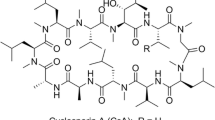
Abstract
Diethyl sulphate-based mutagenesis was performed on fungal strain Tolypocladium inflatum MTCC-3538. Two mutant morphotypes MT1-3538 and MT2-3538 were selected for further chemo-profiling studies. LCMS/MS profiling of fungal crude extract confirmed that the wild-type and mutant strains (MT1-3538, MT2-3538) were competent to produce cyclosporine A. MT2-3538 produced 2.1 fold higher cyclosporine A in comparison to the wild type. Further, LCMS-based high throughput media optimization was performed for MT2-3538 in 20 different media combinations to increase cyclosporine A yield. On the basis of ion-intensity profiling, media combination consisting of Glucose 0.1 g/L; Peptone 0.005 g/L and Valine 0.005 g/L was selected and used for up-scaling purpose. Mutant MT2-3538 with optimized media combination increased cyclosporine yield 16 fold and could potentially be exploited for commercial outcomes.



Similar content being viewed by others
References
Aarnio TH, Agathos SN (1989) Production of extracellular enzymes and cyclosporin byTolypocladium inflatum and morphologically related fungi. Biotechnol Lett 11(11):759–764. https://doi.org/10.1007/BF01026092
Abrol V, Kushwaha M, Arora D, Mallubhotla S, Jaglan S (2021) Mutation, chemoprofiling, dereplication, and isolation of natural products from Penicillium oxalicum. ACS Omega. https://doi.org/10.1021/acsomega.1c00141
Agathos S, Parekh R (1990) Enhancement of cyclosporin production in a Tolypocladium inflatum strain after epichlorohydrin treatment. J Biotechnol 13(1):73–81. https://doi.org/10.1016/0168-1656(90)90132-U
Anjum T, Iram W (2017) Production of cyclosporine A by submerged fermentation. In: Merillon J-M, Ramawat KG (eds) Fungal Metabolites. Springer International Publishing, Cham, pp 1–28
Anjum T, Azam A, Irum W (2012) Production of cyclosporine A by submerged fermentation from a local isolate of Penicillium fellutanum. Indian J Pharm Sci 74(4):372. https://doi.org/10.4103/0250-474X.107082
Barrios-Gonzalez J, Fernandez F, Tomasini A (2003) Microbial secondary metabolites production and strain improvement. Indian J Biotechnol 2(3):322–333
Blount RR. (2018). Secondary metabolism in Tolypocladium: Characterization, ecology, evolution and regulation.
Casareto E, Leoni M, Ronzio E, Magni A. (1998). Antibiotic producing microbe. Google Patents.
Chai Y-J, Cui C-B, Li C-W, Wu C-J, Tian C-K, Hua W (2012) Activation of the dormant secondary metabolite production by introducing gentamicin-resistance in a marine-derived Penicillium purpurogenum G59. Mar Drugs 10(3):559–582. https://doi.org/10.3390/md10030559
Chun G-T, Agathos S (1989) Immobilization of Tolypocladium inflatum spores into porous celite beads for cyclosporine production. J Biotechnol 9(4):237–254. https://doi.org/10.1016/0168-1656(89)90001-1
Demir H, Tari C (2016) Bioconversion of wheat bran for polygalacturonase production by Aspergillus sojae in tray type solid-state fermentation. Int Biodeterior Biodegradation 106:60–66. https://doi.org/10.1016/j.ibiod.2015.10.011
Domratcheva A, Zhgun A, Novak N, Dzhavakhiya V (2018) The influence of chemical mutagenesis on the properties of the cyclosporine A high-producer strain Tolypocladium inflatum VKM F-3630D. Appl Biochem Microbiol 54(1):53–57. https://doi.org/10.1134/S0003683818010027
