Abstract
Chronic kidney disorder may even be a significant health concern in Delta region particularly Dakahlia district, Egypt that’s characterized by permanent loss of kidney function through the years. Mercury considered as a silent threat to the environment and human life, through the world. This work purposes to access the effects of mercury overload in domestic water on the renal function. The study performed in spring 2019 on one hundred participants and conducted in two main areas that are getting ready to Lake Manzala. 100 ml of water, 40 ml of urine and 10 ml of blood samples from patients suffering from kidney diseases and healthy individuals as control group. These samples will be processed and analyzed to determine the incidence of active kidney disease. Mean levels of mercury in water, and urine samples of case group participants exceeded standard references and were significantly higher compared to control group (p = 0.0004 and 0.023 respectively) denoting more exposure to mercury. Proteinuria was significantly higher (p = 0.031) whereas, hemoglobin and hematocrit became significantly lower (p = 0.039 and 0.044 respectively) among case group participants which might be an indicator for kidney affection due to mercury exposure. Mercury overload in domestic water can cause renal impairment. The study showed that urinalysis for Proteinuria and hemoglobin values is commonly used as a screening test to detect kidney damage caused by mercury poisoning. Measures to prevent environmental pollutants and continuous monitoring of water sources in these districts are clearly necessary.
Similar content being viewed by others
Introduction
In many countries, especially developing countries, the quality of domestic water is not ideal, and the poor quality has led to many water-borne diseases. Contamination of Lake Manzala, Egypt with mercury has been previously reported (Salam et al. 2019). The kidneys simply targeted for mercury toxicity with high accumulation notably among the areas of the proximal tubules (Freiberg et al. 1986). Mercury is crucial and prevalent environmental toxicant that induces nephrotic effects. It is also a noxious metal of explicit concern with extremely nephrotoxic effects (Christy et al. 2017). Mercury (Hg) is a persistent bio-accumulative toxic metal with unique physical and chemical properties, which arouses public health concern because its natural and man-made spread still poses high risks to human and environmental health. It is directly disposed into rivers and water systems. The most common forms of organic mercury in ecosystems are methyl- and ethyl mercury compounds, much more toxicological information is available for methyl mercury than for ethyl mercury (Clarkson and Magos 2006). Methyl mercury readily transported by water into the aquatic ecosystems. Humans may expose to mercury through water, air or consumption of contaminated food products (Zhang et al. 2017). The kidneys are easily targeted for mercury toxicity with high accumulation particularly in the areas of the proximal tubules (Nordman et al. 1986). The study of Reyes et al. (2013) confirmed that because the proximal tubule cells are damaged and can enter freely through the glomerular filter, lysosomal sensitivity may also be attributed to proteinuria. Nasiruddin et al. (2018) confirmed changes in mercury, which can lead to proteinuria and renal dysfunction. Urine mercury testing offers additional purposeful image of exposure and additional correct as a result of the mercury is additional focused (Byeong-Jin et al. 2016). Preliminary symptoms of mercury exposure are often assessed by examining kidney function through an easy urine test, and hemoglobin levels are often determined with a simple blood test kit, (Ekawanti and Krisnayanti 2015). Research on the same issue is rarely published, usually related to the geological characteristics of the territory. The research results of El-Kowrany et al. (2016) report, almost all samples collected from source water have mercury content exceeding the maximum allowable concentration of drinking water. People living in these places eat fish or other products, and mercury can cause a range of diseases from mild to the most serious. The health agency must call attention to the safe water supply in the place. We conducted this study to assess mercury exposure among inhabitants of our locality who are on the brink of Lake Manzala and to assess the feasibility of simple diagnostic tools to detect health consequences of such exposure. The present study, emphasis on the effects of mercury overload in domestic water on renal function.
Materials and methods
Participants
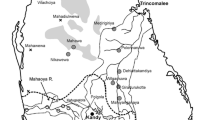
Two areas of Dakahlia Governorate (Fig. 1) show the significant differences between kidney disease patient and non-patient samples. The study was conducting on 100 participants in the spring of 2019 and was conducting in two main areas that are getting ready to Lake Manzala. Representative samples from 100 participants at the time of the study, most of them were male (70%) Table1. A structured questionnaire was using to collect socio-demographic information from the participants. The consumption of water volume isn’t identified to be a risk factor (Roudakova and Monga, 2014). Area 1 close to fishing activities (65 anglers, kidney diseases patients), while area 2 (35 non-kidney diseases patients).
Methods
For each participant; water sample (100 ml) will collect. Before sample collection, tap water was flown for an adequate time (1–2 min) to release the pipe. The concentration of the heavy metal studied (Mercury, Hg) was estimated using a graphite-furnace atomic-absorption spectrometer (Buck scientific company, USA) utilizing the digestion technique based on the APHA standard (2012). Before study process, consent was obtained from the participants. Forty ml of random spot urine and 10 ml of blood samples from patients suffering from kidney diseases as well as from healthy individuals of same age and gender serving as the control group. These samples will process and analyzed in same lab for active kidney diseases incidence will be determined. Calibration standards and quality control samples are prepared fresh every day. Reference standard material used to evaluate the precision and accuracy of the program. The data coded, entered and processed on computer using SPSS program. Continuous variables are expressing as mean ± standard deviation (SD). Statistical differences between mean groups were administered using analysis of variance, where p < 0.05 is significant.
