Abstract
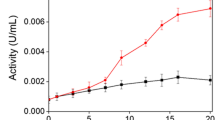
This study is the first report on purification, characterization, and application of laccase derived from the white-rot fungus, Pleurotus ostreatus HK35 (Hungary strain), in Congo Red decolorization. The purification process involved ammonium sulfate precipitation, dialysis, anion exchange chromatography, and ultrafiltration, yielding a specific laccase activity of 15.26 U/mg and a 30.21% recovery rate. The purified enzyme, with a molecular weight of approximately 34 kilodaltons, displayed optimal activity at a temperature of 60 °C and pH 4.0 when using 2,2′-azino-bis (3-ethylbenzthiazoline-6-sulphonic acid) (ABTS) as a substrate. The enzyme maintained over 82.02 ± 1.01% of its activity at temperatures up to 50 °C after 180 min but displayed less than 5% of its activity at 60 and 70 °C. Notably, the enzyme’s activity was significantly enhanced by Pb(NO3)2, whereas β-mercaptoethanol completely inhibited the activity. Utilizing the Box–Behnken design, we optimized Congo Red decolorization efficiency to 91.05 ± 0.82% at 100 mg/L Congo Red, 1.5 mM mediator concentration, and 1.6 U/mL laccase activity. Analysis of Variance (ANOVA) suggested the model was significant, and all variables significantly influenced decolorization efficiency.







Similar content being viewed by others
Data availability
The datasets supporting the results of this article are not publicly available but can be obtained upon reasonable request from the corresponding author.
References
Abdelgalil SA, Attia AR, Reyed RM, Soliman NA (2020) Partial purification and biochemical characterization of a new highly acidic NYSO laccase from Alcaligenes faecalis. J Genet Eng Biotechnol 18(1):79. https://doi.org/10.1186/s43141-020-00088-w
Amin S, Rastogi RP, Chaubey MG, Jain K, Divecha J, Desai C, Madamwar D (2020) Degradation and toxicity analysis of a reactive textile diazo dye-direct Red 81 by newly isolated Bacillus sp. DMS2. Front Microbiol. https://doi.org/10.3389/fmicb.2020.576680
Bagewadi ZK, Mulla SI, Ninnekar HZ (2017) Purification and immobilization of laccase from Trichoderma harzianum strain HZN10 and its application in dye decolorization. J Genet Eng Biotechnol 15(1):139–150. https://doi.org/10.1016/j.jgeb.2017.01.007
Barreto WJ, Doi SMO, Afonso R, Bernardino ND (2012) A kinetic study of the biodegradation of textile dye mixtures by Pleurotus ostreatus fungus. Monatshefte für Chemie - Chem Monthly 143(10):1389–1395. https://doi.org/10.1007/s00706-012-0753-1
Ben Ayed A, Hadrich B, Sciara G, Lomascolo A, Bertrand E, Faulds CB, Zouari-Mechichi H, Record E, Mechichi T (2022) Optimization of the decolorization of the reactive black 5 by a laccase-like active cell-free supernatant from Coriolopsis gallica. Microorganisms. https://doi.org/10.3390/microorganisms10061137
Benzina O, Frikha F, Zouari-Mechichi H, Woodward S, Belbahri L, Mnif E, Mechichi T (2012) Enhanced decolourization of the azo dye Sirius rose BB by laccase–HBT system. 3 Biotech. 2(2):149–57. https://doi.org/10.1007/s13205-011-0041-5
Benzina O, Daâssi D, Zouari-Mechichi H, Frikha F, Woodward S, Belbahri L, Rodriguez-Couto S, Mechichi T (2013) Decolorization and detoxification of two textile industry effluents by the laccase/1-hydroxybenzotriazole system. Environ Sci Pollut Res Int 20(8):5177–5187. https://doi.org/10.1007/s11356-013-1491-6
