Abstract
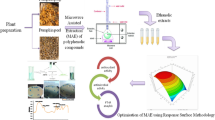
The present study compares three methods viz. microwave assisted extraction (MAE), ultrasonic-assisted extraction (UAE) and conventional solvent extraction (CSE) for extraction of phenolic compounds from black carrot pomace (BCP). BCP is the major by-product generated during processing and poses big disposal problem. Box–Behnken design using response surface methodology was employed to investigate and optimize the MAE of phenolics, antioxidant activity and colour density from BCP. The conditions for maximum recovery of polyphenolics were: microwave power (348.07 W), extraction time (9.8 min), solvent–solid ratio (19.3 mL/g) and ethanol concentration (19.8%). Under these conditions, the extract contained total phenolic content of 264.9 ± 10.02 mg gallic acid equivalents (GAE)/100 mL, antioxidant capacity (AOC) of 13.14 ± 1.05 µmol Trolox equivalents (TE)/mL and colour density of 68.63 ± 5.40 units. The total anthocyanin content at optimized condition was 753.40 ± 31.6 mg/L with low % polymeric colour of 7.40 ± 0.42. At optimized conditions, MAE yielded higher colour density (68.63 ± 5.40), polyphenolic content (264.9 ± 10.025 mg GAE/100 mL) and AOC (13.14 ± 1.05 µmol TE/mL) in a short time as compared to UAE and CSE. Overall results clearly indicate that MAE is the best suited method for extraction in comparison to UAE and CSE. The phenolic rich extract can be used as an effective functional ingredient in foods.



Similar content being viewed by others
References
Alara OR, Abdurahman NH, Afolabi HK, Olalere OA (2018) Efficient extraction of antioxidants from Vernonia cinerea leaves: comparing response surface methodology and artificial neural network. J Basic Appl Sci 7:276–285
Arvindekar AU, Laddha KS (2015) An efficient microwave-assisted extraction of anthraquinones from Rheum emodi: optimisation using RSM, UV and HPLC analysis and antioxidant studies. Ind Crops Prod 52:6574–6582
Azlim AA, Ahmed JKC, Syed ZI, Mustapha SK, Aisyah M, Kamarul RK (2010) Total phenolic content and primary antioxidant activity of methanolic and ethanolic extracts of aromatic plants leaves. Int Food Res J 17(4):1077–1083
Barba FJ, Esteve MJ, Frígola A (2014) Bioactive components from leaf vegetable products. Stud Nat Prod Chem 41:321–346
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant” power: the FRAP assay. Anal Biochem 239:70–76
Chan CH, Yusoff R, Ngoh GC, Kung FWL (2011) Microwave-assisted extractions of active ingredients from plants. J Chromatogr A 1218:6213–6225
Chemat F, Vian MA, Cravotto G (2012) Green extraction of natural products: concept and principles. Int J Mol Sci 12:824–829
Dahmoune F, Spigno G, Moussi K, Remini H, Cherbal A, Madani K (2014) Pistacia lentiscus leaves as a source of phenolic compounds: microwave-assisted extraction optimized and compared with ultrasound-assisted and conventional solvent extraction. Ind Crops Prod 61:31–40
Dani C, Oliboni LS, Bonatto D, Vanderlinde R, Salvador M, Henriques JA (2007) Phenolic content and antioxidant activities of white and purple juices manufactured with organically-produced grapes. Food Chem Toxicol 45:2574–2580
Danisman G, Arslan E, Toklucu AK (2015) Kinetic analysis of anthocyanin degradation and polymeric colour formation in grape juice during heating Czech. J Food Sci 33(2):103–108
Galanakis CM (2012) Recovery of high added-value components from food wastes: conventional, emerging technologies and commercialized applications. Trends Food Sci Technol 26:68–87
Garofulic IE, Dragović-Uzelac V, Jambrak AR, Jukic M (2013) The effect of microwave assisted extraction on the isolation of anthocyanins and phenolic acids from sour cherry Marasca (Prunus cerasus var. marasca). J Food Eng 117:437–442
