Abstract
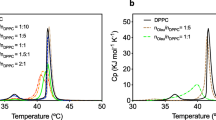
Liposomes were developed with bioactive constituents (omega-3, omega-6, tocopherol) incorporated in acid food. They were made of soy phosphatidylcholine (SPC) allowing the encapsulation of antioxidant vitamin C (VC) and tocopherol. Stearic acid (SA) or calcium stearate (CaS) was added as a bilayer stabilizer. The structural and oxidative stability of the liposomes were studied considering the heat effect of pasteurization. Size was analyzed by light scattering; shape and structure were studied by optical and transmission electron microscopy, respectively. Membrane packing was studied with merocyanine 540. Surface charge and oxidative stability were analyzed by zeta potential and ORAC method, respectively. The liposomes showed significant stability in all of the parameters mentioned above and an important protective effect over thermolabile VC. To confirm their applicability in food, the rheological behavior and a sensory evaluation of liposomes with vitamin C and bioactive constituents were studied. The sensory evaluation of liposomes in orange juice was performed by the overall acceptability and triangular tests with 40 and 78 potential consumers, respectively. The incorporation of all liposomal formulation did not change the acceptability of orange juice. Noteworthy, SPC and SPC:SA systems had rheological behavior similar to a Newtonian fluid whereas that SPC:CaS presented a pseudoplastic one, both considered excellent for larger scale production. From all the obtained results, we can conclude that these liposomal formulations are suitable for food industry applications, incorporating bioactive constituents and generating functional orange juice that conserves its bioactivity after pasteurization.





Similar content being viewed by others
References
Abioye AO, Abioye VF, Ade-Omowaye BIO, Adedeji AA (2013) Kinetic modeling of ascorbic acid loss in baobab drink at pasteurization and storage temperatures. J Environ Sci Toxicol Food Technol 7(2):17–23. doi:10.9790/2402-0721723
Anzaldúa-Morales A (1994) La evaluación sensorial de los alimentos en la teoría y la práctica. Acribia, Zaragoza
Atala E, Vásquez L, Speisky H, Lissi E, López-Alarcón C (2009) Ascorbic acid contribution to ORAC values in berry extracts: an evaluation by the ORAC-pyrogallol red methodology. Food Chem 113:331–335. doi:10.1016/j.foodchem.2008.07.063
Atkinson J, Epand RF, Epand RM (2008) Tocopherols and tocotrienols in membranes: a critical review. Free Radic Biol Med 44(5):739–764. doi:10.1016/j.freeradbiomed.2007.11.010
Bangham AD (1972) Model membranes. Chem Phys Lipids 4:386–392. doi:10.1016/0009-3084(72)90069-2
Bernik DL, Disalvo EA (1993) Gel state surface properties of phosphatidylcholine liposomes as measured with merocyanine 540. Biochim Biophys Acta 1146:169–177. doi:10.1016/0005-2736(93)90352-Z
Biesalski HK, Dragsted LO, Elmadfa I, Grossklaus R, Müller M, Schrenk D, Walter P, Weber P (2009) Bioactive compounds: definition and assessment of activity. Nutrition 25:1202–1205. doi:10.1016/j.nut.2009.04.023
Chen C, Zhu S, Huang T, Wang S, Yan X (2013) Analytical techniques for single-liposome characterization. Anal Methods 5(9):2150–2157. doi:10.1039/C3AY40219C
Disalvo EA, Arroyo J, Bernik DL (2003) Surface properties of liposomes as measured with fluorescent methods. In: Duzgunes N (ed) Methods in enzymology: vol-367, part-A: liposomes, 1st edn. Academic Press, New York, pp. 213–233
Fukuzawa K (2008) Dynamics of lipid peroxidation and antioxidion of alpha-tocopherol in membranes. J Nutr Sci Vitaminol (Tokyo) 54(4):273–285. doi:10.3177/jnsv.54.273
Gramlich G, Zhang J, Nau WN (2002) Increased antioxidant reactivity of vitamin C at low pH in model Membranes. J Am Chem Soc 124(38):11252–11253. doi:10.1021/ja026927b
Hincha DK (2003) Effects of calcium-induced aggregation on the physical stability of liposomes containing plant glycolipids. Biochim Biophys Acta 1611:180–186. doi:10.1016/S0005-2736(03)00053-1
Hincha DK (2008) Effects of alpha-tocopherol (vitamin E) on the stability and lipid dynamics of model membranes mimicking the lipid composition of plant chloroplast membranes. FEBS Lett 582:3687–3692. doi:10.1016/j.febslet.2008.10.002
Henriksen I, Våagen SR, Sande SA, Smistad G, Karlsen J (1997) Interactions between liposomes and chitosan II: effect of selected parameters on aggregation and leakage. Int J Pharm 146(2):193–203. doi:10.1016/S0378-5173(96)04801-6
Jenski LJ, Zerouga M, Stillwell W (1995) Omega-3 fatty acid-containing liposomes in cancer therapy. Proc Soc Exp Biol Med 210(3):227–233. doi:10.3181/00379727-210-43943
Keller BC (2001) Liposomes in nutrition. Trends Food Sci Technol 12:25–31. doi:10.1016/S0924-2244(01)00044-9
Kris-Etherton PM, Hecker KD, Bonanome A, Coval SM, Binkoski AE, Hilpert KF, Griel AE, Etherton TD (2002) Bioactive compounds in foods: their role in the prevention of cardiovascular disease and cancer. Am J Med 113(9B):71S–88S. doi:10.1016/S0002-9343(01)00995-0
Lee KW, Lip GYH (2003) The role of omega-3 fatty acids in the secondary prevention of cardiovascular disease. QJM 96:465–480. doi:10.1093/qjmed/hcg092
Lelkes PI, Miller IR (1980) Perturbations of membrane structure by optical probes. I. Location and structural sensitivity of merocyanine 540 bound to phospholipid membranes. J Membr Biol 52:1–15. doi:10.1007/BF01869001
López LB, Suárez MM (2003) Fundamentos de Nutrición normal. El Ateneo, Buenos Aires
Marsanasco, M., Márquez, A. L., Wagner, J. R., Alonso SdelV., Chiaramoni, N. S. (2011). Liposomes as vehicles for vitamins E and C: an alternative to fortify orange juice and offer vitamin C protection after heat treatment. Food Res Int 44: 3039–3046. doi:10.1016/j.foodres.2011.07.025.
