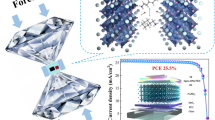
Abstract
We report a series of π-conjugated polymers (P1-F, P2-Cl, and P3-OMe) with three different functional groups (fluorine, chlorine, and methoxy) on their conjugated side chains. Although all three polymers showed identical photophysical properties by varying the functional group, they showed a notable difference in their dipole moment difference between the ground and excited state (Δµge) values. Furthermore, photovoltaic properties of fullerene organic solar cells (FOSCs)/non-fullerene organic solar cells (NFOSCs) were significantly affected concerning the functional group in the π-conjugated polymer. Interestingly, halogen-substituted polymers (P1-F and P2-Cl) showed an enhanced PCE than methoxy-substituted polymer (P3-OMe) in both NFOSCs and FOSCs. Also, the FOSCs were much affected upon functional group modulation than did in NFOSCs. The difference in the photovoltaic properties of P1-F, P2-Cl and P3-OMe based OSCs was further analyzed by atomic force microscopy, space charge limited current method, water contact angle and transient photoluminescence measurements. Overall, our work sheds light on the importance of side chain functional group modulation of donor polymers for efficient F and NFOSCs.
Graphical Abstract
We report a series of π-conjugated polymers (P1-F, P2-Cl, and P3-OMe) with three different functional groups (fluorine, chlorine, and methoxy) on their conjugated side chains. Although all three polymers showed identical photophysical properties by varying the functional group, they showed a notable difference in their dipole moment difference between the ground and excited state (Δµge) values and photovoltaic properties (fullerene and non-fullerene organic solar cells).







Similar content being viewed by others
References
H. Yao, L. Ye, H. Zhang, S. Li, S. Zhang, J. Hou, Chem. Rev. 116, 7379–7457 (2016)
G. Zhang, J. Zhao, P.C.Y. Chow, K. Jiang, J. Zhang, Z. Zhu, J. Zhang, F. Huang, H. Yan, Chem. Rev. 118, 3447–3507 (2018)
S. Li, C.-Z. Li, M. Shi, H. Chen, ACS Energy Lett. 5, 1554–1567 (2020)
L. Lu, T. Zheng, Q. Wu, A.M. Schneider, D. Zhao, L. Yu, Chem. Rev. 115, 12666–12731 (2015)
C. Yan, S. Barlow, Z. Wang, H. Yan, A.K.-Y. Jen, S.R. Marder, X. Zhan, Nat. Rev. Mater. 3, 18003 (2018)
P. Meredith, W. Li, A. Armin, Adv. Energy Mater. 10, 2001788 (2020)
H. Yao, J. Hou, Angew. Chem. Int. Ed. 61, e202209021 (2022)
Y. Cui, Y. Xu, H. Yao, P. Bi, L. Hong, J. Zhang, Y. Zu, T. Zhang, J. Qin, J. Ren, Z. Chen, C. He, X. Hao, Z. Wei, J. Hou, Adv. Mater. 33, 2102420 (2021)
H. Fu, Z. Wang, Y. Sun, Angew. Chem. Int. Ed. 58, 4442–4453 (2019)
L.-W. Feng, J. Chen, S. Mukherjee, V.K. Sangwan, W. Huang, Y. Chen, D. Zheng, J.W. Strzalka, G. Wang, M.C. Hersam, D. DeLongchamp, A. Facchetti, T.J. Marks, ACS Energy Lett. 5, 1780–1787 (2020)
