Abstract
Background
Human gliomas are aggressive brain tumors characterized by uncontrolled cell proliferation. Differential expression of Polycomb repressive complex 2 (PRC2) has been reported in various subtypes of glioma. However, the role of PRC2 in uncontrolled growth in glioma and its underlying molecular mechanisms remain to be elucidated.
Objective
We aimed to investigate the functional role of PRC2 in human glioblastoma cell growth by silencing SUZ12, the non-catalytic core component of PRC2.
Methods
Knockdown of SUZ12 was achieved by infecting T98G cells with lentivirus carrying sequences specifically targeting SUZ12 (shSUZ12). Gene expression was examined by quantitative PCR and western analysis. The impact of shSUZ12 on cell growth was assessed using a cell proliferation assay. Cell cycle distribution was analyzed by flow cytometry, and protein stability was evaluated in cycloheximide-treated cells. Subcellular localization was examined through immunofluorescence staining and biochemical cytoplasmic-nuclear fractionation. Gene expression analysis was also performed on human specimens from normal brain and glioblastoma patients.
Results
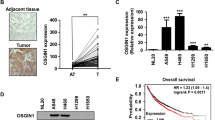
SUZ12 knockdown (SUZ12 KD) led to widespread decrease in the PRC2-specific histone mark, accompanied by a slowdown of cell proliferation through G1 arrest. In SUZ12 KD cells, the degradation of CDKN1B protein was reduced, resulting from alterations in the MYC-SKP2-CDKN1B axis. Furthermore, nuclear localization of CDKN1B was enhanced in SUZ12 KD cells. Analysis of human glioblastoma samples yielded increased expression of EZH2 and MYC along with reduced CDKN1B compared to normal human brain tissue.
Conclusion
Our findings suggest a novel role for SUZ12 in cell proliferation through post-translational regulation of CDKN1B in glioblastoma.




Similar content being viewed by others
Abbreviations
- EZH2:
-
Enhancer of zeste homolog 2
- KD:
-
Knockdown
- PRC2:
-
Polycomb repressive complex 2
- SUZ12:
-
Suppressor of zeste 12 protein homolog
- EED:
-
Embryonic ectoderm development
- MGMT:
-
O-6-methylguanine-DNA methyltransferase
- H3K27me3:
-
Histone H3 lysine 27 trimethylation
- BMI1:
-
BMI1, Polycomb ring finger
- PCGF2:
-
Polycomb group ring finger 2
- RNF2:
-
Ring figure protein 2
- CCNE:
-
Cylclin E1
- RB:
-
Retinoblastoma 1
- LMNB1:
-
Lamin B1
- CHX:
-
Chlorohexidine
- CDK:
-
Cyclin-dependent kinase
- CCND1:
-
Cyclin D1
- CDKN1B:
-
Cyclin-dependent kinase inhibitor 1B
References
Alexander BM, Pinnell N, Wen PY et al (2012) Targeting DNA repair and the cell cycle in glioblastoma. J Neurooncol 3:463–477. https://doi.org/10.1007/s11060-011-0765-4
Aloia L, Di Stefano B, Di Croce L (2013) Polycomb complexes in stem cells and embryonic development. Development 12:2525–2534. https://doi.org/10.1242/dev.091553
Au SL, Wong CC, Lee JM et al (2012) Enhancer of zeste homolog 2 epigenetically silences multiple Tumor suppressor microRNAs to promote Liver cancer Metastasis. Hepatology 2:622–631. https://doi.org/10.1002/hep.25679
Benetatos L, Vartholomatos G, Hatzimichael E (2014) Polycomb group proteins and MYC: the cancer connection. Cell Mol Life Sci 2:257–269. https://doi.org/10.1007/s00018-013-1426-x
