Abstract
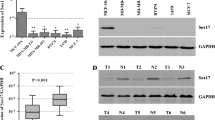
Sex-determining region Y-related high mobility group box 4 (SOX4) has been proven to serve as a critical role in cancer development and progression. However, little is known about the pathological role of SOX4 in breast cancer patients. The purpose of this study is to measure the expression of SOX4 in breast cancer patients and to explore the clinical significance of SOX4. Using RT-PCR and Western blot, messenger RNA (mRNA) and protein expression of SOX4 were measured in breast cancer tissues and adjacent normal mammary tissues. The relationship of SOX4 expression with clinical characteristics of 148 breast cancer patients was analyzed by immunohistochemistry. In the present study, our results indicated that SOX4 mRNA and protein were highly expressed in breast cancer tissues compared with adjacent normal mammary tissues and positively correlated with clinical stage (I–II vs. III–IV; P = 0.008), T classification (T1–T2 vs. T3–T4; P = 0.013), N classification (N0–N1 vs. N2–N3; P < 0.001), M classification (M0 vs. M1; P = 0.001), estrogen receptor (negative vs. positive; P = 0.029), progesterone receptor (negative vs. positive; P = 0.004), and histological grade (G1 vs. G2–G3; P = 0.033) in breast cancer patients. Furthermore, we also found that SOX4 protein overexpression was an unfavorable prognostic factor in breast cancer patients (P < 0.001), regardless of clinical stage, tumor size, lymph node metastasis, and distant metastasis. Finally, high SOX4 expression was an independent poor prognostic factor for pancreatic patients through multivariate analysis (P = 0.033). In conclusion, SOX4 overexpression serves as an unfavorable prognostic biomarker in breast cancer patients.




Similar content being viewed by others
References
Bombonati A, Sgroi DC. The molecular pathology of breast cancer progression. J Pathol. 2011;223:307–17.
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA: A Cancer J Clin. 2014;64:9–29.
Hong W, Dong E. The past, present and future of breast cancer research in China. Cancer Lett. 2014;351:1–5.
Lu J, Steeg PS, Price JE, Krishnamurthy S, Mani SA, Reuben J, et al. Breast cancer metastasis: challenges and opportunities. Cancer Res. 2009;69:4951–3.
Radenkovic S, Milosevic Z, Konjevic G, Karadzic K, Rovcanin B, Buta M, et al. Lactate dehydrogenase, catalase, and superoxide dismutase in tumor tissue of breast cancer patients in respect to mammographic findings. Cell Biochem Biophys. 2013;66:287–95.
Radenkovic S, Konjevic G, Jurisic V, Karadzic K, Nikitovic M, Gopcevic K. Values of mmp-2 and mmp-9 in tumor tissue of basal-like breast cancer patients. Cell Biochem Biophys. 2014;68:143–52.
Radenkovic S, Konjevic G, Isakovic A, Stevanovic P, Gopcevic K, Jurisic V. Her2-positive breast cancer patients: correlation between mammographic and pathological findings. Radiat Prot Dosim. 2014;162:125–8.
Jafarnejad SM, Ardekani GS, Ghaffari M, Li G. Pleiotropic function of SRY-related HMG box transcription factor 4 in regulation of tumorigenesis. Cell Mol Life Sci: CMLS. 2013;70:2677–96.
Vervoort SJ, van Boxtel R, Coffer PJ. The role of SRY-related HMG box transcription factor 4 (SOX4) in tumorigenesis and metastasis: friend or foe? Oncogene. 2013;32:3397–409.
Peluso S, Chiappetta G. High-mobility group A (HMGA) proteins and breast cancer. Breast Care (Basel, Switzerland). 2010;5:81–5.
Zhang J, Liang Q, Lei Y, Yao M, Li L, Gao X, et al. SOX4 induces epithelial-mesenchymal transition and contributes to breast cancer progression. Cancer Res. 2012;72:4597–608.
Sinner D, Kordich JJ, Spence JR, Opoka R, Rankin S, Lin SC, et al. Sox17 and Sox4 differentially regulate beta-catenin/T-cell factor activity and proliferation of colon carcinoma cells. Mol Cell Biol. 2007;27:7802–15.
Andersen CL, Christensen LL, Thorsen K, Schepeler T, Sorensen FB, Verspaget HW, et al. Dysregulation of the transcription factors SOX4, CBFB and SMARCC1 correlates with outcome of colorectal cancer. Br J Cancer. 2009;100:511–23.
