Abstract
Hypoxia, which activates a variety of signaling pathways to enhance tumor cell growth and metabolism, is among the primary features of tumor cells. Hypoxia-inducible factors (HIFs) have a substantial impact on a variety of facets of tumor biology, such as epithelial-mesenchymal transition, metabolic reprogramming, angiogenesis, and improved radiation resistance. HIFs induce hypoxia-adaptive responses in tumor cells. Many academics have presented preclinical and clinical research targeting HIFs in tumor therapy, highlighting the potential applicability of targeted HIFs. In recent years, the discovery of numerous pharmacological drugs targeting the regulatory mechanisms of HIFs has garnered substantial attention. Additionally, HIF inhibitors have attained positive results when used in conjunction with traditional oncology radiation and/or chemotherapy, as well as with the very promising addition of tumor immunotherapy. Immune checkpoint inhibitors (CPIs), which are employed in a range of cancer treatments over the past decades, are essential in tumor immunotherapy. Nevertheless, the use of immunotherapy has been severely hampered by tumor resistance and treatment-related toxicity. According to research, HIF inhibitors paired with CPIs may be game changers for multiple malignancies, decreasing malignant cell plasticity and cancer therapy resistance, among other things, and opening up substantial new pathways for immunotherapy drug development. The structure, activation mechanisms, and pharmacological sites of action of the HIF family are briefly reviewed in this work. This review further explores the interactions between HIF inhibitors and other tumor immunotherapy components and covers the potential clinical use of HIF inhibitors in combination with CPIs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The majority of solid tumors lack adequate oxygenation areas and are hypoxic (pO2 pressure < 8 mmHg) [1]. Low oxygen (O2) supply caused by aberrant vascularization and excessive O2 demand by tumor cells, which show remarkably increased proliferation and aggravated metabolic activity, are the causes of tumor hypoxia [2]. The biological mechanism known as the hypoxic adaptation response, which is necessary for the survival of cells under hypoxia conditions, involves the stimulation of many molecular signaling pathways that enhance erythropoietin synthesis, angiogenesis, and metabolic reprogramming to promote glycolysis [3]. The activity of transcription factors (TFs) known as hypoxia-induced demand of cells stimulates a variety of signaling pathways necessary for the survival of cells, primarily the TF hypoxia-inducible factors (HIFs), which determines the activation of the hypoxic adaptive response [4, 5].
The onset and metastasis of cancer are significantly influenced by tumor-associated hypoxia. HIFs perform an integral function in the adaptation of tumor cells to hypoxia by enhancing the oncogene transcription and negatively regulating the transcription of the suppressor gene [6]. HIFs are important in a variety of fundamental elements of cancer biology such as angiogenesis [7, 8], maintenance of stem cells [9,10,11], reprogramming of energy metabolism [12, 13], signaling of autocrine growth factors [14, 15], epithelial-mesenchymal transition (EMT) [16,17,18], invasion [19], metastasis [20, 21], and resistance to radiotherapy [22] and chemotherapy [23]. Numerous studies and clinical findings have demonstrated that HIFs are potent targets for the therapy of cancer. The development of tumors, vascularization, and metastasis are initially associated with HIF-1a or HIF-2a levels in both experimental animals and human therapeutic trials. Furthermore, HIF activity is increased by the gain-of-function of oncogenes and viral transforming genes as well as the loss-of-function of genes that inhibit cancers, particularly Von Hippel-Lindau (VHL) genes [24]. Additionally, recent advancements in multi-omics techniques (metabolomics, proteomics, transcriptomics, and genomics) and experimental cancer metabolism modeling have provided new information on the molecular mechanisms of HIFs-deficient cancer cells undergoing hypoxia. A growing variety of pharmacologic treatments have been shown to suppress HIF activity and prevent the growth of tumor xenografts via various molecular mechanisms. The development of pharmacological drugs to modify the HIF signaling system has lately sparked considerable attention. In preclinical and clinical contexts, a variety of methods targeting malignant cells caused by hypoxia are currently being studied.
