Abstract
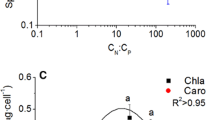
We investigated the physiological response of the symbiotic dinoflagellate Pelagodinium béii to exposure to ultraviolet (UV) radiation, focusing on synthesis and accumulation of UV-absorbing substances (mycosporine-like amino acids, MAAs). For the culture experiments, we used P. béii isolated from the foraminifer Globigerinoides sacculifer, which ubiquitously inhabits the surface waters of oligotrophic oceans. The P. béii produced mainly shinorine, palythenic acid, and mycosporine-glycine as MAAs, and they accumulated MAAs within cells in proportion to UVB exposure within 12 h after UV irradiation. There was no marked accumulation of MAAs with irradiation by only UVA. We observed decreases in growth rate, cellular chlorophyll a content, and photosynthetic activity within 2 days, even under low UVB exposure. Our laboratory experiments showed that P. béii could synthesize MAAs in response to UVB exposure, but that cell growth and photosynthetic function could be inhibited by UVB levels in the surface waters of the natural environment. Our results suggested that free-living P. béii could not survive in the surface waters inhabited by the holobiont of P. béii and its host Globigerinoides sacculifer, even if the stress from nutrient limitation was reduced.







Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Amador-Castro F, Rodriguez-Martinez V, Carrillo-Nieves D (2020) Robust natural ultraviolet filters from marine ecosystems for the formulation of environmental friendlier bio-sunscreens. Sci Total Environ 749:141576. https://doi.org/10.1016/j.scitotenv.2020.141576
Anderson OR (2012) Living together in the plankton: A survey of marine protist symbioses. Acta Protozool 52:1–10. https://doi.org/10.4467/16890027AP.13.0019.1116
Anderson OR, Bé AWH (1976) The ultrastructure of a planktonic foraminifer, Globigerinoides sacculifer (Brady), and its symbiotic dinoflagellates. J Foram Res 6:1–21. https://doi.org/10.2113/gsjfr.6.1.1
Banaszak AT, Santos MGB, LaJeunesse TC, Lesser MP (2006) The distribution of mycosporine-like amino acids (MAAs) and the phylogenetic identity of symbiotic dinoflagellates in cnidarian hosts from the Mexican Caribbean. J Exp Mar Biol Ecol 337:131–146. https://doi.org/10.1016/j.jembe.2006.06.014
Bé AWH (1977) An ecological, zoogeographic and taxonomic review of recent planktonic foraminifera. In: Ramsay ATS (ed) Oceanic micropaleontology, vol 1. Academic Press, pp 1–100
Bé AWH, Spero HJ, Anderson OR (1982) Effects of symbiont elimination and reinfection on the life processes of the planktonic foraminifer Globigerinoides sacculifer1. Mar Biol 70:73–86. https://doi.org/10.1007/BF00397298
Bé AWH, Anderson OR, Faber WW Jr, Caron DA (1983) Sequence of morphological and cytoplasmic changes during gametogenesis in the planktonic foraminifer Globigerinoides sacculifer (Brady). Micropaleontology 29(3):310–325. https://doi.org/10.2307/1485737
Booth CR, Morrow JH (1997) The penetration of UV into natural waters. Photochem Photobiol 65(2):254–257. https://doi.org/10.1111/j.1751-1097.1997.tb08552.x
Britt AB (1996) DNA damage and repair in plants. Annu Rev Plant Physiol Plant Mol Biol 47:75–100. https://doi.org/10.1146/annurev.arplant.47.1.75
Buma AGJ, Engelen AH, Gieskes WWC (1997) Wavelength-dependent induction of thymine dimers and growth rate reduction in the marine diatom Cyclotella sp. exposed to ultraviolet radiation. Mar Ecol Prog Ser 153:91–97. https://doi.org/10.3354/meps153091
Callone AI, Carignan M, Montoya NG, Carreto JI (2006) Biotransformation of mycosporine like amino acids (MAAs) in the toxic dinoflagellate Alexandrium tamarense. J Photochem Photobiol, B 84(2006):204–212. https://doi.org/10.1016/j.jphotobiol.2006.03.001
Caron DA (2000) Symbiosis and mixotrophy among pelagic microorganisms. In: Kirchman DL (ed) Microbial ecology of the oceans. Wiley Series in Ecological and Applied Microbiology, New York, pp 495–523
Carreto JI, Carignan MO, Montoya NG (2001) Comparative studies on mycosporine-like amino acids, paralytic shellfish toxins and pigment profiles of the toxic dinoflagellates Alexandrium tamarense, A. catenella and A. minutum. Mar Ecol Prog Ser 223:49–60. https://doi.org/10.3354/meps223049