Hong N, Yaylayan VA, Raghavan GSV, Paré JRJ, Belanger JMR (2001) Microwave assisted extraction of phenolic compounds from grape seed. Nat Prod Res 15:197–204
Karabegović IT, Stojičević SS, Veličković DT, Todorović ZB, Nikolić NC, Lazić ML (2014) The effect of different extraction techniques on the composition and antioxidant activity of cherry laurel (Prunus laurocerasus) leaf and fruit extracts. Ind Crops Prod 54:142–148
Khandare V, Walia S, Singh M, Kaur C (2011) Black carrot (Daucus carota ssp. sativus) juice: processing effects on antioxidant composition and color. Food Bioprod Process 89:482–486
Kumar M, Dahuja A, Sachdev A, Kaur C, Varghese E, Saha S, Sairam KVSS (2018a) Black Carrot (Daucus carota ssp.) and black soybean (Glycine max (L.) Merr.) anthocyanin extract: a remedy to enhance stability and functionality of fruit juices by copigmentation. Waste Biomass Valorization. https://doi.org/10.1007/s12649-018-0450-3
Kumar M, Dahuja A, Sachdev A, Kaur C, Varghese E, Saha S, Sairam KVSS (2018b) Valorization of Black carrot marc: antioxidant properties and enzyme assisted extraction of flavonoids. Res J Biotechnol 13(11):12–21
Lachman J, Hamouz K, Šulc M, Orsák M, Dvořák P (2008) Differences in phenolic content and antioxidant activity in yellow and purple-fleshed potatoes grown in the Czech Republic. Plant Soil Environ 54:1–6
Li H, Deng Z, Wu T, Liu R, Loewen S, Tsao R (2012) Microwave-assisted extraction of phenolics with maximal antioxidant activities in tomatoes. Food Chem 130:928–936
Maran J, Manikandan S, Vigna Nivetha C, Dinesh R (2013) Ultrasound assisted extraction of bioactive compounds from Nephelium lappaceum L. fruit peel using central composite face centered response surface design. Arab J Chem 18:235–239
Meyed (Fruit Juice Industry Corporation) (2011) Turkey fruit juice like products industry report. http://www.meyed.org.tr. Accessed 10 Mar 2016
Mun’im A, Nurpriantia S, Setyaningsih R, Syahdi RR (2017) Optimization of microwave-assisted extraction of active compounds, antioxidant activity and angiotensin converting enzyme (ACE) inhibitory activity from Peperomia pellucida (L.) Kunth. J Young Pharm 9(1):73–78
Myers RH, Montgomery DC (2002) Response surface methodology: process and product optimization using designed experiments, 3rd edn. Wiley, New York
Nayak B, Dahmoune F, Moussi K, Remini H, Dairi S, Aoun O, Khodir M (2015) Comparison of microwave, ultrasound and accelerated-assisted solvent extraction for recovery of polyphenols from Citrus sinensis peels. Food Chem 187:507–516
Nour V, Trandafir I, Cosmulescu S (2014) Antioxidant capacity, phenolic compounds and minerals content of blackcurrant (Ribes nigrum L.) leaves as influenced by harvesting date and extraction method. Ind Crops Prod 53:133–139
Rumbaoa RGO, Cornago DF, Geronimo IM (2009) Phenolic content and antioxidant capacity of Philippine potato (Solanum tuberosum) tubers. J Food Compos Anal 22:546–550
Sala FJ, Burgos J, Condon S, Lopez P, Raso J (1995) Effect of heat and ultrasound on microorganisms and enzymes. In: Gould GW (ed) New methods of food preservation. Blackie Academic and Professional, London, pp 176–204
Setyaningsih W, Saputro IE, Palma M, Barroso CG (2015) Optimisation and validation of the microwave-assisted extraction of phenolic compounds from rice grains. Food Chem 169:141–149
Singh JP, Kaur A, Shevkani K, Singh N (2016a) Composition, bioactive compounds and antioxidant activity of common Indian fruits and vegetables. J Food Sci Technol. https://doi.org/10.1007/s13197-016-2412-8
Singh JP, Kaur A, Singh N, Nim L, Shevkani K, Kaur H, Arora DS (2016b) In vitro antioxidant and antimicrobial properties of jambolan (Syzygium cumini) fruit polyphenols. LWT Food Sci Technol 65:1025–1030