Immune checkpoint inhibitors (CPIs) play an obvious function in immunotherapy. The advantages of combining numerous CPIs have recently provided novel insight into how to resolve ongoing adverse immunological events. The growing body of research pointing to the potential benefits of combining HIF inhibitors with CPIs for enhancing antitumor immune responses and reducing malignant cell plasticity and treatment resistance will be examined in this review.
2 Structure of HIFs
Human tissues have three distinct HIFs, HIF-1, HIF-2, and HIF-3, which are strictly modulated by alterations in oxygen tension [25]. While HIF-3's function is less understood, HIF-1 and HIF-2 are transcriptional modulators with both distinct and overlapped target genes. The expression of HIF-2 and HIF-3 in the human endothelium starts with chronic hypoxia, whereas HIF-1 controls the acute response to hypoxia. Unlike HIF-2α and HIF-3α, which are only expressed in certain tissues, HIF-1α, a 120 kDa oxygen-sensitive subunit ubiquitously, is expressed in all tissues.
HIFs are composed of heterodimers and the subunits α and β. Hypoxia induces the production of the HIF-1/2/3α alpha subunits, which are found in cell membranes. In the nucleus, beta subunits (aryl hydrocarbon receptor nuclear translocator) HIF-1β/aryl hydrocarbon receptor nuclear translocator (ARNT), HIF-2β (ARNT2), and HIF-3β (ARNTL), respectively, are expressed constitutively [26]. The PER-ARNT-SIM(PAS) and basic helix-loop-helix (bHLH) motifs that facilitate heterodimerization, as well as DNA binding, are located at the amino-terminal end of both the α and β subunits [27]. Two transactivation domains (TADs) (i.e., N-TAD and C-TAD) and oxygen-dependent degradation domain (ODD) form the carboxy-terminal motif of HIF-1/2α, which controls both the proteins’ transcriptional activity and stability, respectively, [27]. Additionally, nuclear localization signals C-NLS and N-NLS, correspondingly on the C- and N-termini of the α subunits point them toward the nucleus [28]. The N-TAD domain is the aspect where HIF-1α and HIF-2α differ the most from one another, sharing 48% of their amino acid sequence identity [29]. HIF-3α shares bHLH and PAS motifs with HIF-1/2α although it does not have the C-terminal transactivation motif [30]. Nonetheless, alternative splicing (AS) of HIF-3α and the use of various promoters lead to at least four distinct HIF-3α mRNA variants that encode for ≥ isoforms [31]. The inhibitory PAS motif protein, a shortened protein that blocks HIF-1/2 function in cell culture, is the HIF-3 variant that has received the most research to date [32]. Conversely, it was discovered that the other human HIF-3 variants upregulated gene levels, proving that HIF-3 is also a crucial transcriptional modulator of hypoxia signaling [33, 34]. The HIF subunit domains are shown in Fig. 1.
3 HIF stability modulation
The stability of all three HIF-α proteins is regulated by oxygen [26]. Factor-inhibiting hypoxia-inducible factor-1 (FIH-1), Prolinehydroxylases (PHDs), and hydroxylase enzymes hydroxylate alpha subunits posttranslationally under normoxia. Irrespective of oxygen tension, HIF-1α is often inactivated in healthy cells yet commonly maintained in cancerous cells [35, 36]. A crucial step that triggers the expression of numerous genes implicated in diverse biological processes is the transportation of stable HIF-1α to the nucleus as well as its interaction with HIF-1β ARNT [24]. These hydroxylase enzymes aim to polyubiquitinate and degrade the alpha subunits under normoxia [37]. Specialized proline residues within ODD domains are subjected to hydroxylation reliant on PHD, which recruits the VHL tumour suppressor protein (pVHL) as well as other protein cofactors and causes the 26S proteasome to degrade the alpha subunits. PHDs need iron ions (Fe2+), ascorbic acid, 2-oxoglutarate, and molecular oxygen to hydroxylate HIF-α [38]. Additionally, various PHD isoforms have varied HIF specificities. For example, PHD-2 activity is mostly HIF-1α-specific [39], while PHD-3 controls HIF-2α levels primarily [40]. The heterodimeric complex's transcriptional activity is regulated by the second hydroxylase, FIH-, by hydroxylating one asparagine residue in the transactivation motifs of HIF-1/2 [41]. Such posttranslational modification precludes the dependent mobilization of the co-activators CREB-binding protein (CBP, sometimes referred to as CREBBP) and p300, which exhibit histone acetyltransferase activity, serving as a late-stage key phase in the HIF activation process [42]. Molecular oxygen is also necessary for the FIH-1 action. It's worth noting that FIH-1 selectively hydroxylates HIF-1α and needs less oxygen tension to stay active than PHD-2 [43].
