Abstract
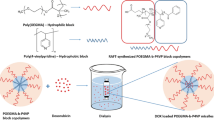
Asymmetric block copolymers PAAc-b-PAAm (DBC) and PAAm-b-PAAc-b-PAAm (TBC) comprised poly(acrylic acid) and polyacrylamide have been evaluated as potential vehicles for doxorubicin (Dox) targeted delivery. The processes of Dox release from the DBC and TBC micelles under the influence of the drug’s concentration gradient and various surrounding medium, such as NaCl solution and aqueous medium at pH= 5.75 and 9, were examined. The previously established tendency to significantly reduce the Dox encapsulation degree by the DBC and TBC micelles at the increase of the size of their “corona” was fully confirmed (Kunitskaya et al. 2018). Noticed that PAAm block’s lengthening complicated the Dox release against all environments under study. It was revealed that micelles which easily and effectively encapsulate Dox as easily "give away" the drug during dialysis. Increasing pH of the DBC and TBC solutions to 9 prevented drug release. It can be stipulated by enhancing interaction between Dox and micellar “core” caused by the ionization of –COOH groups contained in PAAc blocks. Preliminary biological investigations in vitro against human T-leukemia cells demonstrate essential anticancer activity of Dox/DBC and Dox/TDC compositions, as compared to pure Dox.





Similar content being viewed by others
References
Adams ML, Lavasanifar A, Gl K (2003) Amphiphilic block copolymers for drug delivery. J Pharm Sci 92(7):1343–1355. https://doi.org/10.1002/jps.10397
Bae J, Maurya A, Shariat-Madar Z, Murthy SN, Jo S (2015) Novel redox-responsive amphiphilic copolymer micelles for drug delivery: synthesis and characterization. AAPS J 17:1357–1368. https://doi.org/10.1208/s12248-015-9800-2
Bodratti AM, Alexandridis P (2018) Amphiphilic block copolymers in drug delivery: advances in formulation structure and performance. Expert Opin Drug Deliv 15(11):1085–1104. https://doi.org/10.1080/17425247.2018.1529756
Cabral H, Miyata K, Osada K, Kataoka K (2018) Block copolymer micelles in nanomedicine applications. Chem Rev 118(14):6844–6892. https://doi.org/10.1021/acs.chemrev.8b00199
Choucair A, Lavigueur C, Eisenberg A (2004) Polystyrene-b-poly(acrylic acid) vesicle size control using solution properties and hydrophilic block length. Langmuir 20(10):3894–3900. https://doi.org/10.1021/la035924p
Choucair A, Soo PL, Eisenberg A (2005) Active loading tunable release of doxorubicin from block copolymer vesicle. Langmuir 21:9308–9313. https://doi.org/10.1021/la050710o
Gref R, Minamitake Y, Peracchia MT, Trebetskoy VS, Torchilin VP, Langer R (1994) Biodegradable long-circulating polymeric nanospheres. Science 263:1600–1603. https://doi.org/10.1126/science.8128245
Hruby M, Konak C, Ulbrich K (2005) Polymeric micellar pH-sensitive drug delivery system for doxorubicin. J Controlled Release 103:137–148. https://doi.org/10.1016/j.jconrel.2004.11.017
Kumar V, Gupta B, Kumar G, Pandey M, Aiazian E, Parmar VS, Kumar J, Watterson AC (2010) Novel PEGylated amdhiphilic copolymers as nanocarriers for drug delivery: synthesis, characterization and curcum encapsulation. J Macromol Sci 47:1154–1160. https://doi.org/10.1080/10601325.2010.518549
Kunitskaya L, Zheltonozhskaya T, Permyakova N (2012) Block Copolymers of Polyacrilamide and Poly(ethylene oxide as nanocarriers for drug delivery: micellization and bulk structure. Macromol Symp 1:317–318. https://doi.org/10.1002/masy.201100070