Mechanism↓ of kidney damage
The accumulation of Hg2+ in renal tubular cells, primarily those of the proximal tubule, occurs rapidly so, it appears to be the most sensitive to the toxic effects of mercury and is usually the first segment of the nephron affected by exposure to mercuric compounds (Dewanjee et al. 2013). Mercury-related kidney damage will be due to renal tubular dysfunction with elevated albumin, metastasis, and nephrotic syndrome (Fig. 2) with the membranous nephropathy pattern (Zalups 2000). Various reports indicate that mercury can cause various kidney damage, including proteinuria, glomerular disease, and membranous glomerulonephritis (Miller et al. 2013).
Results and discussion
Hundred participants were recruited for this study. Tables 2, 3 show means levels of mercury in water and urine, protein in urine and Proteinuria as well as the characteristics of haematological parameters. Case (patients) group (65): regarding area1 (near the lake Manzala), diagnosed with kidney diseases patients supported physical and clinical examination as a main group. The principal activity of this group participant became fishing.
The study assessed the concentrations of mercury (Hg) obtained from surface and ground water bodies used for drinking and other domestic purposes. The results confirmed that the kidney diseases patients had compatible values of mercury in water and urine exceeded than the permissible limits of WHO (2017), Iyengar and Woittiez (1988). Mean levels (mg/l) of mercury in water samples have been 0.007 of the case group who have ages over 60 with kidney diseases, exceeded WHO (2017) permissible limits of 0.006 for mercury. That is frequently according to Habiba et al. (2016); Song et al. (2016) who stated that the renal burden of mercury increases with age. NKF (2016) reported that 30% of aged 65 and older can also develop chronic kidney disorder later in life. Generally, mercury concentrations were high in water bodies that were close to the fishing activities in this community. Fish seems to be the main source of human methyl mercury poisoning. Through unclear mechanisms, various fish species tend to have a high rate of methyl mercury bioaccumulation (Mozaffarian and Rimm, 2006). The results confirmed that the case group had low Hb and Hct concentrations due to chronic mercury intoxication. This finding became almost like that of previous studies reporting that mercury exposure could decrease Hb levels (Calisi et al. 2011). As kidney function declines, this affects the number of red blood cells (Franco et al. 2005); decreases in either Hb or Hct levels. The significance of such changes in renal functions is of toxicological importance. High urinary mercury levels within the anglers indicate that a pathological process within the kidney can also occur due to mercury. Within the study location, most of the water sources for lifestyle come from an equivalent well. Among the case group participants, protein in urine became 1.74 ± 1.05 g / L. This considered indicating Proteinuria since this level was + 2 and ≥ 0.3 g/L (Clarkson et al. 2003). Different chelating agents are recommended for acute inorganic mercury poisoning. Sometime, chelation therapy might consider on the basis of dose (ASTDR 2017). The FDA approved DMSA for use in children. Doctors also recommend the use of glutathione and N-acetyl cysteine (NAC), but they promote the deposition of Hg in the brain and kidneys (Sharma et al. 2014). Owing to the fatal side effects of chelation therapy, simultaneous treatment of combination therapy is preferable to mono-therapy, even if the dose is lower. In addition, after thorough investigation and clinical trials, vitamins and minerals and potent antioxidants from plants may be used to treat acute exposure (Nasiruddin et al. 2018).
Conclusions
Our research spots the light on the problem of mercury toxicity within a cohort of inhabitants on the fringe of Lake Manzala. This study shows that the inhabitants are exposed to mercury toxicity from its possible consecutive health hazards. Furthermore, the study showed that urinalysis for Proteinuria and hemoglobin values is commonly used as a screening test to detect kidney damage caused by mercury poisoning. The possibility of such a thing simple tools is paramount in developing countries with limited resources. The health risks and consequences of environmental pollution must be considered, and therefore the present study will help authorities have an effect on and solve this issue. Chelating therapy is the preferred medical treatment. These treatments also play an important role in renal detoxification associated with chelating agent nephrotoxicity. Continuous monitoring of the quality of water resources in these districts recommended and additional studies are clearly necessary.
Change history
22 July 2022
Missing Open Access funding information has been added in the Funding Note.
