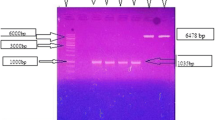
Abstract
The biotechnological application of bacterial xylanases requires a high thermostability, a catalytically active state for a broad pH range. The Bacillus amyloliquefaciens (MTCC 1270) xynA gene was amplified and cloned into the pCold vector and was expressed in Escherichia coli to evaluate the expressed proteins’ thermostability. The pCold, compared to other similar vectors, has unique properties—including pH and temperature tolerance due to the presence of the cspA promoter. The recombinant xynA-pCold (rxynApC) showed the expression of xynA gene with a molecular weight of ~ 27 kDa, confirmed on SDS-PAGE. The rxynApC exhibits optimal activity at 70 °C and pH 8.0. The residual activity of the recombinant enzyme was 90% at pH 8.0. The thermal decomposition temperature (Td) value for the rxynApC enzyme was 93.33 °C obtained from the thermogravimetric analysis, indicating the potent stability of the cloned enzyme. The specific activity of native xylanase and rxynApC under optimal conditions was 32.35 and 105.5 U/mg, respectively. The structural model of the xynA gene was predicted using the in silico tool along with the active site (containing four important Tyr-166, Gly-7, Try-69 and Arg-112 amino acids). The predicted biophysical parameters of the in silico model were similar to the experimental results. The unique feature of the cspA promoter is that it gave a high expression of rxynApC enzyme having alkali and thermostable properties with high yield in surrogate host E. coli. Thus, the recombinant xynA gene can potentially be applied to different industrial needs by looking at its thermostability and enhanced enzyme activity.







Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Adrio J-L, Demain AL (2010) Recombinant organisms for production of industrial products. Bioeng Bugs 1:116–131. https://doi.org/10.4161/bbug.1.2.10484
Altschul S (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402. https://doi.org/10.1093/nar/25.17.3389
Badon MM, Tekverk DG, Vishnosky NS, Woolridge EM (2019) Establishing the oxidative tolerance of Thermomyces lanuginosus xylanase. J Appl Microbiol 127:508–519. https://doi.org/10.1111/jam.14306
Basit A, Jiang W, Rahim K (2020) Xylanase and its industrial applications. In: Basso TP (ed) Biomass. IntechOpen, London (10.5772/intechopen.92156)
Bhardwaj N, Kumar B, Verma P (2019) A detailed overview of xylanases: an emerging biomolecule for current and future prospective. Bioresour Bioprocess 6:40. https://doi.org/10.1186/s40643-019-0276-2
Bjerga GEK, Williamson AK (2015) Cold shock induction of recombinant arctic environmental genes. BMC Biotechnol 15:78. https://doi.org/10.1186/s12896-015-0185-1
Chakdar H, Kumar M, Pandiyan K et al (2016) Bacterial xylanases: biology to biotechnology. 3 Biotech 6:150. https://doi.org/10.1007/s13205-016-0457-z
Dave G, Modi H (2018) FT-IR method for estimation of phytic acid content during bread-making process. Food Measure 12:2202–2208. https://doi.org/10.1007/s11694-018-9836-y
Driss D, Haddar A, Ghorbel R, Chaabouni SE (2014) Production of Xylooligosaccharides by immobilized his-tagged recombinant xylanase from Penicillium occitanis on nickel-chelate eupergit C. Appl Biochem Biotechnol 173:1405–1418. https://doi.org/10.1007/s12010-014-0932-0
Drula E, Garron M-L, Dogan S et al (2022) The carbohydrate-active enzyme database: functions and literature. Nucleic Acids Res 50:D571–D577. https://doi.org/10.1093/nar/gkab1045
Fu Z, Fan G, Zhu Y et al (2020) Soluble expression of a novel feruloyl esterase from Burkholderia pyrrocinia B1213 in Escherichia coli and optimization of production conditions. Biotechnol Biotechnol Equip 34:732–746. https://doi.org/10.1080/13102818.2020.1803129
Gomes LH, Duarte KMR, Andrino FG, Tavares FCA (2000) A simple method for DNA isolation from Xanthomonas spp. Sci Agric 57:553–555. https://doi.org/10.1590/S0103-90162000000300028
Gupta K, Thomas D, Vidya S et al (2005) Detailed protein sequence alignment based on spectral similarity score (SSS). BMC Bioinform 6:105. https://doi.org/10.1186/1471-2105-6-105
He F (2011) Bradford protein assay. Bio-Protoc. https://doi.org/10.21769/BioProtoc.45
Kuroda K, Ueda M (2014) Generation of arming yeasts with active proteins and peptides via cell surface display system: cell surface engineering, bio-arming technology. In: Mapelli V (ed) Yeast metabolic engineering. Springer, New York, pp 137–155 (10.3390/biom3030632)
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Liao H, Zheng H, Li S et al (2015) Functional diversity and properties of multiple xylanases from Penicillium oxalicum GZ-2. Sci Rep 5:12631. https://doi.org/10.1038/srep12631
Liu M, Li J, Rehman AU et al (2022) Sensitivity of family GH11 Bacillus amyloliquefaciens xylanase A (BaxA) and the T33I mutant to Oryza sativa xylanase inhibitor protein (OsXIP): an experimental and computational study. Enzyme Microb Technol 156:109998. https://doi.org/10.1016/j.enzmictec.2022.109998
