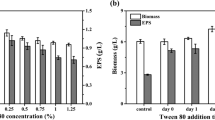
Abstract
In this study, the submerged culture conditions for exopolysaccharide (EPS) production by Ganoderma cantharelloideum M. H. Liu were screened and optimized, and the physicochemical and biological properties of EPS were investigated. Results showed that the glucose and tryptone were the best C and N sources for the maximum EPS production, respectively. Under the optimal culture conditions, the EPS production achieved 1.60 g/L at day 6 in a 5 L stirred tank reactor. Two purified fractions (i.e., Fr-I and Fr-II) were obtained from the G. cantharelloideum EPS by gel permeation chromatography. Fr-II had a higher yield (87.54%), carbohydrate (95.86%), and protein (3.07%) contents and lower molecular weight (74.56 kDa) than that of Fr-I. Both Fr-I and Fr-II were polysaccharide-protein complexes with different monosaccharide compositions and chemical structures. Fr-II also exhibited stronger radical scavenging abilities, antioxidant capacities, and α-amylase and α-glycosidase inhibitory activities in vitro than that of Fr-I. Therefore, Fr-II obtained from G. cantharelloideum EPS might be explored as potential natural functional components or supplements for applications in food, medicine, and cosmetics.








Similar content being viewed by others
References
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Bitter T, Muir HM (1962) A modified uronic acid carbazole reaction. Anal Biochem 4:330–334. https://doi.org/10.1016/0003-2697(62)90095-7
Cao J, Zhang HJ, Xu CP (2014) Culture characterization of exopolysaccharides with antioxidant activity produced by Pycnoporus sanguineus in stirred-tank and airlift reactors. J Taiwan Inst Chem E 45:2075–2080. https://doi.org/10.1016/j.jtice.2014.05.005
Chen L, Wang Z, Zhang B, Ge M, Ng H, Niu Y, Liu L (2019) Production, structure and morphology of exopolysaccharides yielded by submerged fermentation of Antrodia cinnamomea. Carbohyd Polym 205:271–278. https://doi.org/10.1016/j.carbpol.2018.10.070
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Calorimetric method for determination of sugars and related substances. Anal Chem 28:350–366. https://doi.org/10.1021/ac60111a017
Fang QH, Zhong JJ (2002) Submerged fermentation of higher fungus Ganoderma lucidum for production of valuable bioactive metabolites-ganoderic acid and polysaccharide. Biochem Eng J 10:61–65. https://doi.org/10.1016/S1369-703X(01)00158-9
He P, Geng L, Wang Z, Mao D, Wang J, Xu C (2012) Fermentation optimization, characterization and bioactivity of exopolysaccharides from Funalia trogii. Carbohyd Polym 89:17–23. https://doi.org/10.1016/j.carbpol.2012.01.093
Jaros D, Köbsch J, Rohm H (2018) Exopolysaccharides from Basidiomycota: Formation, isolation and techno-functional properties. Eng Life Sci 18:743–752. https://doi.org/10.1002/elsc.201800117
Kachlishvili E, Penninckx MJ, Tsiklauri N, Elisashvili V (2005) Effect of nitrogen source on lignocellulolytic enzyme production by white–rot basidiomycetes under solid-state cultivation. World J Microb Biot 22:391–397. https://doi.org/10.1007/s11274-005-9046-8
Kang Q, Chen S, Li S, Wang B, Liu X, Hao L, Lu J (2019) Comparison on characterization and antioxidant activity of polysaccharides from Ganoderma lucidum by ultrasound and conventional extraction. Int J Biol Macromol 124:1137–1144. https://doi.org/10.1016/j.ijbiomac.2018.11.215
Kim DH, Yang BK, Jeong SC, Park JB, Cho SP, Das S, Yun JW, Song CH (2001) Production of a hypoglycemic, extracellular polysaccharide from the submerged culture of the mushroom, Phellinus linteus. Biotechnol Lett 23:513–517. https://doi.org/10.1023/A:1010312513878
Kim HO, Yun JW (2005) A comparative study on the production of exopolysaccharides between two entomopathogenic fungi Cordyceps militaris and Cordyceps sinensis in submerged mycelial cultures. J Appl Microbiol 99:728–738. https://doi.org/10.1111/j.1365-2672.2005.02682.x
Meng F, Liu X, Jia L, Song Z, Deng P, Fan K (2010) Optimization for the production of exopolysaccharides from Morchella esculenta SO-02 in submerged culture and its antioxidant activities in vitro. Carbohyd Polym 79:700–704. https://doi.org/10.1016/j.carbpol.2009.09.032
Osińska-Jaroszuk M, Jarosz-Wilkołazka A, Jaroszuk-Ściseł J, Szałapata K, Nowak A, Jaszek M, Ozimek E, Majewska M (2015) Extracellular polysaccharides from ascomycota and basidiomycota: production conditions, biochemical characteristics, and biological properties. World J Microb Biot 31:1823–1844. https://doi.org/10.1007/s11274-015-1937-8