References
Agency for Toxic Substances and Disease Registry (ATSDR) (2017), ATSDR - Medical Management Guidelines (MMGs): Mercury, (n.d.), https://www.atsdr.cdc.gov/MMG/MMG.asp?id=106&tid=24
APHA (2012) Standard methods for the examination of water and wastewater (22nd Ed) American Public Health Association, American Water Works Association (AWWA), and Water Environment Federation (WEF): Washington, DC
Bridges CC, Zalups RK (2017) The aging kidney and the nephrotoxic effects of mercury. J Toxicol Environ Health Part B Crit Rev 20(2):55–80
Calisi A, Lionetto MG, Sanchez-Hernandez JC, Schettino T (2011) (2011) Effect of heavy metal exposure on blood hemoglobin concentration and met hemoglobin percentage in Lumbricus terrestris. Eco-Toxicol 20(4):847–854
Clarkson TW, Magos L (2006) The toxicology of mercury and its chemical compounds. Crit Rev Toxicol 36(8):609–662
Clarkson TW, Magos L, Myers GJ (2003) The toxicology of mercury-current exposures and clinical manifestations. N Engl J Med 349(18):1731–1737
Dewanjee S, Sahu R, Karmakar S, Gangopadhyay M (2013) Toxic effects of lead exposure in Wistarrats: involvement of oxidative stress and the beneficial role of edible jute (Corchorus olitorius) leaves. Food Chem Toxicol 55:78–91
Ekawanti A, Krisnayanti BD (2015) Effect of mercury exposure on renal function and hematological parameters among Artisanal and small-scale gold miners at Sekotong, West Lombok Indonesia. J Health Pollut 5(9):25–32. https://doi.org/10.5696/2156-9614-5-9.25
El-Kowrany SI, El- Zamarany EA, El-Nouby KA et al (2016) Water pollution in the Middle Nile Delta, Egypt: an environmental study. J Adv Res 7(5):781–794. https://doi.org/10.1016/j.jare.2015.11.005
Franco A, Budiha MV, Fikfak MD (2005) Long-term effects of elemental mercury on renal function in miners of Idrija Mercury Mine. Ann Occup Hyg 49(6):521–527. https://doi.org/10.1093/annhyg/mei022
Freiberg L, Nordberg G, Vouk V (1986) Handbook on the toxicology of metals. Elsevier Science Publishers, New York, NY, pp 676–718
Habiba G, Abebe G, Bravo AG, Ermias D, Staffan A, Bishop K (2016) Mercury human exposure in populations living around Lake Tana (Ethiopia). Biol Trace Elem Res 175(2):237–243. https://doi.org/10.1007/s12011-016-0745-9
Iyengar GV, Woittiez JRW (1988) Trace elements in human clinical specimen; evaluation of literature data to identity references values. Clinic Chem 34(3):474–481
Miller S, Pallan S, Gangji AS, Lukic D, Clase CM (2013) Mercury-associated nephrotic syndrome: a case report and systematic review of the literature. Am J Kidney Dis 62(1):135–138
Mozaffarian D, Rimm EB (2006) Fish intake, contaminants, and human health: evaluating the risks and the benefits. JAMA 296(15):1885–1899
Nasiruddin RM, Tangpong J, Masudur Md, Md R (2018) Toxicol-dynamics of lead cadmium, mercury and arsenic- induced kidney toxicity and treatment strategy: a mini review. Toxicol Rep 5:704–713. https://doi.org/10.1016/j.toxrep.2018.05.012
National Kidney Foundation (NKF) (2016) Diabetes - A major risk factor for kidney disease, (2016) Available from https://www.kidney.org/atoz/content/diabetes
Nordman H, Berlin M (1986) Titanium. In: Fir Berg L, Nordberg GF, Vou KV (eds) Handbook on the toxicology of metals. Elsevier Science Publishers, New York, NY, pp 676–718
Reyes JL, Molina-Jijón E, Rodríguez-Muñoz R et al (2013) Tight junction proteins and oxidative stress in heavy metals-induced nephrotoxicity. Biomed Res Int. https://doi.org/10.1155/2013/730789
Roudakova K, Monga M (2014) The evolving epidemiology of stone disease, Indian. J Ur 30(1):44–48. https://doi.org/10.4103/0970-1591.124206
Salam KI, Abed El-Ghany SM, Mohammed MA (2019) Heavy metal residues in some fishes from Manzala Lake, Egypt, and their health-risk assessment. J Food Sci 84(7):1957–1965. https://doi.org/10.1111/1750-3841.14676.Epub
Sharma B, Singh S, Siddiqi NJ (2014) Biomedical implications of heavy metals induced imbalances in redox systems. Biomed Res Int. https://doi.org/10.1155/2014/640754
Song Y, Lee CK, Kim KH et al (2016) Factors associated with total mercury concentrations in maternal blood, cord blood, and breast milk among pregnant women in Bussan, Korea. Asia Pac J Clin Nutr 25(2):340–349. https://doi.org/10.6133/apjcn.2016.25.2.16
World Health Organization (WHO 2017) Guidelines for drinking-water quality, Fourth edition incorporating the first addendum; pp 1–631
Ye BJ, Kim BG, Jeon MJ et al (2016) (2016) Evaluation of mercury exposure level, clinical diagnosis and treatment for mercury intoxication. Ann Occup Environ Med. https://doi.org/10.1186/s40557-015-0086-8
Zalups RK (2000) Molecular interactions with mercury in the kidney. Pharmacol Rev 52:113–143
Zhang Z, Gao X, Guo M, Jiang H, Cao Y, Zhang N (2017) The protective effect of Baicalin against lead-induced renal oxidative damage 1 in mice. Biol Trace Elem Res 175:129–135
Acknowledgements
I sincerely thank all people’s help for their cooperation and assistance throughout this study.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). The author(s) received no specific funding for this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
I have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.