Carreto JI, Carignan MO, Montoya NG (2005) A high-resolution reverse-phase liquid chromatography method for the analysis of mycosporine-like amino acids (MAAs) in marine organisms. Mar Biol 146:237–252. https://doi.org/10.1007/s00227-004-1447-y
Dunlap CW, Shick MJ (1998) Ultraviolet radiation-absorbing mycosporine-like amino Acids in coral reef organisms. A biochemical and environmental perspective. J Phycol 34:418–430. https://doi.org/10.1046/j.1529-8817.1998.340418.x
Fujiki T, Takagi H, Kimoto K, Kurasawa A, Yuasa T, Mino Y (2014) Assessment of algal photosynthesis in planktic foraminifers by fast repetition rate fluorometry. J Plankton Res 36(6):1403–1407. https://doi.org/10.1093/plankt/fbu083
Häder D-P (2000) Effects of solar UV-B radiation on aquatic ecosystems. Adv Space Res 26(12):2029–2040. https://doi.org/10.1016/S0273-1177(00)00170-8
Häder D-P, Kumar HD, Smith RC, Worrest RC (1998) Effects on aquatic ecosystems. J Photochem Photobiol B, Biol 46(1–3):53–68. https://doi.org/10.1016/S1011-1344(98)00185-7
Häder D-P, Williamson CE, Wängberg S-Å, Rautio M, Rose KC, Gao K, Helbling EW, Sinhah RP, Worresti R (2015) Effects of UV radiation on aquatic ecosystems and interactions with other environmental factors. Photochem Photobiol Sci 14:108. https://doi.org/10.1039/C4PP90035A
Hannach G, Sigleo AC (1998) Photoinduction of UV-absorbing compounds in six species of marine phytoplankton. Mar Ecol Prog Ser 174:207–222. https://doi.org/10.3354/meps174207
Hillebrand H, Dürselen C-D, Kirschtel D, Pollingher U, Zohary T (1999) Biovolume calculation for pelagic and benthic microalgae. J Phycol 35:403–424. https://doi.org/10.1046/j.1529-8817.1999.3520403.x
Jeffrey SW, MacTavish HS, Dunlap WC, Vesk M, Groenewoud K (1999) Occurrence of UVA- and UVB-absorbing compounds in 152 species (206 strains) of marine microalgae. Mar Ecol Prog Ser 189:35–51. https://doi.org/10.3354/meps189035
Klisch M, Häder D-P (2002) Wavelength dependence of mycosporine-like amino acid synthesis in Gyrodinium dorsum. J Photochem Photobiol B: Biol 66:60–66. https://doi.org/10.1016/S1011-1344(01)00276-7
Kolber Z, Falkowski PG (1993) Use of active fluorescence to estimate phytoplankton photosynthesis in situ. Limnol Oceanogr 38:1646–1665. https://doi.org/10.4319/lo.1993.38.8.1646
Kolber Z, Zehr J, Falkowski PG (1988) Effects of growth irradiance and nitrogen limitation on photosynthetic energy conversion in photosystem II. Plant Physiol 88:923–929. https://doi.org/10.1104/pp.88.3.923
Kuwahara VS, Taguchi S (2015) Estimating the diurnal cycle and daily insolation of ultraviolet and photosynthetically active radiation at the sea surface. Photochem Photobiol 91(5):1103–1111. https://doi.org/10.1111/php.12474
Llewellyn CA, Airs RL (2010) Distribution and abundance of MAAs in 33 species of microalgae across 13 classes. Mar Drugs 8:1273–1291. https://doi.org/10.3390/md8041273
Murray J (1897) On the distribution of the pelagic foraminifera at the surface and on the floor of the ocean. Nat Sci 11:17–27
Porra RJ, Thompson WA, Kriedemann PE (1989) Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys Acta 975:384–394. https://doi.org/10.1016/S0005-2728(89)80347-0
Rastogi PR, Richa SRP, Singh SP, Häder D-P (2010) Photoprotective compounds from marine organisms. J Ind Microbiol Biotechnol 37(6):537–558. https://doi.org/10.1007/s10295-010-0779-5
Shaked Y, de Vargas C (2006) Pelagic photosymbiosis: rDNA assessment of diversity and evolution of dinoflagellate symbionts and planktonic foraminiferal hosts. Mar Ecol Prog Ser 325:59–71. https://doi.org/10.3354/meps325059
Shick JM (2004) The continuity and intensity of ultraviolet irradiation affect the kinetics of biosynthesis, accumulation, and conversion of mycosporine-like amino acids (MAAs) in the coral Stylophora pistillata. Limnol Oceanogr 49(2):442–458. https://doi.org/10.4319/lo.2004.49.2.0442
Shick JM, Dunlap WC (2002) Mycosporine-like amino acids and related gadusols: biosynthesis, accumulation, and UV-protective functions in aquatic organisms. Annu Rev Physiol 64:223–262. https://doi.org/10.1146/annurev.physiol.64.081501.155802
Shick JM, Lesser MP, Jokiel PL (1996) Effects of ultraviolet radiation on corals and other coral reef organisms. Global Change Biol 2:527–545. https://doi.org/10.1111/j.1365-2486.1996.tb00065.x