Because the hydroxylases in hypoxia lack an oxygen substrate, HIF-1α accumulates, moves to the nucleus, and afterward forms complexes with HIF-1β, its co-factor. In normoxia, HIF protein content and transcriptional activity are kept low by the action of both PHD-2 and FIH-1 [44]. Low oxygen tension, on the other hand, reduces the activity of PHD-2 and FIH-1 and stabilizes the HIF-alpha subunit. Following translocation to the nucleus, the alpha subunits dimerize with the beta subunits for the purpose of generating HIF complexes that are transcriptionally active [45]. By attaching to the hypoxia response element (HRE) sequences within promoters of their distinct and shared target genes, HIF-1/2 facilitates the endothelium hypoxic response and upregulates those genes [30]. Numerous genes, such as those involved in cell proliferation [46], metastasis [20, 47,48,49], glycolysis [50, 51], pH control [52], and angiogenesis [53] are activated by the HRE, a TF-binding domain found in the promoter sequences of target genes. Despite reports of HIF-elicited negative transcriptional modulation, it nearly completely occurs indirectly [54, 55]. Lastly, under hypoxia, the Sirtuin 1 (SIRT1) induced by hypoxia preferentially deacetylates HIF-1α and enhances HIF-1 activity [56]. The control of HIF subunits in normoxia and hypoxia is depicted in Fig. 2.
HIF- subunits are hydroxylated only when Iron, oxygen, and 2-oxoglutarate are present under normoxia. The pVHL E3 ligase complex can bind to the HIF-subunit via hydroxylation, thus facilitating polyubiquitination and eventual degradation by the 26S proteasome. The PHD is inhibited in hypoxia (< 5% O2). HIF-subunits are shielded from pVHL-driven destruction and move to the nucleus after which they combine with HIF-1 to produce heterodimers. The HIF heterodimers bind to HRE present in the target genes’ DNA modulatory regions, stimulating their transcription by enlisting the transcriptional co-activators CBPP/p300, stimulating the transcription of several HIF target genes involved in tumor cell proliferation/survival, EMT, angiogenesis, metastasis/invasion, ODD metabolic reprogramming, and immunoregulation
4 Specificity of HIF isoforms in the tumor immune microenvironment
A critical factor in deciding whether a tumor will progress or shrink during the course of its development and how it responds to therapy is the complex landscape known as the tumor microenvironment (TME), which is made up of immune and stromal cells. Research demonstrates the TME cells’ extraordinary heterogeneity, flexibility, and interconnectedness [57]. It was demonstrated that IL-4, IL-13, and CXCL1 suppression reduced protumorigenic myeloid-derived suppressor cells (MDSCs) and tumor-associated macrophages (TAMs) and that elevated levels of CXCL9 and CXCL10 resulted in greater infiltration of antitumor NK and CTLs cells [58]. Hypoxic stress is a key microenvironmental component that can inhibit antitumor immunity and activates many pathways that lead to the formation of resistant cancerous cells [59,60,61].