Bettin F, da Rosa LO, Montanari Q, Calloni R, Gaio TA, Malvessi E, da Silveira MM, Dillon AJP (2011) Growth kinetics, production, and characterization of extracellular laccases from Pleurotus sajor-caju PS-2001. Process Biochem 46(3):758–764. https://doi.org/10.1016/j.procbio.2010.12.002
Bhavsar S, Dudhagara P, Tank S (2018) R software package based statistical optimization of process components to simultaneously enhance the bacterial growth, laccase production and textile dye decolorization with cytotoxicity study. PLoS One 13(5):e0195795. https://doi.org/10.1371/journal.pone.0195795
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Budikania TS, Afriani K, Widiana I, Saksono N (2019) Decolorization of azo dyes using contact glow discharge electrolysis. J Environ Chem Eng 7(6):103466. https://doi.org/10.1016/j.jece.2019.103466
Cristóvão R, Tavares A, Loureiro J, Boaventura R, de Rebello A, Macedo ME (2008) Optimisation of reactive dye degradation by laccase using Box-Behnken design. Environ Technol 29:1357–1364. https://doi.org/10.1080/09593330802379615
Daâssi D, Frikha F, Zouari-Mechichi H, Belbahri L, Woodward S, Mechichi T (2012) Application of response surface methodology to optimize decolourization of dyes by the laccase-mediator system. J Environ Manage 108:84–91. https://doi.org/10.1016/j.jenvman.2012.04.039
Daâssi D, Zouari-Mechichi H, Frikha F, Martinez MJ, Nasri M, Mechichi T (2013) Decolorization of the azo dye acid orange 51 by laccase produced in solid culture of a newly isolated Trametes trogii strain. 3 Biotech 3(2):115–125. https://doi.org/10.1007/s13205-012-0076-2
Das A, Bhattacharya S, Panchanan G, Navya BS, Nambiar P (2016) Production, characterization and Congo red dye decolourizing efficiency of a laccase from Pleurotus ostreatus MTCC 142 cultivated on co-substrates of paddy straw and corn husk. J Genet Eng Biotechnol 14(2):281–288. https://doi.org/10.1016/j.jgeb.2016.09.007
Dhiman K, Poonam S (2020) Production, partial purification and characterization of laccase from rhizospheric bacteria Pseudomonas putida strain LUA15.1. Res J Biotechnol 15:144–152
Ezike TC, Ezugwu AL, Udeh JO, Eze SOO, Chilaka FC (2020) Purification and characterisation of new laccase from Trametes polyzona WRF03. Biotechnol Rep 28:e00566. https://doi.org/10.1016/j.btre.2020.e00566
Freixo Mdo R, Karmali A, Arteiro JM (2012) Production, purification and characterization of laccase from Pleurotus ostreatus grown on tomato pomace. World J Microbiol Biotechnol 28(1):245–254. https://doi.org/10.1007/s11274-011-0813-4
Giardina P, Faraco V, Pezzella C, Piscitelli A, Vanhulle S, Sannia G (2010) Laccases: a never-ending story. Cell Mol Life Sci 67(3):369–385. https://doi.org/10.1007/s00018-009-0169-1
Glazunova OA, Moiseenko KV, Savinova OS, Fedorova TV (2020) Purification and characterization of two novel laccases from Peniophora lycii. J Fungi (Basel). https://doi.org/10.3390/jof6040340
Gupta A, Jana AK (2018) Effects of wheat straw solid contents in fermentation media on utilization of soluble/insoluble nutrient, fungal growth and laccase production. 3 Biotech 8(1):35. https://doi.org/10.1007/s13205-017-1054-5
Iark D, Buzzo A, Garcia JAA, Côrrea VG, Helm CV, Corrêa RCG, Peralta RA, Peralta Muniz Moreira RF, Bracht A, Peralta RM (2019) Enzymatic degradation and detoxification of azo dye Congo red by a new laccase from Oudemansiella canarii. Bioresour Technol 289:121655. https://doi.org/10.1016/j.biortech.2019.121655