Shick JM, Romaine-Lioud S, Romaine-Lioud S, Ferrier-Pagès C, Gattuso J-P (1999) Ultraviolet-B radiation stimulates shikimate pathway-dependent accumulation of mycosporine-like amino acids in the coral Stylophora pistillata despite decreases in its population of symbiotic dinoflagellates. Limnol Oceanogr 44(7):1667–1682. https://doi.org/10.4319/lo.1999.44.7.1667
Siano R, Montresor M, Probert I, Not F, de Vargas C (2010) Pelagodinium gen. Nov and P. béii comb. nov., a dinoflagellate symbiont of planktonic foraminifera. Protist 161(3):385–399. https://doi.org/10.1016/j.protis.2010.01.002
Sinha RP, Singh SP, Häder D-P (2007) Database on mycosporines and mycosporine-like amino acids (MAAs) in fungi, cyanobacteria, macroalgae, phytoplankton and animals. J. Photochem. Photobiol B, Biol 89:29–35. https://doi.org/10.1016/j.jphotobiol.2007.07.006
Taira H, Taguchi S (2017) Cellular Mycosporine-like amino acids protect photosystem II of the Dinoflagellate Scrippsiella sweeneyae from ultraviolet radiation damage. J Photochem Photobiol B, Biol 174:27–34. https://doi.org/10.1016/j.jphotobiol.2017.07.015
Takagi H, Kimoto K, Fujiki T, Kurasawa A, Moriya K, Hirano H (2016) Ontogenetic dynamics of photosymbiosis in cultured planktic foraminifers revealed by fast repetition rate fluorometry. Mar Micropaleontol 122:44–52. https://doi.org/10.1016/j.marmicro.2015.10.003
Takagi H, Kimoto K, Fujiki T, Moriya K (2018) Effect of nutritional condition on photosymbiotic consortium of cultured Globigerinoides sacculifer (Rhizaria, Foraminifera). Symbiosis 76:25–39. https://doi.org/10.1007/s13199-017-0530-3
Takagi H, Kimoto K, Fujiki T, Saito H, Schmidt C, Kucera M, Moriya K (2019) Characterizing photosymbiosis in modern planktonic foraminifera. Biogeosciences 16:3377–3396. https://doi.org/10.5194/bg-16-3377-2019
Tedetti M, Sempéré R (2006) Penetration of ultraviolet radiation in the marine environment. A review. Photochem and Photobiol 82:389–397. https://doi.org/10.1562/2005-11-09-IR-733
Thimijan RW, Heins RD (1983) Photometric, radiometric, and quantum light units of measure: a review of procedures for interconversion. HortScience 18(6):818–822
Whitehead K, Hedges JI (2002) Analysis of mycosporine-like amino acids in plankton by liquid chromatography electrospray ionization mass spectrometry. Mar Chem 80:27–39. https://doi.org/10.1016/S0304-4203(02)00096-8
Whitehead K, Hedges JI (2003) Electrospray ionization tandem mass spectrometric and electron impact mass spectrometric characterization of mycosporine-like amino acids. Rapid Commun Mass Spectrom 17:2133–2138. https://doi.org/10.1002/rcm.1162
Wu D, Hu Q, Yan Z, Chen W, Yan C, Huang X, Zhang J, Yang P, Deng H, Wang J (2012) Structural basis of ultraviolet-B perception by UVR8. Nature 484:214–219. https://doi.org/10.1038/nature10931
Acknowledgements
We thank Dr. K. Kimoto and Dr. V. S. Kuwahara for their valuable discussions. We appreciate the constructive suggestions of Prof. T. Nishino at Kobe University. We also thank Ms. T. Amimoto of the Natural Science Center for Basic Research and Development (N-BARD) at Hiroshima University for MS analysis. This work was partially supported by Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (grant numbers 16K00532 and 22H02428).
Funding
This work was partially supported by Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (grant numbers 16K00532 and 22H02428).
Author information
Authors and Affiliations
Contributions
A. Y. Hoshihara and T. Fujiki designed the study. A. Y. Hoshihara carried out the experiments. A. Y. Hoshihara, S. Shigeoka and M. Hirayama analyzed the samples. All authors interpreted the results and wrote the paper.
Corresponding author
Ethics declarations
Conflicts of interest/Competing interests
Not applicable.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yamamoto Hoshihara, A., Fujiki, T., Shigeoka, S. et al. Physiological response of the symbiotic dinoflagellate Pelagodinium béii to ultraviolet radiation: synthesis and accumulation of mycosporine-like amino acids. Symbiosis 86, 281–292 (2022). https://doi.org/10.1007/s13199-022-00839-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-022-00839-y