postmeal URLi, versus 0.27 U/kg for lispro (p = 0.005 and p = 0.019, respectively). The total daily bolus insulin dose at baseline was 0.45 U/kg for mealtime URLi, 0.49 U/kg for postmeal URLi, and 0.44 U/kg for lispro. At week 26, the total daily bolus dose was 0.53 U/kg for mealtime URLi and 0.50 U/kg for postmeal URLi versus 0.48 U/kg for lispro (p = 0.029 and p = 0.523, respectively). The ratio of prandial to total insulin dose at week 26 was similar in each treatment group (~ 62%).
Safety and Tolerability
A summary of adverse events for each treatment group in Japanese patients is outlined in Table 2. Overall, the incidences of treatment emergent adverse events (TEAEs) related to the study treatment, serious adverse events (SAEs), and discontinuations from the study treatment due to an AE were low and similar across treatment groups. In addition, the majority of TEAEs related to the study treatment were mild in severity, and resolved without study insulin dose adjustment. There were no discontinuations from the study due to an AE. At week 26, mean change from baseline for body weight was 0.6 kg for lispro, − 0.1 kg for mealtime URLi, and no change for postmeal URLi.
Note that hypoglycemia events, including incidence and rate, are best represented in Table 3 and Fig. 5.
Incidence and rate of hypoglycemia (with or without symptoms) from week 0 to week 26 in Japanese patients. Rate and incidence of a documented and nocturnal hypoglycemia (BG < 54 mg/dL) and b documented symptomatic and asymptomatic postmeal hypoglycemia (BG < 54 mg/dL). Data are LSM + SE for event rate and LSM for incidence. BG blood glucose, LSM least squares mean, SE standard error, URLi ultra-rapid lispro
During the 26-week treatment period, the incidence and rate of severe hypoglycemia in Japanese patients were low, with no significant differences between treatment groups. There were no significant differences in the incidence of documented symptomatic hypoglycemia between mealtime or postmeal URLi and mealtime lispro. The rate of documented symptomatic hypoglycemia (events/patients/30 days) and the rate of non-nocturnal hypoglycemia (events/patients/year) with glucose below 54 mg/dL was significantly lower for mealtime URLi compared to lispro (p = 0.013 and p = 0.003, respectively) (Table 3).
The rates of documented and nocturnal hypoglycemia were significantly lower in URLi-treated patients in comparison to lispro (p = 0.002 and p = 0.028, respectively). Both the postmeal hypoglycemia rate and incidence were low. The rate and incidence of postprandial hypoglycemia for the timepoint of 0–4 h was statistically significantly lower for mealtime URLi compared to lispro (rate [SE] 4.68 [0.754] URLi vs. 7.75 [1.268] lispro; incidence 67.7% URLi vs. 79.6% lispro), and in the late postprandial period (more than 4 h after the meal) (see Fig. 5b). There were no clinically significant treatment differences in the rate and incidence of postprandial hypoglycemia between postmeal URLi and lispro (Fig. 5).
Discussion
PRONTO-T1D demonstrated that mealtime and postmeal URLi are non-inferior to lispro in terms of change in HbA1c following 26 weeks of treatment [21]. In addition, mealtime URLi was superior to lispro at reducing 1- and 2-h PPG excursions during the meal test [21]. In our subpopulation analysis of Japanese patients with T1DM, URLi demonstrated effective glycemic control when used in combination with basal insulin, with no clinically significant difference in safety profile observed. This is the first study to report the efficacy and safety of URLi compared to lispro in this patient population, and provides valuable information for clinicians and policy-makers in Japan.
As per the overall population, mealtime URLi and postmeal URLi were comparable to lispro in terms of change in mean HbA1c levels. The upper limit of 95% CI of LSM differences between URLi (mealtime and postmeal) and lispro was lower than 0.4%, which was the pre-specified non-inferiority margin for the overall population [21]. Consistent mean HbA1c profiles were observed between Japanese patients and the overall population from study entry through week 26.
After 26 weeks of treatment, mealtime URLi demonstrated statistically significantly lower PPG excursions following the MMTT in comparison to lispro at all timepoints (ranging from 30 min to 4 h postmeal) among Japanese patients. Although mealtime URLi did not show a statistically significant difference in controlling HbA1c levels in comparison to lispro, we believe that URLi will benefit patients by reducing PPG. An excess in glucose excursions could result in glucose variability and consequently heightened risk of hyperglycemia and/or hypoglycemia [25]. An important observation is that the overall trend in PPG excursions in the MMTT was consistently lower for mealtime URLi compared to lispro in Japanese patients, as seen in the overall population. Interestingly, the estimated treatment differences between mealtime URLi and lispro were more pronounced in Japanese patients in comparison to the overall population [21].