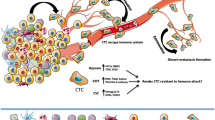
The TME is governed by specific microenvironmental factors, which in turn are regulated by cross-cellular communication, which produces diverse signaling outputs in different cell types. In hypoxia, for instance, hypoxia enhances the secretion of CCL28 by tumor cells in a HIF-1α-dependent mode, which enhances the recruitment of CCR10+ T reg cells to the tumor site, thus inhibiting the functions of cytotoxic T cells and accelerating tumor growth [62, 63]. Adenosine and the A2a receptor work together to promote immune checkpoint expression in hypoxic environments, which suppresses T cells [64]. Additionally, HIF-1α increases the expression of a protein called ADAM10 (a disintegrin and metalloproteinase domain-containing protein 10) in cancerous cells, causing major histocompatibility complex class I chain-related molecule (MICA) to be shed from a tumor cell's surface. The NKG2D activator receptor on NK and T cells is downregulated by soluble MICA, allowing cancer cells to evade [65]. Eventually, hepatocellular carcinoma (HCC) cells express ectonucleoside triphosphate diphosphohydrolase 2 (ENTPD2) when hypoxia-driven HIF-1α (but not HIF-2α) is present, which facilitates the onset and progression of syngeneic Hepa1-6 HCC tumors in mice by increasing the infiltration of MDSC into the tumor mass [66]. Overall, the HIF family of TFs controls a variety of TME activities known to modify the metabolic activity and aggressiveness of tumors as well as the environment's immunosuppressive conditions that favor tumor development. Immune checkpoint blockade (ICB) is used to accomplish the latter goal.
Additionally, there is proof that the immunological and non-immune constituents of the TME interact extensively, whereas the efficacy of adoptively transplanted tumor-specific CD8 + T cells in a syngeneic murine model of lung cancer is increased by the overexpression of superoxide dismutase (SOD3) in endothelial cells (EC) and the consequent stabilization of HIF-2α rather than HIF-1α [67]. Previous research illustrated that antisense HIF-1 and B7-1-T may enhance NK cell and CD8 T cell-elicited anticancer immune response and trigger tumor rejection by downregulating HIF-1 expression [68]. The effect of HIF transcription factors on tumor-immune cell interactions in TME is shown in Fig. 3. According to Lequeux et al., therapies that prevent HIF-1/HIF-1 dimerization can change the tumor's immunosuppressive environment into one that is permissive to NK and CD8+ effector T cell infiltration. These techniques may be utilized to enhance cancer immunotherapy regimens including ICB treatments and cancer vaccination in melanoma individuals who are not responding to treatment [69]. Furthermore, HIF-1 suppression and DC-based immunotherapy were confirmed to enhance survival in a breast cancer model by enhancing the proliferation and activities of cytotoxic T cells and enhancing the synthesis of type 1 interferon (IFN) [70]. Collectively, the above evidence highlights the complex and dynamic interactions between HIF-activated tumor cells and TME immune cells that affect tumor advancement, treatment response, and aggressiveness.
The production of tumor cell-specific HIF in tumor cells affects the interactions between tumor and immune cells, which control cytotoxicity and therapeutic effects induced by immune cells against tumors.
5 HIF associated with immune checkpoint blockade
Hypoxia regulates multiple major immune checkpoint molecules, such as the V-domain Ig suppressor of T-cell activation (VISTA), the cluster of differentiation 47 (CD47), human leukocyte antigen G (HLA-G), and PD-L1. The interactions between HIF and these key immune checkpoints will be highlighted next (Fig. 4).
5.1 PD-L1
By regulating the expression of the immune checkpoint PD-L1 on the MDSCs' surface, hypoxia increases the immunosuppressive characteristics of MDSCs against T cells [71,72,73]. Inhibiting PD-L1 under hypoxic settings encourages T cell activation driven by MDSC while attenuating IL-6 and IL-10 synthesis from MDSCs. [72]. It has been shown that hypoxia can increase the levels of PD-L1 protein in human breast and prostate carcinoma cells via HIF-1α [73]. MDSCs derived from the spleen are greatly promoted to express PD-L1 when exposed to hypoxia, according to data from mice models that bear the B16-F10 melanoma [72]. Additional research revealed that after stabilizing in hypoxic cells, HIF-1α attaches to the HRE in the PD-L1 gene’s proximal promoter [72]. Additionally, HIF-2α is also implicated in the overexpression of PD-L1 which is remarkably correlated with VHL mutation and HIF-2α stability among patients with clear cell renal cell carcinoma (ccRCC) [71]. Analysis of paraganglioma and pheochromocytoma samples revealed a substantial correlation of PD-L2 expression with carbonic anhydrase 9 (CAIX) and hypoxia-driven HIF-1α, which was not the case with PD-L1 expression [74]. These findings suggest that inhibiting HIF-1α and PD-L1/PD-L2 simultaneously might be an effective way to increase the activity of cytotoxic T cells.