Dong Y, Cui C-B, Li C-W, Hua W, Wu C-J, Zhu T-J, Gu Q-Q (2014) Activation of dormant secondary metabolite production by introducing neomycin resistance into the deep-sea fungus, Aspergillus versicolor ZBY-3. Mar Drugs 12(8):4326–4352. https://doi.org/10.3390/md12084326
Dreyfuss M, Härri E, Hea H, Kobel H, Pache W, Tscherter H (1976) Cyclosporin A and C. Eur J Appl Microbiol 3(2):125–133. https://doi.org/10.1007/BF00928431
Duetz WA, Rüedi L, Hermann R, O’Connor K, Büchs J, Witholt B (2000) Methods for intense aeration, growth, storage, and replication of bacterial strains in microtiter plates. Appl Environ Microbiol 66(6):2641–2646. https://doi.org/10.1128/AEM.66.6.2641-2646.2000
El-Kashef D, El-Kenawi A, Suddek G, Salem H (2017) Allicin ameliorates kidney function and urinary bladder sensitivity in cyclosporine A-treated rats. Hum Exp Toxicol 36(7):681–691. https://doi.org/10.1177/0960327116660864
Fang S-M, Wu C-J, Li C-W, Cui C-B (2014) A practical strategy to discover new antitumor compounds by activating silent metabolite production in fungi by diethyl sulphate mutagenesis. Mar Drugs 12(4):1788–1814. https://doi.org/10.3390/md12041788
Ferreira-Torres C, Micheletti M, Lye G (2005) Microscale process evaluation of recombinant biocatalyst libraries: application to Baeyer-Villiger monooxygenase catalysed lactone synthesis. Bioprocess Biosyst Eng 28(2):83–93. https://doi.org/10.1007/s00449-005-0422-4
Gilger BC, Wilkie DA, Clode AB, McMullen RJ, Utter ME, Komaromy AM, Brooks DE, Salmon JH (2010) Long-term outcome after implantation of a suprachoroidal cyclosporine drug delivery device in horses with recurrent uveitis. Vet Ophthalmol 13(5):294–300. https://doi.org/10.1111/j.1463-5224.2010.00807.x
Hoffmann GR (1980) Genetic effects of dimethyl sulfate, diethyl sulfate, and related compounds. MRGTEM 75(1):63–129. https://doi.org/10.1016/0165-1110(80)90028-7
Huang-jian Y, De-sen W, Zhu-lan Z, Wen-li T, Lin-ying R, Guan-rong Q, Yun-yang L (2017) Isolation, purification and structural identification of cyclosporine analogues from fermentation broth of strain Fusarium solani sp FIM116. Nat Prod Res Dev 29(2):240. https://doi.org/10.16333/j.1001-6880.2017.2.010
Irum W, Anjum T (2012) Production enhancement of Cyclosporin ‘A’by Aspergillus terreus through mutation. Afr j Biotechnol 11(7):1736–1743. https://doi.org/10.5897/AJB10.1330
Kahn JN, Byrne KM, Kaplan L, Monaghan RJ. (1994). Aspergillus niger mutants. Google Patents.
Keller NP, Turner G, Bennett JW (2005) Fungal secondary metabolism-from biochemistry to genomics. Nat Rev Microbiol 3(12):937. https://doi.org/10.1038/nrmicro1286
Ko SY, Kobel H, Besemer-Rosenwirth B, Seebach D, Traber R, Wenger R, Bollinger P. (2001). Cyclosporin fermentation process. Google Patents.
Li C-W, Xia M-W, Cui C-B, Peng J-X, Li D-H (2016) A novel oxaphenalenone, penicimutalidine: activated production of oxaphenalenones by the diethyl sulphate mutagenesis of marine-derived fungus Penicillium purpurogenum G59. RSC Adv 6(85):82277–82281. https://doi.org/10.1039/C6RA17087K
Lin J, Chen X, Cai X, Yu X, Liu X, Cao Y, Che Y (2011) Isolation and characterization of aphidicolin and chlamydosporol derivatives from Tolypocladium inflatum. J Nat Prod 74(8):1798–1804. https://doi.org/10.1021/np200431k
Manonmani AM, Geetha I, Balaraman K. (2016). Process for the Production of Cyclosporin-A Using the Fungus Tolypocladium sp. Strain NRRL No.: 18950. Google Patents.
