Abstract
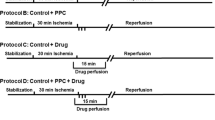
We and others have demonstrated a protective effect of pacing postconditioning (PPC) against ischemia/reperfusion (I/R) injury. However, the mechanisms underlying this protection are not completely clear. In the present study, we evaluated the effects of calcium release from the sarcoplasmic reticulum (SR) and the novel intracellular acidic stores (AS). Isolated rat hearts (n = 6 per group) were subjected to coronary occlusion followed by reperfusion using a modified Langendorff system. Cardiac hemodynamics and contractility were assessed using a data acquisition program, and cardiac injury was evaluated by creatine kinase (CK) and lactate dehydrogenase (LDH) levels. Hearts were subjected to 30 min of regional ischemia, produced by ligation of the left anterior descending (LAD) coronary artery, followed by 30 min of reperfusion. The hearts were also subjected to PPC (3 cycles of 30 s of left ventricle (LV) pacing alternated with 30 s of right atrium (RA) pacing) and/or were treated during reperfusion with agonists or antagonists of release of calcium from SR or AS. PPC significantly (P < 0.05) normalized LV, contractility, and coronary vascular dynamics and significantly (P < 0.001) decreased heart enzyme levels compared to the control treatments. The blockade of SR calcium release resulted in a significant (P < 0.01) recovery in LV function and contractility and a significant reduction in CK and LDH levels (P < 0.01) when applied alone or in combination with PPC. Interestingly, the release of calcium from AS alone or in combination with PPC significantly improved LV function and contractility (P < 0.05) and significantly decreased the CK and LDH levels (P < 0.01) compared to the control treatments. An additive effect was produced when agonism of calcium release from AS or blockade of calcium release from the SR was combined with PPC. Calcium release from AS and blockade of calcium release from the SR protect the heart against I/R. Combining calcium release from acidic stores or blockade of calcium release from the SR with PPC produced a synergistic protective effect.





Similar content being viewed by others
References
Babiker FA, Joseph S, Juggi J (2014) The protective effects of 17beta-estradiol against ischemia-reperfusion injury and its effect on pacing postconditioning protection to the heart. J Physiol Biochem 70:151–162
Babiker FA, Lorenzen-Schmidt I, Mokelke E et al (2010) Long-term protection and mechanism of pacing-induced postconditioning in the heart. Basic Res Cardiol 105:523–533
Babiker FA, van Golde J, Vanagt WY, Prinzen FW (2012) Pacing postconditioning: impact of pacing algorithm, gender, and diabetes on its myocardial protective effects. J Cardiovasc Transl Res 5:727–734
Baumgartner HK, Gerasimenko JV, Thorne C et al (2009) Calcium elevation in mitochondria is the main Ca2+ requirement for mitochondrial permeability transition pore (mPTP) opening. J Biol Chem 284:20796–20803
Billington RA, Bellomo EA, Floriddia EM, Erriquez J, Distasi C, Genazzani AA (2009) A transport mechanism for NAADP in a rat basophilic cell line. FASEB J 20:521–523
Churchill GC, Okada Y, Thomas JM, Genazzani AA, Patel S, Galione A (2002) NAADP mobilizes Ca(2+) from reserve granules, lysosome-related organelles, in sea urchin eggs. Cell 111:703–708
Danial NN, Korsmeyer SJ (2004) Cell death: critical control points. Cell 116:205–219
Djerada Z, Peyret H, Dukic S, Millart H (2013) Extracellular NAADP affords cardioprotection against ischemia and reperfusion injury and involves the P2Y11-like receptor. Biochem Biophys Res Commun 434:428–433
Dong S, Teng Z, Lu FH et al (2010) Post-conditioning protects cardiomyocytes from apoptosis via PKC(epsilon)-interacting with calcium-sensing receptors to inhibit endo(sarco)plasmic reticulum-mitochondria crosstalk. Mol Cell Biochem 341:195–206
Drenger B, Ostrovsky IA, Barak M, Nechemia-Arbely Y, Ziv E, Axelrod JH (2011) Diabetes blockade of sevoflurane postconditioning is not restored by insulin in the rat heart: phosphorylated signal transducer and activator of transcription 3- and phosphatidylinositol 3-kinase-mediated inhibition. Anesthesiology 114:1364–1372
du Toit EF, Opie LH (1994) Antiarrhythmic properties of specific inhibitors of sarcoplasmic reticulum calcium ATPase in the isolated perfused rat heart after coronary artery ligation. J Am Coll Cardiol 23:1505–1510
Ferdinandy P, Schulz R, Baxter GF (2007) Interaction of cardiovascular risk factors with myocardial ischemia/reperfusion injury, preconditioning, and postconditioning. Pharmacol Rev 59:418–458
Ferrera R, Benhabbouche S, Bopassa JC, Li B, Ovize M (2009) One hour reperfusion is enough to assess function and infarct size with TTC staining in Langendorff rat model. Cardiovasc Drugs Ther 23:327–331, Sponsored by the International Society of Cardiovascular Pharmacotherapy
Franco L, Zocchi E, Usai C, Guida L, Bruzzone S, Costa A, De Flora A (2001) Paracrine roles of NAD+ and cyclic ADP-ribose in increasing intracellular calcium and enhancing cell proliferation of 3T3 fibroblasts. J Biol Chem 276:21642–21648