Postmeal URLi resulted in statistically significantly greater PPG excursions in comparison to lispro at 30 min during the MMTT; however, postmeal URLi and lispro were numerically similar in terms of PPG excursions at 1 and 2 h after the meal, with a statistically significant reduction in PPG excursions evident at the later timepoint of 3 h for postmeal URLi compared to lispro. This indicates that URLi is efficacious when administered at the commencement of a meal and up to 20 min after the start of the meal. This finding reflects the fast onset and short duration of action observed in URLi clinical pharmacology studies [26, 27]. As described earlier, many patients inject mealtime insulin after the start of a meal as a result of various inevitable reasons [22]. In addition, injecting URLi 15 min after a meal led to lower PPG excursions compared to injecting lispro 15 min after a meal [28]. Taken together, we believe that URLi will provide improved PPG control for people who are unable to inject mealtime insulin before or at the start of a meal, with the advantage of being on-label use for postmeal administration in Japan.
In Japanese patients, the overall daily average blood glucose levels in the 10-point SMBG profiles were similar between treatments, except for blood glucose levels at the midday and evening premeal timepoints where lispro showed lower PPG levels compared to mealtime URLi, and higher PPG levels for postmeal URLi 1 h following the midday and evening timepoints in comparison to lispro. The SMBG results observed in this subgroup analysis suggest the possibility of further optimization of URLi dosing in conjunction with basal insulin. For example, premeal dosing of URLi did not cause a premeal dip in blood glucose as was observed in the lispro arm, although postmeal blood glucose was the same or lower in premeal dosing of URLi compared to lispro in Japanese patients and in the overall population [21]. It is therefore possible that additional titration of basal insulin could lead to better overall blood glucose control with the use of URLi. Further studies are warranted to explore this possibility.
In a clinical pharmacology study, URLi demonstrated a “fast-on” and “fast-off” pharmacokinetic (PK) and pharmacodynamic (PD) profile [29]. Our current findings regarding lower blood glucose for lispro treatment at the premeal, midday, and evening timepoints, may be due to drug concentration and action being of longer duration for lispro in comparison to URLi. In addition, for mealtime URLi, study investigators may have looked at the postmeal blood glucose levels and ceased titrating insulin once they observed a satisfactorily smaller postprandial blood glucose excursion. Although study investigators were instructed to achieve premeal blood glucose targets, they were also permitted to observe postprandial SMBG data. In the study, postprandial SMBG levels from 10-point SMBG profiles should have been evaluated for optimization of prandial insulin dosing. Also, site personnel were allowed to request additional blood glucose monitoring from patients and/or assess blood glucose values at other times (such as postprandial measurements) to inform clinical management decisions. In terms of postmeal URLi, our findings at the premeal, midday, and evening timepoints may be the result of higher postprandial blood glucose, hypothetically due to lack of postmeal inhibition of net splanchnic glucose output [30].
Importantly, the improvements in glycemic control and PPG excursions in Japanese patients occurred without increasing the risk of hypoglycemia. Overall, URLi was well tolerated by Japanese patients. The incidence of SAEs and TEAEs related to the study treatment was low and similar across treatment groups. There were no discontinuations from the study due to an AE, and no deaths during this 26-week trial.
Furthermore, it should be noted that the frequency of hypoglycemia at least 4 h after the meal was significantly lower for mealtime URLi compared to mealtime lispro. This finding may be due to differences in PK and PD profiles between URLi and lispro. Specifically, URLi exhibits reduced late exposure and reduced late insulin action, with a shorter duration of insulin action compared with lispro [29]. This indicates that URLi may lower both postprandial blood glucose and the frequency of fasting hypoglycemia.
It is well established that achieving glycemic control, as determined by HbA1c levels, is effective in reducing the risk of diabetes complications. In addition, epidemiology studies indicate that elevated PPG levels increase the risk of cardiovascular disease [11, 31, 32], and controlling PPG excursions is essential for achieving recommended HbA1c goals [33, 34]. In comparison to lispro, URLi may not only match the time action profile of normal insulin secretion more closely but may also lower PPG excursions, with an acceptable safety profile, as described in this Japanese subgroup analysis of the PRONTO-T1D trial.