Illuri R, Kumar M, Eyini M, Veeramanikandan V, Almaary KS, Elbadawi YB, Biraqdar MA, Balaji P (2021) Production, partial purification and characterization of ligninolytic enzymes from selected basidiomycetes mushroom fungi. Saudi J Biol Sci 28(12):7207–7218. https://doi.org/10.1016/j.sjbs.2021.08.026
Isanapong J (2021) Production of Laccase from Pleurotus sp. from Hungary on Agricultural Biomass by Solid-State Fermentation. In: Thai Society for Biotechnology International Conference Online, Bangkok, Thailand. pp 575–580
Jeznabadi EK, Jafarpour M, Eghbalsaied S (2016) King oyster mushroom production using various sources of agricultural wastes in Iran. Int J Recycl Org Waste Agric 5(1):17–24. https://doi.org/10.1007/s40093-015-0113-3
Junior JA, Vieira YA, Cruz IA, da Silva VD, Aguiar MM, Torres NH, Bharagava RN, Lima ÁS, de Souza RL, Romanholo Ferreira LF (2020) Sequential degradation of raw vinasse by a laccase enzyme producing fungus Pleurotus sajor-caju and its ATPS purification. Biotechnol Rep 25:e00411. https://doi.org/10.1016/j.btre.2019.e00411
Kang S-F, Liao C-H, Po S-T (2000) Decolorization of textile wastewater by photo-fenton oxidation technology. Chemosphere 41(8):1287–1294. https://doi.org/10.1016/S0045-6535(99)00524-X
Karp SG, Faraco V, Amore A, Letti LA, Thomaz Soccol V, Soccol CR (2015) Statistical optimization of laccase production and delignification of sugarcane bagasse by Pleurotus ostreatus in solid-state fermentation. Biomed Res Int. https://doi.org/10.1155/2015/181204
Kasiri MB, Modirshahla N, Mansouri H (2013) Decolorization of organic dye solution by ozonation; optimization with response surface methodology. Int J Ind Chem 4(1):3. https://doi.org/10.1186/2228-5547-4-3
Kaur M, Sharma S, Sodhi HS (2022) Chapter 10 - An eco-friendly approach for the degradation of azo dyes and their effluents by Pleurotus florida. In: Dar GH, Bhat RA, Qadri H, Hakeem KR (eds) Microbial Consortium and Biotransformation for Pollution Decontamination. Elsevier, pp 209–242. https://doi.org/10.1016/B978-0-323-91893-0.00006-7
Leo VV, Passari AK, Muniraj IK, Uthandi S, Hashem A (2019) Elevated levels of laccase synthesis by Pleurotus pulmonarius BPSM10 and its potential as a dye decolorizing agent. Saudi J Biol Sci 26(3):464–468. https://doi.org/10.1016/j.sjbs.2018.10.006
Leontievsky AA, Vares T, Lankinen P, Shergill JK, Pozdnyakova NN, Myasoedova NM, Kalkkinen N, Golovleva LA, Cammack R, Thurston CF, Hatakka A (1997) Blue and yellow laccases of ligninolytic fungi. FEMS Microbiol Lett 156(1):9–14. https://doi.org/10.1016/S0378-1097(97)00393-5
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56(3):658–666. https://doi.org/10.1021/ja01318a036
Liu W, Chao Y, Yang X, Bao H, Qian S (2004) Biodecolorization of azo, anthraquinonic and triphenylmethane dyes by white-rot fungi and a laccase-secreting engineered strain. J Ind Microbiol Biotechnol 31(3):127–132. https://doi.org/10.1007/s10295-004-0123-z
Liu X, Deng W, Yang Y (2021) Characterization of a novel laccase LAC-Yang1 from white-rot fungus Pleurotus ostreatus strain Yang1 with a strong ability to degrade and detoxify chlorophenols. Molecules. https://doi.org/10.3390/molecules26020473
Lonappan L, Rouissi T, Laadila MA, Brar SK, Hernandez Galan L, Verma M, Surampalli RY (2017) Agro-industrial-produced laccase for degradation of diclofenac and identification of transformation products. ACS Sustain Chem Eng 5(7):5772–5781. https://doi.org/10.1021/acssuschemeng.7b00390
Manavalan A, Manavalan T, Murugesan K, Kutzner A, Thangavelu KP, Heese K (2015) Characterization of a solvent, surfactant and temperature-tolerant laccase from Pleurotus sp. MAK-II and its dye decolorizing property. Biotechnol Lett 37(12):2403–2409. https://doi.org/10.1007/s10529-015-1937-7