Freixa X, Bellera N, Ortiz-Perez JT et al (2012) Ischaemic postconditioning revisited: lack of effects on infarct size following primary percutaneous coronary intervention. Eur Heart J 33:103–112
Galione A (2006) NAADP, a new intracellular messenger that mobilizes Ca2+ from acidic stores. Biochem Soc Trans 34:922–926
Galione A, Patel S, Churchill GC (2000) NAADP-induced calcium release in sea urchin eggs. Biol Cell 92:197–204
Gan R, Hu G, Zhao Y et al (2012) Post-conditioning protecting rat cardiomyocytes from apoptosis via attenuating calcium-sensing receptor-induced endo(sarco)plasmic reticulum stress. Mol Cell Biochem 361:123–134
Genazzani AA, Billington RA (2002) NAADP: an atypical Ca2+-release messenger? Trends Pharmacol Sci 23:165–167
Hahn JY, Song YB, Kim EK et al (2013) Ischemic postconditioning during primary percutaneous coronary intervention: the effects of postconditioning on myocardial reperfusion in patients with ST-segment elevation myocardial infarction (POST) randomized trial. Circulation 128:1889–1896
Hausenloy DJ, Yellon DM (2009) Preconditioning and postconditioning: underlying mechanisms and clinical application. Atherosclerosis 204:334–341
Heidemann AC, Schipke CG, Kettenmann H (2005) Extracellular application of nicotinic acid adenine dinucleotide phosphate induces Ca2+ signaling in astrocytes in situ. J Biol Chem 280:35630–35640
Limalanathan S, Andersen GO, Klow NE, Abdelnoor M, Hoffmann P, Eritsland J (2014) Effect of ischemic postconditioning on infarct size in patients with ST-elevation myocardial infarction treated by primary PCI results of the POSTEMI (POstconditioning in ST-Elevation Myocardial Infarction) randomized trial. J Am Heart Assoc 3:e000679
Nichols M, Townsend N, Scarborough P, Rayner M (2014) Cardiovascular disease in Europe 2014: epidemiological update. Eur Heart J 35:2929
Osada M, Netticadan T, Tamura K, Dhalla NS (1998) Modification of ischemia-reperfusion-induced changes in cardiac sarcoplasmic reticulum by preconditioning. Am J Physiol 274:H2025–H2034
Patel S (2004) NAADP-induced Ca2+ release—a new signalling pathway. Biol Cell 96:19–28, Under the auspices of the European Cell Biology Organization
Piper HM, Abdallah Y, Schafer C (2004) The first minutes of reperfusion: a window of opportunity for cardioprotection. Cardiovasc Res 61:365–371
Piper HM, Garcia-Dorado D, Ovize M (1998) A fresh look at reperfusion injury. Cardiovasc Res 38:291–300
Piper HM, Kasseckert S, Abdallah Y (2006) The sarcoplasmic reticulum as the primary target of reperfusion protection. Cardiovasc Res 70:170–173
Pipicz M, Varga ZV, Kupai K, Gaspar R, Kocsis GF, Csonka C, Csont T (2015) Rapid ventricular pacing-induced postconditioning attenuates reperfusion injury: effects on peroxynitrite, RISK and SAFE pathways. Br J Pharmacol 172:3472–3483
Smani T, Calderon-Sanchez E, Gomez-Hurtado N et al (2010) Mechanisms underlying the activation of L-type calcium channels by urocortin in rat ventricular myocytes. Cardiovasc Res 87:459–466
Szydlowska K, Tymianski M (2010) Calcium, ischemia and excitotoxicity. Cell Calcium 47:122–129
Tsang A, Hausenloy DJ, Mocanu MM, Yellon DM (2004) Postconditioning: a form of “modified reperfusion” protects the myocardium by activating the phosphatidylinositol 3-kinase-Akt pathway. Circ Res 95:230–232
Vanagt WY, Cornelussen RN, Poulina QP et al (2006) Pacing-induced dys-synchrony preconditions rabbit myocardium against ischemia/reperfusion injury. Circulation 114:I264–I269
Xie GH, Rah SY, Yi KS, Han MK, Chae SW, Im MJ, Kim UH (2003) Increase of intracellular Ca2+ during ischemia/reperfusion injury of heart is mediated by cyclic ADP-ribose. Biochem Biophys Res Commun 307:713–718
Yamasaki M, Churchill GC, Galione A (2005) Calcium signalling by nicotinic acid adenine dinucleotide phosphate (NAADP). FEBS J 272:4598–4606
Yetgin T, Magro M, Manintveld OC et al (2014) Impact of multiple balloon inflations during primary percutaneous coronary intervention on infarct size and long-term clinical outcomes in ST-segment elevation myocardial infarction: real-world postconditioning. Basic Res Cardiol 109:403
Yeung HM, Kravtsov GM, Ng KM, Wong TM, Fung ML (2007) Chronic intermittent hypoxia alters Ca2+ handling in rat cardiomyocytes by augmented Na+/Ca2+ exchange and ryanodine receptor activities in ischemia-reperfusion. Am J Physiol Cell Physiol 292:C2046–C2056
Yu G, Zucchi R, Ronca-Testoni S, Ronca G (2000) Protection of ischemic rat heart by dantrolene, an antagonist of the sarcoplasmic reticulum calcium release channel. Basic Res Cardiol 95:137–143
Zhao ZQ, Corvera JS, Halkos ME, Kerendi F, Wang NP, Guyton RA, Vinter-Johansen J (2003) Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol 285:H579–H588
Acknowledgments
This study was supported by College of Graduate Studies and a grant no. YM 08/11 from Kuwait University, Kuwait.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Rights and permissions
About this article
Cite this article
Khalaf, A., Babiker, F. Discrepancy in calcium release from the sarcoplasmic reticulum and intracellular acidic stores for the protection of the heart against ischemia/reperfusion injury. J Physiol Biochem 72, 495–508 (2016). https://doi.org/10.1007/s13105-016-0498-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-016-0498-0