Liao YL, Sun YM, Chau GY, Chau YP, Lai TC, Wang JL, et al. Identification of SOX4 target genes using phylogenetic footprinting-based prediction from expression microarrays suggests that overexpression of SOX4 potentiates metastasis in hepatocellular carcinoma. Oncogene. 2008;27:5578–89.
Hur W, Rhim H, Jung CK, Kim JD, Bae SH, Jang JW, et al. SOX4 overexpression regulates the p53-mediated apoptosis in hepatocellular carcinoma: clinical implication and functional analysis in vitro. Carcinogenesis. 2010;31:1298–307.
Liu P, Ramachandran S, Ali Seyed M, Scharer CD, Laycock N, Dalton WB, et al. Sex-determining region Y box 4 is a transforming oncogene in human prostate cancer cells. Cancer Res. 2006;66:4011–9.
Moreno CS. The sex-determining region Y-box 4 and homeobox c6 transcriptional networks in prostate cancer progression: crosstalk with the Wnt, Notch, and PI3K pathways. Am J Pathol. 2010;176:518–27.
Shen R, Pan S, Qi S, Lin X, Cheng S. Epigenetic repression of microRNA-129-2 leads to overexpression of SOX4 in gastric cancer. Biochem Biophys Res Commun. 2010;394:1047–52.
Fang CL, Hseu YC, Lin YF, Hung ST, Tai C, Uen YH, et al. Clinical and prognostic association of transcription factor SOX4 in gastric cancer. PLoS One. 2012;7:e52804.
Huang HY, Cheng YY, Liao WC, Tien YW, Yang CH, Hsu SM, et al. SOX4 transcriptionally regulates multiple sema3/plexin family members and promotes tumor growth in pancreatic cancer. PLoS One. 2012;7:e48637.
Zhang H, Alberich-Jorda M, Amabile G, Yang H, Staber PB, Di Ruscio A, et al. SOX4 is a key oncogenic target in C/EBPalpha mutant acute myeloid leukemia. Cancer Cell. 2013;24:575–88.
Wang L, Li Y, Yang X, Yuan H, Li X, Qi M, et al. ERG-SOX4 interaction promotes epithelial-mesenchymal transition in prostate cancer cells. Prostate. 2014;74:647–58.
Jafarnejad SM, Ardekani GS, Ghaffari M, Martinka M, Li G. SOX4-mediated dicer expression is critical for suppression of melanoma cell invasion. Oncogene. 2013;32:2131–9.
Wang L, Zhang J, Yang X, Chang YW, Qi M, Zhou Z, et al. SOX4 is associated with poor prognosis in prostate cancer and promotes epithelial-mesenchymal transition in vitro. Prostate Cancer Prostatic Dis. 2013;16:301–7.
Vervoort SJ, Lourenco AR, van Boxtel R, Coffer PJ. SOX4 mediates TGF-beta-induced expression of mesenchymal markers during mammary cell epithelial to mesenchymal transition. PLoS One. 2013;8:e53238.
Tiwari N, Tiwari VK, Waldmeier L, Balwierz PJ, Arnold P, Pachkov M, et al. SOX4 is a master regulator of epithelial-mesenchymal transition by controlling EZH2 expression and epigenetic reprogramming. Cancer Cell. 2013;23:768–83.
Jafarnejad SM, Wani AA, Martinka M, Li G. Prognostic significance of SOX4 expression in human cutaneous melanoma and its role in cell migration and invasion. Am J Pathol. 2010;177:2741–52.
Wang C, Zhao H, Lu J, Yin J, Zang L, Song N, et al. Clinicopathological significance of SOX4 expression in primary gallbladder carcinoma. Diagn Pathol. 2012;7:41.
Ramezani-Rad P, Geng H, Hurtz C, Chan LN, Chen Z, Jumaa H, et al. SOX4 enables oncogenic survival signals in acute lymphoblastic leukemia. Blood. 2013;121:148–55.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Guo-Dong Song and Yu Sun are co-first authors.
Rights and permissions
About this article
Cite this article
Song, GD., Sun, Y., Shen, H. et al. SOX4 overexpression is a novel biomarker of malignant status and poor prognosis in breast cancer patients. Tumor Biol. 36, 4167–4173 (2015). https://doi.org/10.1007/s13277-015-3051-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3051-9