Mateašik A, Šikurová L, Chorvát Jr D (2002) Interaction of merocyanine 540 with charged membranes. Bioelectrochemistry 55:173–175. doi:10.1016/S1567-5394(01)00140-2
McClements DJ (1999) Food emulsions. Principles, practice and techniques. CRC Press, Boca Raton
Meilgaard M, Civille GV, Carr TB (1999) Sensory evaluation techniques. CRC Press, Florida
Monroig O, Navarro JC, Amat F, González P, Hontoria F (2007) Oxidative stability and changes in the particle size of liposomes used in the Artemia enrichment. Aquaculture 266:200–210. doi:10.1016/j.aquaculture.2006.12.016
Mosca M, Ceglie A, Ambrosone L (2011) Effect of membrane composition on lipid oxidation in liposomes. Chem Phys Lipids 164:158–165. doi:10.1016/j.chemphyslip.2010.12.006
Nacka F, Cansell M, Gouygou JP, Gerbeaud C, Méléard P, Entressangles B (2001) Physical and chemical stability of marine lipid-based liposomes under acid conditions. Colloids Surf B Biointerfaces 20:257–266. doi:10.1016/S0927-7765(00)00205-8
Niva M (2007) ‘All foods affect health’: understandings of functional foods and healthy eating among health-oriented Finns. Appetite 48:384–393. doi:10.1016/j.appet.2006.10.006
Primo Yúfera E (1998) Química de los Alimentos. Síntesis, Madrid
Prior RL, Hoang H, Gu L, Wu X, Bacchiocca M, Howard L, Hampsch-Woodill M, Huang D, Ou B, Jacob R (2003) Assays for hydrophilic and lipophilic antioxidant capacity (oxygen radical absorbance capacity (ORAC(FL)) of plasma and other biological and food samples. J Agric Food Chem 51(11):3273–3279. doi:10.1021/jf0262256
Santa Cruz MJ, Martínez C, Varela P (2005) Principios básicos del análisis sensorial. In: Hough G, Fiszman S (eds) Estimación de la vida útil sensorial de los alimentos, 1st edn. Martín Impresores S. L., Madrid, pp. 24–26
Sharma SK, Mulvaney SJ, Rizvi SSH (2003) Ingeniería de los alimentos. Operaciones prácticas y de laboratorio. Limusa S. A., Distrito Federal
Soto-Arriaza MA, Sotomayor CP, Lissi EA (2008) Relationship between lipid peroxidation and rigidity in L-α-phosphatidylcholine-DPPC vesicles. J Colloid Interface Sci 323:70–74. doi:10.1016/j.jcis.2008.04.034
Sukhija PS, Palmquist DL (1990) Dissociation of calcium soaps of long-chain fatty acids in fumen fluid. J Dairy Sci 73:1784–1787. doi:10.3168/jds.S0022-0302(90)78858-3
Yeap PK, Lim KO, Chong CS, Teng TT (2008) Effect of calcium ions on the density of lecithin and its effective molecular volume in lecithin–water dispersions. Chem Phys Lipids 151:1–9. doi:10.1016/j.chemphyslip.2007.09.001
Yoshioka H (1991) Surface modification of haemoglobin-containing liposomes with polyethylene glycol prevents liposome aggregation in blood plasma. Biomaterials 12:861–864. doi:10.1016/0142-9612(91)90075-L
Acknowledgments
This work was funded by the following sources:
Universidad Nacional de Quilmes-Project PUNQ974/11.
Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) – Project PIP-CONICET # 11220110100214.
Ministerio Nacional de Ciencia y Tecnología (MINCyT)- Project: MINCyT-CAPES N°. BR/11/01 (2012–2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
Highlights
Highlight 1 Liposomes with omega-3, omega-6 and vitamins E and C were obtained.
Highlight 2 Liposomes were stable in structure, morphology, size, oxidation and membrane packing.
Highlight 3 High stability of liposomes was maintained before and after pasteurization.
Highlight 4 Liposomes presented a protective effect over the thermolabile vitamin C.
Highlight 5 Liposomes showed an excellent acceptability of commercial orange juice.
Rights and permissions
About this article
Cite this article
Marsanasco, M., Piotrkowski, B., Calabró, V. et al. Bioactive constituents in liposomes incorporated in orange juice as new functional food: thermal stability, rheological and organoleptic properties. J Food Sci Technol 52, 7828–7838 (2015). https://doi.org/10.1007/s13197-015-1924-y
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-015-1924-y