In Japan, recombinant human insulin and insulin analogues (such as insulin glulisine, insulin aspart, and insulin lispro) are currently available as mealtime insulin products. Following approval in Europe [35], URLi was recently granted approval in Japan under the trade name Lyumjev (March 2020) [36]. Recently, Fiasp®, composed of insulin aspart with two added excipients to ensure formulation stability with accelerated absorption [37], was approved and launched in Japan for mealtime and postmeal administration. Aside from Fiasp®, currently available products could be administered up to 15 min prior to commencing a meal (according to the Japan label) [19, 31, 32]; thus, Fiasp® was the only postmeal product available in Japan before the launch of Lyumjev. In a recent quantitative survey, substantial burden was reported by Japanese patients with diabetes regarding the current injection timing of mealtime insulin [22]. Hence, there is a medical need for faster-acting products in Japan.
A key limitation of the study design was the use of an open-label design for the postmeal treatment arm. In addition, the MMTT insulin dose may not have been fit for Japanese patients who had a relatively lower body weight compared to patients in the overall population. The 500-rule was used to calculate the MMTT insulin dose for the majority of Japanese patients using the pattern adjustment method, and a limitation of the 500-rule method is that it does not account for body weight. As mentioned, Japanese patients in this subgroup analysis were, on average, lighter than patients in the overall population. In addition, the small number of patients in this subpopulation analysis limited the likelihood of obtaining statistical differences in this population.
Conclusion
This 26-week randomized controlled trial indicates that URLi is effective and comparable to lispro in terms of overall glycemic control when administered as prandial insulin in combination with basal insulin, either immediately before or up to 20 min after the start of a meal in Japanese patients with T1DM. URLi provides effective PPG control when administered immediately before the start of a meal. Furthermore, URLi was well tolerated, with an acceptable safety profile in this population.
References
World Health Organization (WHO). Global report on diabetes 2016. Geneva: WHO; 2016. https://www.who.int/diabetes/global-report/en/. Accessed 11 Mar 2020.
Sherwani SI, Khan HA, Ekhzaimy A, Masood A, Sakharkar MK. Significance of HbA1c test in diagnosis and prognosis of diabetic patients. Biomarker Insights. 2016;11:95–104.
Bonora E, Tuomilehto J. The pros and cons of diagnosing diabetes with A1C. Diabetes Care. 2011;34(Suppl 2):S184–S190190.
American Diabetes Association. Standards of medical care in diabetes-2019. Abridged for primary care providers. Clin Diabetes. 2019;37:11–34.
Stark Casagrande S, Fradkin JE, Saydah SH, Rust KF, Cowie CC. The prevalence of meeting A1C, blood pressure, and LDL goals among people with diabetes, 1988–2010. Diabetes Care. 2013;36:2271–9.
Japan Diabetes Clinical Data Management Study Group. 2018. https://jddm.jp/data/index-2017.html#data_03. Accessed 11 Mar 2020
Monnier L, Lapinski H, Colette C. Contributions of fasting and postprandial plasma glucose increments to the overall diurnal hyperglycemia of type 2 diabetic patients: variations with increasing levels of HbA(1c). Diabetes Care. 2003;26:881–5.
Standl E, Schnell O, Ceriello A. Postprandial hyperglycemia and glycemic variability: should we care? Diabetes Care. 2011;34(Suppl 2):S120–S127127.
Madsbad S. Impact of postprandial glucose control on diabetes-related complications: how is the evidence evolving? J Diabetes Compl. 2016;30:374–85.
DECODE Study Group, the European Diabetes Epidemiology Group. Glucose tolerance and cardiovascular mortality: comparison of fasting and 2-h diagnostic criteria. Arch Intern Med. 2001;161:397–405.
Meigs JB, Nathan DM, D'Agostino RB Sr, Wilson PW. Fasting and postchallenge glycemia and cardiovascular disease risk: the Framingham Offspring Study. Diabetes Care. 2002;25:1845–50.
Hanefeld M, Fischer S, Julius U, et al. Risk factors for myocardial infarction and death in newly detected NIDDM: the Diabetes Intervention Study, 11-year follow-up. Diabetologia. 1996;39:1577–83.
Brod M, Nikolajsen A, Weatherall J, Pfeiffer KM. Understanding post-prandial hyperglycemia in patients with type 1 and type 2 diabetes: a web-based survey in Germany, the UK, and USA. Diabetes Ther. 2016;7:335–48.
Pfeiffer KM, Sandberg A, Nikolajsen A, Brod M. Postprandial glucose and healthcare resource use: a cross-sectional survey of adults with diabetes treated with basal-bolus insulin. J Med Econ. 2018;21:66–73.