B. Zheng, L. Huo, Y. Li, NPG Asia Mater 3, 12 (2020)
S. Li, L. Ye, W. Zhao, H. Yan, B. Yang, D. Liu, W. Li, H. Ade, J. Hou, J. Am. Chem. Soc. 140, 7159–7167 (2018)
K. He, P. Kumar, Y. Yuan, Y. Li, Mater. Adv. 2, 115–145 (2021)
K. Kranthiraja, A. Saeki, A.C.S. Appl, Mater. Interfaces 14, 28936–28944 (2022)
X. Xu, K. Feng, Z. Bi, W. Ma, G. Zhang, Q. Peng, Adv. Mater. 31, 1901872 (2019)
B. Fan, M. Li, D. Zhang, W. Zhong, L. Ying, Z. Zeng, K. An, Z. Huang, L. Shi, G.C. Bazan, F. Huang, Y. Cao, ACS Energy Lett. 5, 2087–2094 (2020)
Z. Zheng, H. Yao, L. Ye, Y. Xu, S. Zhang, J. Hou, Mater. Today 35, 115–130 (2020)
S. Li, W. Zhao, J. Zhang, X. Liu, Z. Zheng, C. He, B. Xu, Z. Wei, J. Hou, Chem. Mater. 32, 1993–2003 (2020)
N. Nakao, S. Ogawa, H.D. Kim, H. Ohkita, T. Mikie, M. Saito, I. Osaka, A.C.S. Appl, Mater. Interfaces 13, 56420–56429 (2021)
T. Jia, J. Zhang, K. Zhang, H. Tang, S. Dong, C.-H. Tan, X. Wang, F. Huang, J. Mater. Chem. A 9, 8975–8983 (2021)
J.J. Rech, L. Yan, Z. Wang, Q. Zhang, S. Bradshaw, H. Ade, W. You, A.C.S. Appl, Polym. Mater. 3, 30–41 (2021)
C. Sun, F. Pan, H. Bin, J. Zhang, L. Xue, B. Qiu, Z. Wei, Z.-G. Zhang, Y. Li, Nat Commun. 9, 743 (2018)
Q. Liu, Y. Jiang, K. Jin, J. Qin, J. Xu, W. Li, J. Xiong, J. Liu, Z. Xiao, K. Sun, S. Yang, X. Zhang, L. Ding, Sci. Bull. 65, 272 (2020)
N. Nakao, M. Saito, T. Mikie, T. Ishikawa, J. Jeon, H.D. Kim, H. Ohkita, A. Saeki, I. Osaka, Adv. Sci. 10, 2205682 (2023). https://doi.org/10.1002/advs.202205682
L. Zhu, M. Zhang, J. Xu, C. Li, J. Yan, G. Zhou, W. Zhong, T. Hao, J. Song, X. Xue, Z. Zhou, R. Zeng, H. Zhu, C.-C. Chen, R.C.I. MacKenzie, Y. Zou, J. Nelson, Y. Zhang, Y. Sun, F. Liu, Nat. Mater. 21, 656–663 (2022)
Y. Tang, H. Sun, Z. Wu, Y. Zhang, G. Zhang, M. Su, X. Zhou, X. Wu, W. Sun, X. Zhang, B. Liu, W. Chen, Q. Liao, H.Y. Woo, X. Guo, Adv. Sci. 6, 1901773 (2019)
Q. Zhang, M.A. Kelly, N. Bauer, W. You, Acc Chem. Res. 50, 2401–2409 (2017)
F. Meyer, Prog. Polym. Sci. 47, 70–91 (2015)
L. Lu, L. Yu, Adv. Mater. 26, 4413–4430 (2014)
Y. Liang, D. Feng, Y. Wu, S.-T. Tsai, G. Li, C. Ray, L. Yu, J. Am. Chem. Soc. 131, 7792–7799 (2009)
K. Kranthiraja, K. Gunasekar, H. Kim, A.-N. Cho, N.-G. Park, S. Kim, B.J. Kim, R. Nishikubo, A. Saeki, M. Song, S.-H. Jin, Adv. Mater. 29, 1700183 (2017)
K. Kranthiraja, S. Kim, C. Lee, K. Gunasekar, V.G. Sree, B. Gautam, K. Gundogdu, S.-H. Jin, B.J. Kim, Adv. Funct. Mater. 27, 1701256 (2017)
K. Kranthiraja, D.X. Long, V.G. Sree, W. Cho, Y.-R. Cho, A. Zaheer, J.-C. Lee, Y.-Y. Noh, S.-H. Jin, Macromolecules 51, 5530–5536 (2018)
T. Xu, L. Lu, T. Zheng, J.M. Szarko, A. Schneider, L.X. Chen, L. Yu, Adv. Funct. Mater. 24, 3432–3437 (2014)
X. Liao, Y. Cui, X. Shi, Z. Yao, H. Zhao, Y. An, P. Zhu, Y. Guo, X. Fei, L. Zuo, K. Gao, F. Lin, Q. Xie, L. Chen, W. Ma, Y. Chen, A.K.-Y. Jen, Mater. Chem. Front. 4, 1507–1518 (2020)