Navada KK, Kulal A (2021) Kinetic characterization of purified laccase from Trametes hirsuta: a study on laccase catalyzed biotransformation of 1,4-dioxane. Biotechnol Lett 43(3):613–626. https://doi.org/10.1007/s10529-020-03038-1
Okamoto K, Yanagi SO, Sakai T (2000) Purification and characterization of extracellular laccase from Pleurotus ostreatus. Mycoscience 41(1):7–13. https://doi.org/10.1007/BF02464380
Oladoye PO, Bamigboye MO, Ogunbiyi OD, Akano MT (2022) Toxicity and decontamination strategies of Congo red dye. Groundw Sustain Dev 19:100844. https://doi.org/10.1016/j.gsd.2022.100844
Othman A, Elshafei A, Elsayed M, Hassan M (2018) Decolorization of cibacron blue 3G-A Dye by Agaricus bisporus CU13 laccase - mediator system: a statistical study for optimization via response surface methodology. Annual Res Rev Biol 25:1–13. https://doi.org/10.9734/ARRB/2018/40772
Patel H, Gupte S, Gahlout M, Gupte A (2014) Purification and characterization of an extracellular laccase from solid-state culture of Pleurotus ostreatus HP-1. 3 Biotech 4(1):77–84. https://doi.org/10.1007/s13205-013-0129-1
Patel SK, Kalia VC, Choi JH, Haw JR, Kim IW, Lee JK (2014b) Immobilization of laccase on SiO2 nanocarriers improves its stability and reusability. J Microbiol Biotechnol 24(5):639–647. https://doi.org/10.4014/jmb.1401.01025
Pradhananga RR, Adhikari L, Shrestha RG, Adhikari MP, Rajbhandari R, Ariga K, Shrestha LK (2017) Wool carpet dye adsorption on nanoporous carbon materials derived from agro-product. C 3(2):12
Radveikienė I, Vidžiūnaitė R, Meškienė R, Meškys R, Časaitė V (2021) Characterization of a yellow laccase from Botrytis cinerea 241. J Fungi (Basel). https://doi.org/10.3390/jof7020143
Rajhans G, Barik A, Sen SK, Raut S (2021) Degradation of dyes by fungi: an insight into mycoremediation. BioTechnologia (Pozn) 102(4):445–455. https://doi.org/10.5114/bta.2021.111109
Ramírez-Cavazos LI, Junghanns C, Ornelas-Soto N, Cárdenas-Chávez DL, Hernández-Luna C, Demarche P, Enaud E, García-Morales R, Agathos SN, Parra R (2014) Purification and characterization of two thermostable laccases from Pycnoporus sanguineus and potential role in degradation of endocrine disrupting chemicals. J Mol Catal B Enzym 108:32–42. https://doi.org/10.1016/j.molcatb.2014.06.006
Rampinelli JR, Melo MP, Arbigaus A, Silveira M, Wagner TM, Gern RMM, Wisbeck E, Bonatti-Chaves M, Furigo Júnior A, Furlan SA (2021) Production of Pleurotus sajor-caju crude enzyme broth and its applicability for the removal of bisphenol A. An Acad Bras Cienc 93(1):e20191153. https://doi.org/10.1590/0001-3765202120191153
Rezaei S, Tahmasbi H, Mogharabi M, Ameri A, Forootanfar H, Khoshayand MR, Faramarzi MA (2015) Laccase-catalyzed decolorization and detoxification of Acid Blue 92: statistical optimization, microtoxicity, kinetics, and energetics. J Environ Health Sci Eng 13(1):31. https://doi.org/10.1186/s40201-015-0183-1
Satheesh Babu S, Mohandass C, Vijayaraj AS, Dhale MA (2015) Detoxification and color removal of Congo red by a novel Dietzia sp. (DTS26) – A microcosm approach. Ecotoxicol Environ Saf 114:52–60. https://doi.org/10.1016/j.ecoenv.2015.01.002
Senthivelan T, Kanagaraj J, Panda RC (2016) Recent trends in fungal laccase for various industrial applications: an eco-friendly approach - a review. Biotechnol Bioprocess Eng 21(1):19–38. https://doi.org/10.1007/s12257-015-0278-7