5.2 HLA-G
The immune checkpoint marker HLA-G, a non-classical MHC-I molecule, is thought to be relevant to immunotherapy [75]. There is evidence that high invasive or metastasis and negative treatment outcomes are correlated with malignant tumors' aberrant HLA-G expression [76]. Published investigations have shown that hypoxia causes HLA-G upregulation at the mRNA level [77]. In the first part of their investigation, Mouillot et al. showed that when exposed to hypoxia, HIF-1α increases the levels of HLA-G mRNA in the HLA-G–negative M8 melanoma cell lines [77]. Since the HLA-G promoter contains a small number of HREs, HIF-1 probably binds to HRE domains to activate HLA-G transcription as the mechanism driving HLA-G expression in response to hypoxia [76]. According to research by Yaghi et al., the HLA-G gene is expressed in glioma cells due to the attachment of HIF-1 to the HRE domain in exon 2; other MHC-I molecules affected by hypoxia in malignancies have received less research [78]. Furthermore, research shows that the expression of Qa-1 in mice and HLA-E in human malignant cells may both be remarkably upregulated by the combined exposure to glucose deprivation and oxygen. As a result, the cells can engage with the blocking CD94/NKG2 receptor on activated T cells and avoid being recognized by CD8+ T cells [79].
5.3 CD47
The transmembrane immune checkpoint protein CD47 also referred to as integrin-associated protein, is present on the surface of both solid and hematologic tumor cells. Also, CD47 upregulation is associated with an unfavorable clinical prognosis [80]. The predominant process underlying CD47-elicited immune escape is its interplay with SIRP, which is expressed at a high level in myeloid-lineage hematopoietic cells such as MDSCs and TAMs. Through this connection, cancer cells are prevented from being phagocytosed by a strong “don’t eat me signal” delivered by SIRP phosphorylation [80]. Recent research has shown that by specifically binding to its promoter, HIF-1α may modulate the CD47 gene’s transcription. Additionally, the capacity of macrophages to phagocytose breast carcinoma cells is improved when CD47 is inhibited [81]. MDSCs and macrophages are blocked from expressing prophagocytic signaling in pancreatic adenocarcinoma due to CD47 overexpression under hypoxia [82]. Moreover, both innate and adaptive immunity are negatively impacted by the CD47-SIRP axis. The CD47-SIRP axis has proven to be a valuable immunotherapy target in cancer, and the use of anti-CD47 antibodies in various solid tumors is now being studied [80].
5.4 VISTA
An investigation conducted recently showed that the hypoxic TME of colorectal cancer (CRC) mouse models and humans overexpresses VISTA, a B7 family negative checkpoint modulator [83]. When HIF-1α attaches to the HRE in the VISTA promoter, myeloid cells, such as DCs, macrophages, and MDSCs, selectively express VISTA in the hypoxic TME [84]. VISTA expression brought on by hypoxia can inhibit T cell function and proliferation [83, 85]. Additionally, hypoxic lymphocytes have elevated levels of co-stimulatory and co-inhibitory receptors like OX40, CTLA-4, CD137, and LAG-3 than lymphocytes under normoxia environments [86]. This association between HIF-1 and these higher levels of co-inhibitory and co-stimulatory receptors has been demonstrated.
Drugs that disrupt these pathways are now used to treat a wide range of malignancies and exhibited sustained therapeutic effects in some cancer patients. The next generation of immune checkpoints tends to function in synergy with chemotherapy or other CPIs due to its distinct action mechanism in comparison to previous anticancer methods.