Singleton VL, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteau reagent. Methods Enzymol 299:152–178
Sochar J, Ryvolova M, Krystofova O, Salas P, Hubalek J, Adam V (2010) Fully automated spectrometric protocols for determination of antioxidant activity: advantages and disadvantages. Molecules 15(12):8618–8640
Song J, Li D, Liu C, Zhang Y (2011) Optimized microwave-assisted extraction of total phenolics (TP) from Ipomoea batatas leaves and its antioxidant activity. Innov Food Sci Emerg 12:282–287
Sun Y, Liao X, Wang Z, Hu X, Chen F (2007) Optimization of microwave-assisted extraction of anthocyanins in red raspberries and identification of anthocyanin of extracts using high-performance liquid chromatography–mass spectrometry. Eur Food Res Technol 225:511–523
Sun Y, Liu J, Kennedy JF (2010) Extraction optimization of antioxidant polysaccharides from the fruiting bodies of Chroogom phisrutilus (Schaeff. Fr.) O.K. Miller by Box–Behnken statistical design. Carbohydr Polym 82:209–214
Sutivisedsak N, Cheng HN, Willett JL, Lesch WC, Tangsrud RR, Biswas A (2010) Microwave-assisted extraction of phenolics from bean (Phaseolus vulgaris L.). Food Res Int 43:516–519
Tameshia SB, Parameswarakumar M, Kequan Z, Sean O (2010) Microwave-assisted extraction of phenolic antioxidant compounds from peanut skins. Food Chem 120:1185–1192
Tanongkankit Y, Sablani SS, Chiewchan N, Devahastin S (2013) Microwave-assisted extraction of sulforaphane from white cabbages: effects of extraction condition, solvent and sample pretreatment. J Food Eng 117:151–157
Tchabo W, Ma Y, Engmann FN, Zhang H (2015) Ultrasound-assisted enzymatic extraction (UAEE) of phytochemical compounds from mulberry (Morus nigra) must and optimization study using response surface methodology. Ind Crops Prod 63:214–225
Wang WD, Xu SY (2007) Degradation kinetics of anthocyanins in blackberry juice and concentrate. J Food Eng 82:271–275
Wrolstad RE, Durst RW, Lee J (2005) Tracking color and pigment changes in anthocyanin products. Trends Food Sci Technol 16:423–428
Wu T, Yan J, Liu R, Marcone MF, Aisa HA, Tsao R (2012) Optimization of microwave-assisted extraction of phenolics from potato and its downstream waste using orthogonal array design. Food Chem 133:1292–1298
Xiao W, Han L, Shi B (2008) Microwave-assisted extraction of flavonoids from Radix astragali. Sep Purif Technol 62:614–618
Xu OLJ, Hou H, Hu J, Liu B (2018) Optimized microwave extraction, characterization and antioxidant capacity of biological polysaccharides from Eucommia ulmoides. Sci Rep 8:6561
Yang Z, Zhai W (2010) Optimization of microwave-assisted extraction of anthocyanins from purple corn (Zea mays L.) cob and identification with HPLC–MS. Innov Food Sci Emerg Technol 11:470–476
Yin GH, Dang YL (2008) Optimization of extraction technology of the Lycium barbarum polysaccharides by Box–Behnken statistical design. Carbohydr Polym 74:603–610
Zhang B, Yang R, Liu C (2008) Microwave-assisted extraction of chlorogenic acid from flower buds of Lonicera japonica Thunb. Sep Purif Technol 62:480–483
Acknowledgements
The facilities and financial support provided by the ICAR-Indian Agricultural Research Institute (IARI), Science and Engineering Research Board (SERB), Department of Science and Technology (DST), Confederation of Indian Industry (CII), Government of India and Prathista Industries Limited under Prime Minister Fellowship Scheme to undertake the research work are highly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumar, M., Dahuja, A., Sachdev, A. et al. Valorisation of black carrot pomace: microwave assisted extraction of bioactive phytoceuticals and antioxidant activity using Box–Behnken design. J Food Sci Technol 56, 995–1007 (2019). https://doi.org/10.1007/s13197-018-03566-9
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-018-03566-9