Bretones G, Acosta JC, Caraballo JM et al (2011) SKP2 oncogene is a direct MYC target gene and MYC down-regulates p27(KIP1) through SKP2 in human Leukemia cells. J Biol Chem 11:9815–9825. https://doi.org/10.1074/jbc.M110.165977
Cao R, Zhang Y (2004) SUZ12 is required for both the histone methyltransferase activity and the silencing function of the EED-EZH2 complex. Mol Cell 1:57–67. https://doi.org/10.1016/j.molcel.2004.06.020
Caraballo JM, Acosta JC, Cortes MA et al (2014) High p27 protein levels in chronic lymphocytic Leukemia are associated to low Myc and Skp2 expression, confer resistance to apoptosis and antagonize Myc effects on cell cycle. Oncotarget 13:4694–4708. https://doi.org/10.18632/oncotarget.2100
Carrano AC, Eytan E, Hershko A et al (1999) SKP2 is required for ubiquitin-mediated degradation of the CDK inhibitor p27. Nat Cell Biol 4:193–199. https://doi.org/10.1038/12013
Chou J, Ferris AC, Chen T et al (2019) Roles of polycomb group proteins enhancer of zeste (E(z)) and polycomb (pc) during metamorphosis and larval leg regeneration in the flour beetle Tribolium castaneum. Dev Biol 1:34–46. https://doi.org/10.1016/j.ydbio.2019.03.002
Chu IM, Hengst L, Slingerland JM (2008) The cdk inhibitor p27 in human cancer: prognostic potential and relevance to anticancer therapy. Nat Rev Cancer 4:253–267. https://doi.org/10.1038/nrc2347
Chung H, Jung YM, Shin DH et al (2008) Anticancer effects of wogonin in both estrogen receptor-positive and -negative human Breast cancer cell lines in vitro and in nude mice xenografts. Int J Cancer 4:816–822. https://doi.org/10.1002/ijc.23182
Chuu CP, Kokontis JM, Hiipakka RA et al (2011) Androgen suppresses proliferation of castration-resistant LNCaP 104-R2 Prostate cancer cells through androgen receptor, Skp2, and c-Myc. Cancer Sci 11:2022–2028. https://doi.org/10.1111/j.1349-7006.2011.02043.x
Clarke J, Penas C, Pastori C et al (2013) Epigenetic pathways and glioblastoma treatment. Epigenetics 8:785–795. https://doi.org/10.4161/epi.25440
Conway E, Healy E, Bracken AP (2015) PRC2 mediated H3K27 methylations in cellular identity and cancer. Curr Opin Cell Biol. 42–48. https://doi.org/10.1016/j.ceb.2015.10.003
Crea F, Hurt EM, Farrar WL (2010) Clinical significance of polycomb gene expression in brain tumors. Mol Cancer 265. https://doi.org/10.1186/1476-4598-9-265
Deb G, Singh AK, Gupta S (2014) EZH2: not EZHY (easy) to deal. Mol Cancer Res 5:639–653. https://doi.org/10.1158/1541-7786.MCR-13-0546
Del Moral-Morales A, Gonzalez-Orozco JC, Hernandez-Vega AM et al (2022) EZH2 mediates proliferation, Migration, and Invasion promoted by Estradiol in Human Glioblastoma cells. Front Endocrinol (Lausanne) 703733. https://doi.org/10.3389/fendo.2022.703733
Dunn GP, Rinne ML, Wykosky J et al (2012) Emerging insights into the molecular and cellular basis of glioblastoma. Genes Dev 8:756–784. https://doi.org/10.1101/gad.187922.112
Gu J, Wang J, You A et al (2021) MiR-137 inhibits the proliferation, invasion and migration of glioma via targeting to regulate EZH2. Genes Genomics 10:1157–1165. https://doi.org/10.1007/s13258-021-01117-9
Ichimura K, Pearson DM, Kocialkowski S et al (2009) IDH1 mutations are present in the majority of common adult gliomas but rare in primary glioblastomas. Neuro Oncol 4:341–347. https://doi.org/10.1215/15228517-2009-025