Marudhu Ramachandran M, Balakrishnan K, Cynthiya A, Mani Sankar M, Panimalar M, Raghuraman T, Siva Ganesa Karthikeyan R (2007) Screening and isolation of cyclosporine-related compound producing soil fungi from the Western Ghats. Tamil Nadu Curr Sci 92(6):726–727
Membreno FE, Espinales JC, Lawitz EJ (2013) Cyclophilin inhibitors for hepatitis C therapy. Clin Liver Dis 17(1):129–139. https://doi.org/10.1016/j.cld.2012.09.008
Miach PJ (1986) Cyclosporin A in organ transplantation. Med J Aust 145(3–4):146–150. https://doi.org/10.5694/j.1326-5377.1986.tb113775.x
Nakajima H, Hamasaki T, Nishimura K, Kondo T, Kimura Y, Udagawa S-i, Sato S (1988) Isolation of 2-Acetylamino-3-hydroxy-4-methyloct-6-enoic Acid, a derivative of the “C9-Amino Acid” residue of Cyclosporins, produced by the fungus Neocosmospora vasinfecta EF Smith. Agric Biol Chem 52(6):1621–1623. https://doi.org/10.1080/00021369.1988.10868854
Nakajima H, Hamasaki T, Tanaka K, Kimura Y, Udagawa S-i, Horie Y (1989) Production of cyclosporin by fungi belonging to the genus Neocosmospora. Agric Biol Chem 53(8):2291–2292. https://doi.org/10.1080/00021369.1989.10869637
Ooijkaas LP, Weber FJ, Buitelaar RM, Tramper J, Rinzema A (2000) Defined media and inert supports: Their potential as solid-state fermentation production systems. Trends Biotechnol 18(8):356–360. https://doi.org/10.1016/S0167-7799(00)01466-9
Patocka J, Nepovimova E, Kuca K, Wu W (2020) Cyclosporine A: chemistry and toxicity - a review. Curr Med Chem 28(10):3925–3934. https://doi.org/10.2174/0929867327666201006153202
Řeháček Z, De-xiu Z (1991) The biochemistry of cyclosporin formation: a review. Process Biochem 26(3):157–166. https://doi.org/10.1016/0032-9592(91)80012-E
Shinde RB, Chauhan NM, Raut JS, Karuppayil SM (2012) Sensitization of Candida albicans biofilms to various antifungal drugs by cyclosporine A. Ann Clin Microbiol 11(1):27. https://doi.org/10.1186/1476-0711-11-27
Stiller C, Opelz G (1991) Should cyclosporine be continued indefinitely? Transplan Proc 1(1):36–40
Survase SA, Annapure US, Singhal RS (2009a) Statistical optimization for improved production of cyclosporin A in solid-state fermentation. J Microbiol Biotechnol 19(11):1385–1392. https://doi.org/10.4014/jmb.0901.0035
Survase SA, Shaligram NS, Pansuriya RC, Annapure US, Singhal RS (2009b) A novel medium for the enhanced production of cyclosporin A by Tolypocladium inflatum MTCC 557 using solid state fermentation. J Microbiol Biotechnol 19(5):462–467. https://doi.org/10.4014/jmb.0805.324
Survase SA, Kagliwal LD, Annapure US, Singhal RS (2011) Cyclosporin A - a review on fermentative production, downstream processing and pharmacological applications. Biotechnol Adv 29(4):418–435. https://doi.org/10.1016/j.biotechadv.2011.03.004
Tanseer S, Anjum T (2011) Modification of c and n sources for enhanced production of cyclosporin’a’by Aspergillus terreus. Braz J Microbiol 42(4):1374–1383. https://doi.org/10.1590/S2517-83822011000400019
Vervoort Y, Linares AG, Roncoroni M, Liu C, Steensels J, Verstrepen KJ (2017) High-throughput system-wide engineering and screening for microbial biotechnology. Curr Opin Biotechnol 46:120–125. https://doi.org/10.1016/j.copbio.2017.02.011
Weiser J, Matha V (1988) The insecticidal activity of cyclosporines on mosquito larvae. J Invertebr Pathol 51(1):92–93. https://doi.org/10.1016/0022-2011(88)90092-4
Wu C-J, Qiao H, Cui C-B, Yang Y, Xu R, Sun C-X, Li D-H, Li C-W (2019) Two new polyketides isolated from a diethyl sulphate mutant of marine-derived Penicillium purpurogenum G59. Nat Prod Res 33(20):2977–2981. https://doi.org/10.1080/14786419.2018.1514397
Yabutani T, Tsujimoto M, Ohira S, Shimizu S, Nakano H (2017) Strain improvement of Lentzea sp 7887 for higher yield per unit volume on hydroxylation of cyclosporine derivative FR901459. Biosci Biotechnol Biochem 81(7):1456–1459. https://doi.org/10.1080/09168451.2017.1314759
Yang X, Feng P, Yin Y, Bushley K, Spatafora JW, Wang C (2018) Cyclosporine biosynthesis in Tolypocladium inflatum benefits fungal adaptation to the environment. Mbio. https://doi.org/10.1128/mBio.01211-18
Zeligs MA (2016) Anti-parasitic methods and compositions utilizing diindolylmethane-related indoles. Google Patents.
Acknowledgements
Sundeep Jaglan gratefully acknowledges the Department of Science Technology (DST), Govt. of India for the financial assistance with Grant No. ECR/2017/001381
Funding
This article is funded by SERB DST INDIA, ECR/2017/001381, Sundeep Jaglan.
Author information
Authors and Affiliations
Contributions
VA executed the experimental work and prepared the original draft of the manuscript. MK did the analytical studies. SM did the formal analysis, supervision, review and editing of the manuscript. SJ contributed for the conceptualization, validation, formal analysis, supervision, editing of the manuscript and funding acquisition. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Abrol, V., Kushwaha, M., Mallubhotla, S. et al. Chemical mutagenesis and high throughput media optimization in Tolypocladium inflatum MTCC-3538 leads to enhanced production of cyclosporine A. 3 Biotech 12, 158 (2022). https://doi.org/10.1007/s13205-022-03219-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03219-x