5.5 CD73
CD73 expression in the tumor microenvironment has been investigated as a predictive biomarker for clinical outcomes in a variety of tumor types, with a statistically significant correlation between high CD73 expression and poor clinical results [87]. This is consistent with adenosine’s activity as an immunosuppressive metabolite [88]. The overexpression of CD73/A2aR is frequently attributed to genetic variations [89], which in turn leads to immunosuppression by modulating the tumor microenvironment [90,91,92]. HIF-1α accumulation has been shown to promote downstream CD73 overexpression, activating the CD73-adenosine pathway and reducing T cell effector activity [93]. In the latest study, Yuan et al. created cancer cell membrane-camouflaged gelatin nanoparticles (CSG@B16F10) to distribute oxygen-producing molecules catalase and CD73siRNA simultaneously, improving tumor oxygenation and reducing CD73-adenosine pathway-mediated T cell immunosuppression [94]. An appealing and potential target in cancer immunotherapy is the immunosuppressive effect of hypoxia signaling on NK cells via the HIF-dependent CD73-adenosinergic pathway. In the context of solid tumors, the administration of drugs that can block CD73 and/or target HIFs in addition to NK cell-based therapies is becoming recognized as an immunotherapeutic approach with substantial promise [95].
In hypoxic tumor cells such as MDSCs, macrophages, and DCs HIF-1 stabilization promotes the overexpression of PD-L1. The increased level of CD47 on the tumor cell surface is attributed to HIF-1. Powerful “don’t eat me” signals are sent to cancer cells when CD47 binds to SIRP, which is extensively upregulated on myeloid-linage hematopoietic cells including TAMs and MDSCs thus preventing phagocytosis. The tumor cell surface expresses a great number of HLA-G when exposed to hypoxia. The overexpressed HLA-G attaches to its inhibitory receptors on immune cells, inducing immunosuppression and facilitating immunological evasion by compromising the DC antigen presentation, activation of suppressor T-cells, and inhibition of cytotoxic attack. Hypoxia may induce VISTA overexpression on myeloid cells such as macrophages, MDSCs, and DCs once HIF-1 interfaces with the HRE in the VISTA promoter, which inhibits the activity and proliferation of T cells. The expression of co-stimulatory factors (OX40 and CD137) and inhibitory immune checkpoints (CTLA4 and LAG-3) on the surface of T cells rises in response to hypoxia. A hypoxic environment inhibits the levels of co-stimulatory molecules such as CD86, CD80, and CD40 on DCs, via the mechanism of stabilizing HIF-1.
6 HIF activators and inhibitors
6.1 HIF agonists
Several pharmaceutical therapies that stimulate the HIF pathway are presently identified, particularly hydroxylase inhibitors such as dimethyloxalylglycine (DMOG), a 2-oxoglutarate mimic that stimulates the expression and activity of HIF in vivo and in vitro [96]. Several other similar hydroxylase inhibitors, such as JNJ1935 and FG-4497, have also been identified [97, 98]. Upon treatment with A-503451A, the expression of erythropoietin, vascular endothelial growth factor, and HIF-dependent genes, were increased at the protein and mRNA levels [99]. Potent HIF activators include iron chelators like desferrioxamine and metals like cobalt [100]. Even though the exact action mechanism of the cardiovascular medication hydralazine, which has been employed to treat cardiovascular diseases for many years, is still unknown, it has been demonstrated to have a potent hydroxylase inhibitory effect [101]. As a result, multiple examples of possible HIF-activating drugs have been presented, as shown in Fig. 5.
6.2 HIF inhibitors
The de-repression in hypoxia-based regulation of the HIFs system makes it more challenging to develop HIF inhibitors rationally. Although the particular processes by which many of these medications directly repress HIFs are still largely unknown, a range of potential HIF inhibitors has been discovered through several screening techniques, many of which included evaluating huge libraries of substances against simple assays of HIF activity. Importantly, HIF suppression is a topic of ongoing research in cancer treatment [6, 102]. There have been reports of HIF inhibitors, such as medications that interfere with a variety of processes, like HIF-1α mRNA expression (e.g. aminoflavone [103]), HIF-1α protein synthesis (e.g. rapamycin [104]), HIF-1α protein stabilization (e.g. HSP90 inhibitors [105]), HIF heterodimerization (e.g. acriflavine [8]), HIF-DNA binding (e.g. echinomycin [11]) and HIF transactivation (e.g. bortezomib [106, 107]). Table 1 presents possible action mechanisms of a few HIF inhibitors (not exhaustive).