Jo S, Lee H, Kim S et al (2011) Inhibition of PCGF2 enhances granulocytic differentiation of acute promyelocytic Leukemia cell line HL-60 via induction of HOXA7. Biochem Biophys Res Commun 1–2:86–91. https://doi.org/10.1016/j.bbrc.2011.10.152
Jo S, Lee H, Kim S et al (2013) Korean red ginseng extract induces proliferation to differentiation transition of human acute promyelocytic Leukemia cells via MYC-SKP2-CDKN1B axis. J Ethnopharmacol 2:700–707. https://doi.org/10.1016/j.jep.2013.09.036
Kim KH, Roberts CW (2016) Targeting EZH2 in cancer. Nat Med 2:128–134. https://doi.org/10.1038/nm.4036
Kim W, Bird GH, Neff T et al (2013a) Targeted disruption of the EZH2-EED complex inhibits EZH2-dependent cancer. Nat Chem Biol 10:643–650. https://doi.org/10.1038/nchembio.1331
Kim E, Kim M, Woo DH et al (2013b) Phosphorylation of EZH2 activates STAT3 signaling via STAT3 methylation and promotes tumorigenicity of glioblastoma stem-like cells. Cancer Cell 6:839–852. https://doi.org/10.1016/j.ccr.2013.04.008
Kissler S, Stern P, Takahashi K et al (2006) In vivo RNA interference demonstrates a role for Nramp1 in modifying susceptibility to type 1 Diabetes. Nat Genet 4:479–483. https://doi.org/10.1038/ng1766
Koumangoye RB, Andl T, Taubenslag KJ et al (2015) SOX4 interacts with EZH2 and HDAC3 to suppress microRNA-31 in invasive Esophageal cancer cells. Mol Cancer 24. https://doi.org/10.1186/s12943-014-0284-y
Li LY (2014) EZH2: novel therapeutic target for human cancer. Biomed (Taipei). https://doi.org/10.7603/s40681-014-0001-6. 1:1
Li T, Luo W, Liu K et al (2015) miR-31 promotes proliferation of colon Cancer cells by targeting E2F2. Biotechnol Lett 3:523–532. https://doi.org/10.1007/s10529-014-1715-y
Liang J, Zubovitz J, Petrocelli T et al (2002) PKB/Akt phosphorylates p27, impairs nuclear import of p27 and opposes p27-mediated G1 arrest. Nat Med 10:1153–1160. https://doi.org/10.1038/nm761
Louis DN, Perry A, Wesseling P et al (2021) The 2021 WHO classification of tumors of the Central Nervous System: a summary. Neuro Oncol 8:1231–1251. https://doi.org/10.1093/neuonc/noab106
Macdonald JI, Dick FA (2012) Posttranslational modifications of the retinoblastoma Tumor suppressor protein as determinants of function. Genes Cancer 11–12:619–633. https://doi.org/10.1177/1947601912473305
Mineura K, Ishiyama Y, Kowada M (1992) Development of resistance to antitumor chloroethylnitrosoureas in vitro in Brain Tumor cells. Acta Oncol 7:755–759. https://doi.org/10.3109/02841869209083866
Miranda TB, Cortez CC, Yoo CB et al (2009) DZNep is a global histone methylation inhibitor that reactivates developmental genes not silenced by DNA methylation. Mol Cancer Ther 6:1579–1588. https://doi.org/10.1158/1535-7163.MCT-09-0013
Montagnoli A, Fiore F, Eytan E et al (1999) Ubiquitination of p27 is regulated by cdk-dependent phosphorylation and trimeric complex formation. Genes Dev 9:1181–1189. https://doi.org/10.1101/gad.13.9.1181
Nadiminty N, Tummala R, Lou W et al (2012) MicroRNA let-7c suppresses androgen receptor expression and activity via regulation of myc expression in Prostate cancer cells. J Biol Chem 2:1527–1537. https://doi.org/10.1074/jbc.M111.278705
Natsume A, Ito M, Katsushima K et al (2013) Chromatin regulator PRC2 is a key regulator of epigenetic plasticity in glioblastoma. Cancer Res 14:4559–4570. https://doi.org/10.1158/0008-5472.CAN-13-0109