M. Li, Y. Zhou, J. Zhang, J. Song, Z. Bo, J. Mater. Chem. A 7, 8889–8896 (2019)
C.F.N. Marchiori, M. Koehler, J. Phys. D: Appl. Phys. 47, 215104 (2014)
S. Honda, H. Ohkita, H. Benten, S. Ito, Adv. Energy Mater. 1, 588–598 (2011)
H. Wang, L. Yang, P.-C. Lin, C.-C. Chueh, X. Liu, S. Qu, S. Guang, J. Yu, W.A. Tang, Small 17, 2007746 (2021)
F. Du, H. Wang, Z. Zhang, L. Yang, J. Cao, J. Yu, W. Tang, Mater. Horiz. 8, 1008–1016 (2021)
K. Kranthiraja, A. Saeki, ACS Appl. Polym. Mater. 3, 2759–2767 (2021)
D. Chen, S. Liu, J. Liu, J. Han, L. Chen, Y. Chen, A.C.S. Appl, Polym. Mater. 3, 1923–1931 (2021)
Acknowledgements
This work was supported by the National Research Foundation (NRF-2018R1A5A1025594 and 2022R1A2C2091150) by the Ministry of Science, ICT of Korea.
Author information
Authors and Affiliations
Contributions
KK, UKA: conceptualization, methodology, formal analysis, writing—original draft. HK, JL: formal analysis, data curation. SSR, RDG, TG: formal analysis, data curation, writing—original draft. S-HJ: writing—review & editing, supervision, project administration, funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. I declare that the above items are certain.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supporting information
Supporting information
Monomer and polymer synthesis, materials and instruments, device fabrication, device measurement and characterization, schemes S1 and S2; 1H and 13C NMR of monomers (Figure S1 and S2); 1H NMR of P1-F, P2-Cl and P3-OMe (Figure S3-S5); CV and TGA of P1-F, P2-Cl and P3-OMe (Figure S6); Normalized absorption spectra of P1-F, P2-Cl and P3-OMe in chlorobenzene and film (Figure S7); DFT simulated HOMO and LUMO of P1-F, P2-Cl and P3-OMe repeating unit (Figure S8); SCLC curves of hole-only only devices of F and NFOSCs (Figure S9); Pictures of CA measurements using water and glycerol (Figure S10); PCE vs absolute difference between the donor and acceptor materials; PCE vs χ (Figure S11); Summary of ground state dipole moment; excited state dipole moment; dipole moment difference between the ground and excited state of polymers (Table S1), SCLC hole mobility of optimized NFOSCs and FOSCs (Table S2); Transient lifetime values of the pristine polymers and their optimized blends. (Table S3); contact angle and surface free energy of donor polymers and acceptor materials (Table S4); Flory-Huggins interaction parameter (χ) obtained from the SFE (Table S5).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kranthiraja, K., Kim, H., Lee, J. et al. Side chain functionalization of conjugated polymer on the modulation of photovoltaic properties of fullerene and non-fullerene organic solar cells. Macromol. Res. 31, 897–905 (2023). https://doi.org/10.1007/s13233-023-00176-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-023-00176-z