Heinemann L, Hompesch M, Flacke F, et al. Reduction of postprandial glycemic excursions in patients with type 1 diabetes: a novel human insulin formulation versus a rapid-acting insulin analog and regular human insulin. J Diabetes Sci Technol. 2011;5:681–6.
Boss AH, Petrucci R, Lorber D. Coverage of prandial insulin requirements by means of an ultra-rapid-acting inhaled insulin. J Diabetes Sci Technol. 2012;6:773–9.
Andersen G, Meiffren G, Lamers D, et al. Ultra-rapid BioChaperone Lispro improves postprandial blood glucose excursions vs insulin lispro in a 14-day crossover treatment study in people with type 1 diabetes. Diabetes Obes Metab. 2018;20:2627–32.
Krasner A, Pohl R, Simms P, Pichotta P, Hauser R, De Souza E. A review of a family of ultra-rapid-acting insulins: formulation development. J Diabetes Sci Technol. 2012;6:786–96.
Heinemann L, Muchmore DB. Ultrafast-acting insulins: state of the art. J Diabetes Sci Technol. 2012;6:728–42.
Humalog [prescribing information]. Eli Lilly and Company; 2015. https://pi.lilly.com/us/humalog-pen-pi.pdf. Accessed 25 Mar 2020
Klaff L, Cao D, Dellva MA, et al. Ultra rapid lispro improves postprandial glucose control compared with lispro in patients with type 1 diabetes: results from the 26-week PRONTO-T1D study. Diabetes Obes Metab. 2020. https://doi.org/10.1111/dom.14100.
Ishii H, Suichi S, Williams P, Demiya S, Aranishi T, Treuer T. Cross-sectional survey in patients with type 1 and type 2 diabetes to understand mealtime insulin unmet needs in Japan: the MINUTES-J study. Diabetes Res Clin Pract. 2020;162:108076.
Harashima SI, Nishimura A, Inagaki N. Attitudes of patients and physicians to insulin therapy in Japan: an analysis of the Global Attitude of Patients and Physicians in Insulin Therapy study. Expert Opin Pharmacother. 2017;18:5–11.
World Health Organization (WHO). Classification of diabetes mellitus. Geneva: WHO; 2019. https://www.who.int/publications-detail/classification-of-diabetes-mellitus. Accessed 11 Mar 2020
Ceriello A, Monnier L, Owens D. Glycaemic variability in diabetes: clinical and therapeutic implications. Lancet Diabetes Endocrinol. 2019;7:221–30.
Leohr J, Dellva MA, Carter K, et al. 1040-P Ultra-rapid lispro (URLi) accelerates insulin lispro absorption and insulin action vs. humalog (lispro) consistently across study populations: a meta-analysis of pharmacokinetic and glucodynamic data. Diabetes. 2020. https://doi.org/10.2337/db20-1040-P
Linnebjerg H, Zhang Q, LaBell E, et al. 1107-P: ultra rapid lispro (URLi) accelerates insulin lispro absorption and insulin action vs. Humalog® (lispro) in patients with T1D. Diabetes. 2019;68:(Suppl 1). https://doi.org/10.2337/db19-1107-P.
Plum-Moerschel L, Leohr J, Liu, R, et al. 1010-P—ultra-rapid lispro (URLi) reduces postprandial glucose excursions vs. Humalog® in patients with T1D at multiple meal-to-dose timing intervals. Amercican Diabetes Association 78th Scientific Session. 2018. https://plan.core-apps.com/tristar_ada18/abstract/807d2f9885450670bb994661e3fdd160.
Shiramoto M, Nasu R, Oura T, Imori M, Ohwaki K. Ultra-rapid lispro results in accelerated insulin lispro absorption and faster early insulin action in comparison with Humalog® in Japanese patients with type 1 diabetes. J Diabetes Investig. 2019. https://doi.org/10.1111/jdi.13195.
Ferrannini E, Bjorkman O, Reichard GA Jr, et al. The disposal of an oral glucose load in healthy subjects. A quantitative study. Diabetes. 1985;34:580–8.
DECODE Study Group, European Diabetes Epidemiology Group. Is the current definition for diabetes relevant to mortality risk from all causes and cardiovascular and noncardiovascular diseases? Diabetes Care. 2003;26:688–96.
Coutinho M, Gerstein HC, Wang Y, Yusuf S. The relationship between glucose and incident cardiovascular events. A metaregression analysis of published data from 20 studies of 95,783 individuals followed for 12.4 years. Diabetes Care. 1999;22:233–40.