Omuro A, DeAngelis LM (2013) Glioblastoma and other malignant gliomas: a clinical review. JAMA 17:1842–1850. https://doi.org/10.1001/jama.2013.280319
Orian JM, Vasilopoulos K, Yoshida S et al (1992) Overexpression of multiple oncogenes related to histological grade of astrocytic glioma. Br J Cancer 1:106–112. https://doi.org/10.1038/bjc.1992.225
Park SC, Lee JM (2022) Ezh2 promotes TRbeta lysine methylation-mediated degradation in hepatocellular carcinoma. Genes Genomics 3:369–377. https://doi.org/10.1007/s13258-021-01196-8
Parreno V, Martinez AM, Cavalli G (2022) Mechanisms of polycomb group protein function in cancer. Cell Res 3:231–253. https://doi.org/10.1038/s41422-021-00606-6
Pasini D, Bracken AP, Jensen MR et al (2004) Suz12 is essential for mouse development and for EZH2 histone methyltransferase activity. EMBO J 20:4061–4071. https://doi.org/10.1038/sj.emboj.7600402
Ryu J, Yoon NA, Lee YK et al (2015) Tristetraprolin inhibits the growth of human glioma cells through downregulation of urokinase plasminogen activator/urokinase plasminogen activator receptor mRNAs. Mol Cells 2:156–162. https://doi.org/10.14348/molcells.2015.2259
Schiffer D, Cavalla P, Fiano V et al (2002) Inverse relationship between p27/Kip.1 and the F-box protein Skp2 in human astrocytic gliomas by immunohistochemistry and Western blot. Neurosci Lett 2:125–128. https://doi.org/10.1016/s0304-3940(02)00483-4
Schuettengruber B, Bourbon HM, Di Croce L et al (2017) Genome regulation by Polycomb and trithorax: 70 years and counting. Cell 1:34–57. https://doi.org/10.1016/j.cell.2017.08.002
Shi B, Liang J, Yang X et al (2007) Integration of estrogen and wnt signaling circuits by the polycomb group protein EZH2 in Breast cancer cells. Mol Cell Biol 14:5105–5119. https://doi.org/10.1128/MCB.00162-07
Shin I, Yakes FM, Rojo F et al (2002) PKB/Akt mediates cell-cycle progression by phosphorylation of p27(Kip1) at threonine 157 and modulation of its cellular localization. Nat Med 10:1145–1152. https://doi.org/10.1038/nm759
Shukla S, Ying W, Gray F et al (2021) Small-molecule inhibitors targeting polycomb repressive complex 1 RING domain. Nat Chem Biol 7:784–793. https://doi.org/10.1038/s41589-021-00815-5
Sicari BM, Troxell R, Salim F et al (2012) c-myc and skp2 coordinate p27 degradation, vascular smooth muscle proliferation, and neointima formation induced by the parathyroid hormone-related protein. Endocrinology 2:861–872. https://doi.org/10.1210/en.2011-1590
Stazi G, Taglieri L, Nicolai A et al (2019) Dissecting the role of novel EZH2 inhibitors in primary glioblastoma cell cultures: effects on proliferation, epithelial-mesenchymal transition, migration, and on the pro-inflammatory phenotype. Clin Epigenetics 1:173. https://doi.org/10.1186/s13148-019-0763-5
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 10:987–996. https://doi.org/10.1056/NEJMoa043330
Suva ML, Riggi N, Janiszewska M et al (2009) EZH2 is essential for glioblastoma cancer stem cell maintenance. Cancer Res 24:9211–9218. https://doi.org/10.1158/0008-5472.CAN-09-1622
Taherbhoy AM, Huang OW, Cochran AG (2015) BMI1-RING1B is an autoinhibited RING E3 ubiquitin ligase. Nat Commun 7621. https://doi.org/10.1038/ncomms8621
Vervoorts J, Luscher B (2008) Post-translational regulation of the Tumor suppressor p27(KIP1). Cell Mol Life Sci 20:3255–3264. https://doi.org/10.1007/s00018-008-8296-7