Kunitskaya LR, Zheltonozhskaya TB, Aleinichenko VA, Roshal AD, Khayetsky IK (2011) The self-assembly of diblock copolymers MePEG-b-PAAm into micellar structures and their interaction with doxorubicin. Mol Cryst Liq Cryst 536:398–404. https://doi.org/10.1080/15421406.2011.538603
Kunitskaya LR, Zheltonozhskaya TB, Stoika RS, Klymchuk DO (2017) Compositions of anticancer drug with micellar nanocarriers and their cytotoxicity. French-Ukrainian J Chem 5:103–120. https://doi.org/10.17721/fujcV5I2P103-120
Kunitskaya L, Zheltonozhskaya T, Destarac M, Mazieres S (2017) Block copolymers containing polyacrylamide and polyacrylic acid: bulk structure and hydrogen bonds system. Mol Cryst Liq Cryst 642:89–98. https://doi.org/10.1080/15421406.2016.1255522
Kunitskaya L, Zheltonozhskaya T, Destarac M, Mazieres S, Klymchuk DO (2018) Micellar structures of PAAc/PAAm block copolymers in the processes of doxorubicin encapsulation. Mol Cryst Liq Cryst 670(1):168–177. https://doi.org/10.1080/15421406.2018.1542077
Layre A, Couvreur H, Chacun H, Richard J, Passirani C, Requier D, Benoit JP, Gref R (2006) Novel composite core-shell nanoparticles as busulfan carriers. J Control Release 111:271–280. https://doi.org/10.1016/j.jconrel.2006.01.002
Nakanishi T, Fukushima S, Okamoto K, Suzuki M, Matsumura Y, Yokoyama M, Okano T, Sakurai Y, Kataoka K (2001) Development of the polymer micelle carrier system for doxorybicin. J Controlled Rel 74:295–302
Noh T, Kook YH, Park C et al (2008) Block copolymer micelles conjugated with anti-EGFR antibody for targeted delivery of anticancer drug. J Polym Sci 46:7321–7330. https://doi.org/10.1002/pola.23036
Papadimitrou S, Bikiaris D (2009) Novell self-assembled core-shell nanoparticles based on crystalline amorphous moieties of aliphatic copolyesters for efficient controlled drug release. J Control Release 138:177–184. https://doi.org/10.1016/j.jconrel.2009.05.013
Soo PL, Luo L, Mayasinger D, Eisenberg A (2002) Incorporation and release of hydrophobic probes in biocompatible polycaproloctone-block-polyethelene oxide micelles: implication of drug delivery. Langmuir 18:9996–10004. https://doi.org/10.1021/la026339b
Wang J, Yao K, Wang C, Tang C, Jiang X (2013) Synthesis and drug delivery of novel amphiphilic block copolymers containing hydrophobic dehydroabietic moiety. J Mater Chem 1(B):2324–2332. https://doi.org/10.1039/C3TB20100G
Zheltonozhskaya T, Permyakova N, Momot L (2009) Intramolecular polycomplexes in block and graft copolymers. In: Khutoryanskiy V, Staikos G (eds) Hydrogen-bonded interpolymer complexes. Formation, structure and application. World Scientific, New Jersey, pp 5–153
Zheltonozhskaya T, Partsevskaya S, Fedorchuk S, Klymchuk D, Gomza Y, Permyakova N, Kunitskaya L (2013) Micellar nanocontainers based on PAAm-b-PEO-b-PAAm triblock copolymers for poorly soluble drugs. Eur Polym J 49:405–418. https://doi.org/10.1016/j.eurpolymj.2012.10.28
Acknowledgements
We thank Dr. Mathias Destarac and Dr. Stéphane Masieres from Université Paul Sabatier (Toulose, France) for use of their laboratory during the synthetic part of the experiment as well as the Institute of Cell Biology NAS of Ukraine for providing biological investigations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest:
On behalf of all authors, the corresponding author states that there no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kunitskaya, L., Zheltonozhskaya, T., Gerda, V. et al. Development polymeric micellar system for targeted delivery of antitumor drugs. Appl Nanosci 12, 1315–1322 (2022). https://doi.org/10.1007/s13204-021-01876-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-021-01876-7