Park JP, Kim SW, Hwang HJ, Yun JW (2001) Optimization of submerged culture conditions for the mycelial growth and exo-biopolymer production by Cordyceps militaris. Lett Appl Microbiol 33:76–81. https://doi.org/10.1046/j.1472-765X.2001.00950.x
Peng YF, Zhang LN (2003) Characterization of a polysaccharide-protein complex from Ganoderma tsugae mycelium by size-exclusion chromatography combined with laser light scattering. J Biochem Biophys Methods 56(1–3):243–252. https://doi.org/10.1016/S0165-022X(03)00062-9
Shi M, Yang Y, Hu X, Zhang Z (2014) Effect of ultrasonic extraction conditions on antioxidative and immunomodulatory activities of a Ganoderma lucidum polysaccharide originated from fermented soybean curd residue. Food Chem 155:50–56. https://doi.org/10.1016/j.foodchem.2014.01.037
Siu KC, Chen X, Wu JY (2014) Constituents actually responsible for the antioxidant activities of crude polysaccharides isolated from mushrooms. J Funct Foods 11:548–556. https://doi.org/10.1016/j.jff.2014.08.012
Tang YJ, Zhong JJ (2001) Submerged fermentation of higher fungi for production of valuable bioactive metabolites. J East China Univ SciTechnol 27:704–711
Wang ZB, Chen BB, Luo L, Yan JK (2016) Fractionation, physicochemical characteristics and biological activities of polysaccharides from Pueraria lobata roots. J Taiwan Inst Chem E 67:54–60. https://doi.org/10.1016/j.jtice.2016.07.029
Wasser S (2002) Medicinal mushrooms as a source of antitumor and immunomodulating polysaccharides. Appl Microbiol Biotechnol 60:258–274. https://doi.org/10.1007/s00253-002-1076-7
Wu J, Shi S, Wang H, Wang S (2016) Mechanisms underlying the effect of polysaccharides in the treatment of type 2 diabetes: a review. Carbohyd Polym 144:474–494. https://doi.org/10.1016/j.carbpol.2016.02.040
Xing RG, Liu S, Guo ZY, Yu HH (2005) Relevance of molecular weight of chitosan and its derivatives and their antioxidant activities in vitro. Bioorgan Med Chem 13:1573–1577. https://doi.org/10.1016/j.bmc.2004.12.022
Xu CP, Kim SW, Hwang HJ, Yun JW (2006) Production of exopolysaccharides by submerged culture of an entomopathogenic fungus, Paecilomyces tenuipes C240 in stirred-tank and airlift reactors. Bioresour Technol 97:770–777. https://doi.org/10.1016/j.biortech.2005.01.042
Xu Y, Zhang X, Yan XH, Zhang JL, Wang LY, Xue H, Jiang GC, Ma XT, Liu XJ (2019) Characterization, hypolipidemic and antioxidant activties of degraded polysaccharides from Ganoderma lucidum. Int J Biol Macromol 135:706–716. https://doi.org/10.1016/j.ijbiomac.2019.05.166
Yan JK, Wang YY, Qiu WY, Shao N (2017) Three-phase partitioning for efficient extraction and separation of polysaccharides from Corbicula fluminea. Carbohyd Polym 163:10–19. https://doi.org/10.1016/j.carbpol.2017.01.021
Yan JK, Wu LX, Qiao ZR, Cai WD, Ma H (2019) Effect of different drying methods on the product quality and bioactive polysaccharides of bitter gourd (Momordica charantia L.) slices. Food Chem 271:588–596. https://doi.org/10.1016/j.foodchem.2018.08.012
Zhang J, Meng G, Zhai G, Yang Y, Zhao H, Jia L (2016) Extraction, characterization and antioxidant activity of polysaccharides of spent mushroom compost of Ganoderma lucidum. Int J Biol Macromol 82:432–439. https://doi.org/10.1016/j.ijbiomac.2015.10.016
Zhen D, Su L, Miao Y, Zhao F, Ren G, Mahfuz S, Song H (2018) Purification, partal characterization and inducing tumor cell apoptosis activity of a polysaccharide from Ganoderma applanatum. Int J Biol Macromol 115:10–17. https://doi.org/10.1016/j.ijbiomac.2018.03.062
Zheng JQ, Mao XJ, Geng LJ, Yang GM, Xu CP (2014) Production optimization, preliminary characterization and bioactivity of exopolysaccharides from Incutis tamaricis (Pat.) Fiasson & Niemela. J Taiwan Inst Chem E 45:725–733. https://doi.org/10.1016/j.jtice.2013.08.006
Acknowledgements
This work was supported by the Major Public Welfare Projects in Henan Province (201300110200), National Science Foundation of China (Grant No U1604176) and Program for Innovative Research Team (in Science and Technology) in University of Henan Province (Grant No 20IRTSTHN022).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declared no conflicts of interest relevant to this article.
Rights and permissions
About this article
Cite this article
Long, Z., Xue, Y., Ning, Z. et al. Production, characterization, and bioactivities of exopolysaccharides from the submerged culture of Ganoderma cantharelloideum M. H. Liu. 3 Biotech 11, 145 (2021). https://doi.org/10.1007/s13205-021-02696-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02696-w