Singh J, Kumar P, Saharan V, Kapoor RK (2019) Simultaneous laccase production and transformation of bisphenol-A and triclosan using Trametes versicolor. 3 Biotech 9(4):129. https://doi.org/10.1007/s13205-019-1648-1
Srijumpa N, Khayankan S (2011) Agricultural wastes as substrates for mushroom cultivation. Thai Agric Res J 29 (2)
Sun Y, Liu ZL, Hu BY, Chen QJ, Yang AZ, Wang QY, Li XF, Zhang JY, Zhang GQ, Zhao YC (2021) Purification and characterization of a thermo- and pH-stable laccase from the litter-decomposing fungus Gymnopus luxurians and Laccase mediator systems for dye decolorization. Front Microbiol 12:672620. https://doi.org/10.3389/fmicb.2021.672620
Tinoco R, Pickard MA, Vazquez-Duhalt R (2001) Kinetic differences of purified laccases from six Pleurotus ostreatus strains. Lett Appl Microbiol 32(5):331–335. https://doi.org/10.1046/j.1472-765x.2001.00913.x
Tychanowicz GK, Zilly A, de Souza CGM, Peralta RM (2004) Decolourisation of industrial dyes by solid-state cultures of Pleurotus pulmonarius. Process Biochem 39(7):855–859. https://doi.org/10.1016/S0032-9592(03)00194-8
Wang HX, Ng TB (2006) Purification of a laccase from fruiting bodies of the mushroom Pleurotus eryngii. Appl Microbiol Biotechnol 69(5):521–525. https://doi.org/10.1007/s00253-005-0086-7
Wang HQ, Yao Z, Sun Y, Zhou Z, Xiong Q, Zhong ZX (2014) Immobilization of γ-glutamyltranspeptidase on silylated mesoporous TiO2 whiskers. Biotechnol Bioprocess Eng 19(2):304–310. https://doi.org/10.1007/s12257-013-0675-8
Wang F, Xu L, Zhao L, Ding Z, Ma H, Terry N (2019) Fungal laccase production from lignocellulosic agricultural wastes by solid-state fermentation: a review. Microorganisms. https://doi.org/10.3390/microorganisms7120665
Zhu L, Wang Y, Zhu F, You L, Shen X (2018) Evaluation of the biosorption characteristics of Tremella fuciformis for the decolorization of cationic dye from aqueous solution. J Polym Environ 26(3):1051–1060. https://doi.org/10.1007/s10924-017-1010-9
Acknowledgements
The authors gratefully acknowledge King Mongkut’s University of Technology North Bangkok, Thailand, for the financial support of this research project under Grant No. KMUTNB-61-GOV-03-37. They thank Miss. Kanokwan Pribwai, Miss. Pawinee Bunkasem, and Miss. Suthasinee Anukunkij for their assistance in this research project. They would also like to thank the Mushroom Research and Development Group, Biotechnology Research and Development Office, Department of Agriculture, Thailand for their contribution in supplying pertinent information concerning the origin and strain specifics of P. ostreatus HK35.
Author information
Authors and Affiliations
Contributions
JI conceptualized the study. JI and SP contributed to writing and editing the manuscript. JI supervised the experimental work, performed the main data analysis, and administered the project and funding. KS and SL performed the experimental work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Ethical approval
Ethical approval was not required.
Informed consent
Not applicable as this study did not involve human participants.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Isanapong, J., Suwannoi, K., Lertlattanapong, S. et al. Purification, characterization of laccase from Pleurotus ostreatus HK35, and optimization for congo red biodecolorization using Box–Behnken design. 3 Biotech 14, 73 (2024). https://doi.org/10.1007/s13205-024-03926-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-024-03926-7