Lu F, Lu M, Lu Z et al (2008) Purification and characterization of xylanase from Aspergillus ficuum AF-98. Biores Technol 99:5938–5941. https://doi.org/10.1016/j.biortech.2007.10.051
McCleary BV, McGeough P (2015) A comparison of polysaccharide substrates and reducing sugar methods for the measurement of endo-1,4-β-xylanase. Appl Biochem Biotechnol 177:1152–1163. https://doi.org/10.1007/s12010-015-1803-z
Murakami MT, Arni RK, Vieira DS et al (2005) Correlation of temperature induced conformation change with optimum catalytic activity in the recombinant G/11 xylanase A from Bacillus subtilis strain 168 (1A1). FEBS Lett 579:6505–6510. https://doi.org/10.1016/j.febslet.2005.10.039
Ninawe S, Kapoor M, Kuhad RC (2008) Purification and characterization of extracellular xylanase from Streptomyces cyaneus SN32. Biores Technol 99:1252–1258. https://doi.org/10.1016/j.biortech.2007.02.016
Paice MG, Bourbonnais R, Desrochers M et al (1986) A xylanase gene from Bacillus subtilis: nucleotide sequence and comparison with B. pumilus gene. Arch Microbiol 144:201–206. https://doi.org/10.1007/BF00410947
Patel DK, Patel K, Patel D, Dave G (2021) Engineering of thermostable phytase–xylanase for hydrolysis of complex biopolymers. 3 Biotech 11:390. https://doi.org/10.1007/s13205-021-02936-z
Ricci L, Umiltà E, Righetti MC et al (2018) On the thermal behavior of protein isolated from different legumes investigated by DSC and TGA: legume proteins investigated by DSC and TGA. J Sci Food Agric 98:5368–5377. https://doi.org/10.1002/jsfa.9078
Rosano GL, Ceccarelli EA (2014) Recombinant protein expression in Escherichia coli: advances and challenges. Front Microbiol. https://doi.org/10.3389/fmicb.2014.00172
Santiago-Hernández A, Vega-Estrada J, del Carmen M-H, Hidalgo-Lara ME (2007) Purification and characterization of two sugarcane bagasse-absorbable thermophilic xylanases from the mesophilic Cellulomonas flavigena. J Ind Microbiol Biotechnol 34:331–338. https://doi.org/10.1007/s10295-006-0202-4
Shatalov AA, Pereira H (2008) Effect of xylanases on peroxide bleachability of eucalypt (E. globulus) kraft pulp. Biochem Eng J 40:19–26. https://doi.org/10.1016/j.bej.2007.11.012
Srivastava P, Mukherjee K (2001) Cloning, characterization, and expression of xylanase gene from Bacillus lyticus in Escherichia coli and Bacillus subtilis. Prep Biochem Biotechnol 31:389–400. https://doi.org/10.1081/PB-100107484
Ujiie M, Roy C, Yaguchi M (1991) Low-molecular-weight xylanase from Trichoderma viride. Appl Environ Microbiol 57:1860–1862. https://doi.org/10.1128/aem.57.6.1860-1862.1991
Voytas D (1998) Agarose Gel Electrophoresis. In: Dunn BM, Speicher DW, Wingfield PT, Coligan JE (eds) Current protocols in protein science. John Wiley and Sons, Inc., Hoboken NJ USA, p A.4F.1-A.4F.3
Walia A, Guleria S, Mehta P et al (2017) Microbial xylanases and their industrial application in pulp and paper biobleaching: a review. 3 Biotech 7:11. https://doi.org/10.1007/s13205-016-0584-6
Xie W, Yu Q, Zhang R et al (2021) Insights into the catalytic mechanism of a novel XynA and structure-based engineering for improving bifunctional activities. Biochemistry 60:2071–2083. https://doi.org/10.1021/acs.biochem.1c00134
Xu X, Liu M, Dai X (2015) Expression of recombinant Bacillus amyloliquefaciens xylanase a in Escherichia coli and Potential Application in Xylan hydrolysis. BioResources 10:4694–4710. https://doi.org/10.15376/biores.10.3.4694-4711
Xuezheng L, Shuoshuo C, Guoying X et al (2010) Cloning and heterologous expression of two cold-active lipases from the Antarctic bacterium Psychrobacter sp. G: cloning and expression of cold-active lipases. Polar Res 29:421–429. https://doi.org/10.1111/j.1751-8369,2010.00189.x
Zarafeta D, Galanopoulou AP, Leni ME et al (2020) XynDZ5: a new thermostable GH10 xylanase. Front Microbiol 11:545. https://doi.org/10.3389/fmicb.2020.00545
Acknowledgements
We thank the Department of Science and Technology for providing INSPIRE Fellowship (No. DST/INSPIRE Fellowship/[IF170394].
Funding
This work was supported by the Department of Science and Technology, Delhi, India (No. DST/INSPIRE Fellowship/[IF170394], Dharti Keyur Patel.
Author information
Authors and Affiliations
Contributions
Material preparation, data collection, and analysis were performed. The first draft of the manuscript was written by Dharti Patel. The idea and design of work with critical revision the work was done by Dr. Gayarti Dave.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Patel, D.K., Dave, G. pCold-assisted expression of a thermostable xylanase from Bacillus amyloliquefaciens: cloning, expression and characterization. 3 Biotech 12, 245 (2022). https://doi.org/10.1007/s13205-022-03315-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03315-y