Ketema EB, Kibret KT. Correlation of fasting and postprandial plasma glucose with HbA1c in assessing glycemic control; systematic review and meta-analysis. Arch Public Health. 2015;73:43.
Woerle HJ, Neumann C, Zschau S, et al. Impact of fasting and postprandial glycemia on overall glycemic control in type 2 diabetes Importance of postprandial glycemia to achieve target HbA1c levels. Diabetes Res Clin Pract. 2007;77:280–5.
European Medicines Agency. Liumjev. https://www.ema.europa.eu/en/medicines/human/EPAR/liumjev#authorisation-details-section. Accessed 2 Jun 2020
Pharmaceutical and Medical Devices Agency. Summary of investigation results. Preparations containing insulin. https://www.pmda.go.jp/files/000235075.pdf#page=4. Accessed 2 Jun 2020
Haahr H, Heise T. Fast-acting insulin aspart: a review of its pharmacokinetic and pharmacodynamic properties and the clinical consequences. Clin Pharmacokinet. 2020;59:155–72.
Acknowledgements
We thank the patients and their families/caregivers, the study investigators and their staff, and the PRONTO-T1D clinical trial team.
Funding
This study was sponsored by Eli Lilly and Company, IN, USA. This report, including the Rapid Service Fee, was sponsored by Eli Lilly Japan K.K. (Kobe, Japan). All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Medical Writing, Editorial and Other Assistance
Medical writing support was provided by Lisa Cossens, and editorial support by Dana Schamberger and Likhitha Kurma, of Syneos Health and funded by Eli Lilly Japan K.K. Project management support was provided by Yumi Sophia Suzuki from Eli Lilly Japan K.K.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Makoto Imori, Hiroshi Nishiyama, and Takeshi Imaoka are employees of Eli Lilly Japan K.K. and minor stockholders of Eli Lilly and Company. Junnosuke Miura discloses the following relationships: receipt of speaker's fees from Taisho Pharma, Novo Nordisk, Novartis, Eli Lilly, Sanofi, Johnson & Johnson, Abbott, Terumo, Boehringer Ingelheim, Kyowa Kirin, Tanabe-Mitsubishi, MSD, Mylan, AstraZeneca, and Astellas; consultant fees from Kowa, Kanro, Terumo, AstraZeneca, and Abbott.
Compliance with Ethics Guidelines
The study was conducted in accordance with the International Council for Harmonization Guidelines for Good Clinical Practice including the Declaration of Helsinki and its later amendments and the Council for International Organisation of Medical Sciences International Ethical Guidelines. An ethical review board at each site (Kobori Clinic, H.E.C. Science Clinic, Jinnouchi Hospital, Heiwadai Hospital, The Institute for Adult Diseases, Naka Memorial Clinic, Tomonaga Clinic, Nihonbashi Sakura Clinic, AMC Nishi-umeda Clinic, Manda Hospital, Fukuoka Red Cross Hospital, Shin Akasaka Clinic, Shinagawa East One Medical Clinic, Sasebo Chuo Hospital, Okuguchi Clinic of Internal Medicine, Sapporo Skin Clinic, Takatsuki Red Cross Hospital, Osaka City University Medical School Hospital, Goshogatani Home Clinic, Tokyo Women's Medical University Hospital) approved all procedures, and all subjects provided written, informed consent.
Data Availability
Eli Lilly and Company provides access to all individual participant data collected during the trial, after anonymization, with the exception of pharmacokinetic or genetic data. Data are available to request 6 months after the indication studied has been approved in the USA and EU and after primary publication acceptance, whichever is later. No expiration date of data requests is currently set once data are made available. Access is provided after a proposal has been approved by an independent review committee identified for this purpose and after receipt of a signed data sharing agreement. Data and documents, including the study protocol, statistical analysis plan, clinical study report, and blank or annotated case report forms, will be provided in a secure data sharing environment. For details on submitting a request, see the instructions provided at https://www.vivli.org.
Author information
Authors and Affiliations
Corresponding author
Additional information
Digital Features
To view digital features for this article go to https://doi.org/10.6084/m9.figshare.12656945.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Miura, J., Imori, M., Nishiyama, H. et al. Ultra-Rapid Lispro Efficacy and Safety Compared to Humalog® in Japanese Patients with Type 1 Diabetes: PRONTO-T1D Subpopulation Analysis. Diabetes Ther 11, 2089–2104 (2020). https://doi.org/10.1007/s13300-020-00892-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-020-00892-0