Recently, efforts have been undertaken to create targeted HIF-2 inhibitors that are presently being tested in clinical trials [108]. Among the many clinical concerns in the context of cancer, chronic hypoxia is one of them, and the prospect of HIF-2-directed therapeutics is now feasible, judging by recent findings that identify a key HIF-2 target gene, namely, tubulin beta-3 chain (TUBB3), which is involved in tumor advancement and chemotherapy [109]. Conversely, diseases characterized by ischemic/hypoxic states and inflammatory disorders might both be treated by stimulating the protective HIF response. The principal targets of the several compounds suggested to trigger HIF signaling are PHD2, PHD3, and VHL. In terms of clinical development, PHD3 inhibitors are the most advanced [110,111,112]. Nevertheless, current research suggests that in addition to regulating the HIF response, FIH1 and PHDs also modulate a variety of other cellular metabolic processes [113], which requires further consideration to reduce any potential negative outcomes.
Multiple HIF inhibitors (yellow boxes) and HIF activators (purple boxes) drugs identified to date. Among them, HIF inhibitors interfere with a variety of processes, such as Hif-α mRNA expression, Hif-α protein synthesis, Hif-α protein stabilization, HIF heterodimerization, HIF-DNA binding, and Hif-α transcriptional activation.
7 Several HIF inhibitors in malignant tumors
A few biological processes that are regulated by hypoxia-dependent HIF-1 and HIF-2 encompass angiogenesis, EMT, proliferation/survival of tumor cells, the cancer stem cells (CSCs) maintenance, metastasis/invasion, metabolic reprogramming, and immunomodulation [145, 146]. Multiple HIF inhibitors are now being researched in pre-clinical and clinical studies since HIF performs a significant function in cancer, and this pathway presents a possible treatment target [147, 148]. Several compounds are now undergoing phase II clinical studies, either alone or in conjunction with available anticancer medications, mostly to treat advanced or resistant malignancies [148]. Several approaches have been put forth to combat hypoxia in cancer, including the use of hypoxia-activated prodrugs (HAPs), suppression of HIF signaling, metabolic intervention, as well as downstream targeting of crucial hypoxic pathways, such as the mTOR and unfolded protein response (UPR) pathways; others are still in the conceptual design phase, such as siRNA-mediated gene treatments and recombinant anaerobic bacteria [149].
Notably, Belzutifan (MK-6482), a specific small molecule inhibitor that targets the TF HIF-2, was shown to shrink tumors and stop tumor growth in over 90% of VHL patients in a 3-year research [150]. Belzutifan recently received FDA approval to treat adult patients with pancreatic neuroendocrine tumors, central nervous system hemangioblastomas, and VHL-associated ccRCC [151]. Belzutifan's effectiveness is now being assessed in multiple other solid tumors, such as glioblastoma (NCT02974738). Furthermore, several clinical trials are currently conducted involving patients with ccRCC evaluating combinatorial treatment approaches including immune-checkpoint therapies or anti-angiogenic therapies used in conjunction with belzutifan, like Belzutifan + Pembrolizumab (PD-1 inhibitor) + Lenvatinib (NCT04736706), Belzutifan + Cabozantinib (TKI) (NCT03634540), and Belzutifan + Lenvatinib (TKI) (NCT04586231) [152,153,154].
The current study discovered that removing stromal hypoxia-inducible factor-2 from animals with pancreatic cancer reduced tumor development and increased median survival, potentially by interfering with immunosuppressive cancer-associated fibroblast-macrophage interaction [155]. PX-478, a HIF-1α inhibitor that could also inhibit HIF-2α, slows the growth of pancreatic ductal adenocarcinoma and esophageal squamous cell cancer both in in vivo and in vitro settings [156, 157]. HIF-1α production and activation are inhibited at various levels by bortezomib because it prevents the recruitment of p300 and hinders the PI3K/Akt/TOR pathway, without affecting HIF-2α [107, 158]. In a similar vein, a range of anti-cancer medications, including those that block the PI-3 K-mTOR pathway, histone deacetylases (HDAC), or topoisomerase, exhibit indirect impacts and varying degrees of effectiveness in reducing the production of and activation of HIF-1/2α [159].