Viglietto G, Motti ML, Bruni P et al (2002) Cytoplasmic relocalization and inhibition of the cyclin-dependent kinase inhibitor p27(Kip1) by PKB/Akt-mediated phosphorylation in Breast cancer. Nat Med 10:1136–1144. https://doi.org/10.1038/nm762
Villa R, Pasini D, Gutierrez A et al (2007) Role of the polycomb repressive complex 2 in acute promyelocytic Leukemia. Cancer Cell 6:513–525. https://doi.org/10.1016/j.ccr.2007.04.009
Visani M, de Biase D, Marucci G et al (2014) Expression of 19 microRNAs in glioblastoma and comparison with other brain neoplasia of grades I-III. Mol Oncol 2:417–430. https://doi.org/10.1016/j.molonc.2013.12.010
Wander SA, Zhao D, Slingerland JM (2011) p27: a barometer of signaling deregulation and potential predictor of response to targeted therapies. Clin Cancer Res 1:12–18. https://doi.org/10.1158/1078-0432.CCR-10-0752
Yamagishi M, Nakano K, Miyake A et al (2012) Polycomb-mediated loss of miR-31 activates NIK-dependent NF-kappaB pathway in adult T cell Leukemia and other cancers. Cancer Cell 1:121–135. https://doi.org/10.1016/j.ccr.2011.12.015
Yamaguchi H, Hung MC (2014) Regulation and role of EZH2 in Cancer. Cancer Res Treat 3:209–222. https://doi.org/10.4143/crt.2014.46.3.209
Yu ZK, Gervais JL, Zhang H (1998) Human CUL-1 associates with the SKP1/SKP2 complex and regulates p21(CIP1/WAF1) and cyclin D proteins. Proc Natl Acad Sci U S A 19:11324–11329. https://doi.org/10.1073/pnas.95.19.11324
Zhang Q, Padi SK, Tindall DJ et al (2014) Polycomb protein EZH2 suppresses apoptosis by silencing the proapoptotic miR-31. Cell Death Dis 10:e1486. https://doi.org/10.1038/cddis.2014.454
Zhang J, Chen L, Han L et al (2015a) EZH2 is a negative prognostic factor and exhibits pro-oncogenic activity in glioblastoma. Cancer Lett 2 Pt B 929–936. https://doi.org/10.1016/j.canlet.2014.11.003
Zhang W, Liu H, Liu W et al (2015b) Polycomb-mediated loss of microRNA let-7c determines inflammatory macrophage polarization via PAK1-dependent NF-kappaB pathway. Cell Death Differ 2:287–297. https://doi.org/10.1038/cdd.2014.142
Zhong Q, Zhong Q, Cai X et al (2023) Identification and validation of an ECM organization-related gene signature as a prognostic biomarker and therapeutic target for glioma patients. Genes Genomics. https://doi.org/10.1007/s13258-023-01413-6
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2011-0014802 and NRF-2021R1A6A1A03038899).
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
SK and SJ primarily performed the experiments and analyzed data. SHP and SSK analyzed data in human glioma tissues. HC initiated, organized, and designed the study, analyzed data, and wrote the manuscript. All authors critically reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare they have no financial interests.
Ethics approval
Human tissue samples were obtained and processed under the approval of the Institutional Review Board of Seoul National University Hospital (IRB approval H-0B05-036-243).
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, S., Jo, S., Paek, S.H. et al. SUZ12 inhibition attenuates cell proliferation of glioblastoma via post-translational regulation of CDKN1B. Genes Genom 45, 1623–1632 (2023). https://doi.org/10.1007/s13258-023-01468-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-023-01468-5