Salman et al. reported the development of 32-134D, a low-molecular-weight compound that suppressed the expression of genes regulated by HIF-1/2 in cancerous cells and prevented tumor growth [160]. The low-molecular-weight drug was developed to specifically target HIF-1/HIF-2, resulting in the degradation of the HIF- component and blocking transcription of HIF-1/2 target genes. This suppression influenced genes involved in glycolysis, angiogenesis, and immune regulation. Furthermore, mice with tumors developed fewer tumors following HIF inhibitor administration.
8 HIF inhibitors combined with conventional chemoradiotherapy
Hypoxia contributes to the failure of conventional cancer therapies such as chemotherapy [161] and radiotherapy [162]. Observations from multiple murine models show that therapies with VEGF receptor inhibitors, such as the small molecule tyrosine kinase inhibitor sunitinib or the anti-VEGFR2 antibody DC-101, decreased the vascularization and growth of primary tumors but accelerated metastasis, likely since deficient angiogenesis contributed to enhanced intratumoral hypoxia as well as increased HIF activity [163,164,165]. The FDA revoked the clearance of the anti-VEGF antibody bevacizumab because it did not affect the advancement of breast cancer [166] which may entail additional angiogenic growth factors being expressed in a HIF-1-dependent way. Conversely, HIF inhibitor remarkably alleviated the spontaneous metastasis of human breast cancer cells to the lungs of mice orthotopic transplant models by interfering with several metastatic process phases [20, 167]. Numerous studies also show that HIFs have a role in radiotherapy [22] and chemotherapy [23] resistance and increasing data suggest that HIF-1 activity could perform a role in the development of resistance to new targeted medicines, including imatinib therapy for chronic myeloid leukemia [168]. Together, these findings imply that the co-administration of HIF inhibitors can increase the effectiveness of antiangiogenic drugs, and mouse model studies corroborate this hypothesis [169]. When conventional chemotherapy is used in conjunction with a HIF inhibitor, it also might be more successful; this effect is underlied by a variety of molecular pathways that depend on the cell type and are specific to chemotherapy.
9 Cross-talk between HIF and tumor immunotherapy
The treatment environment for human tumors in advanced clinical stages has significantly changed over the past several decades, thanks to immunotherapy, a ground-breaking intervention [170]. Adoptive cell transfer (ACT) and CPIs are two examples of immunotherapeutic interventions that use the components of the immune system to combat malignant cells [171]. Unlike conventional treatment approaches, CPIs function by reactivating the host immunity to combat malignant cells. A balanced state is maintained between pro- and anti-inflammatory signals by immune checkpoints [172]. These immune checkpoints encompass a set of stimulating and inhibiting mechanisms responsible for regulating the activity of immune cells [173]. Evidently, hypoxia could be thought of as a possible target in combination with cancer immunotherapy due to HIF’s crucial function in controlling tumor development and immune surveillance. In this part, we investigate the prospect of altering hypoxia to enhance the success of cancer immunotherapies using data from pre-clinical and clinical research.
9.1 HIFs associated with immune checkpoint blockade
The significance of the immune checkpoint in immune tolerance vs. tumor escape of host immune response defines the proportionate risk/benefit ratio [174]. The predominantly applied immunotherapy interventions in the past decade are antibodies against immune inhibition receptors including PD-L, PD-1, and CTLA-4 [175]. The US FDA has licensed three separate classes of CPIs to treat distinct cancers, namely CTLA-4 inhibitors (Ipilimumab), PDL-1 inhibitors (Avelumab, Durvalumab, and Atezolimumab), and PD-1 inhibitors (Cemiplimab, Pembrolizumab, and Nivolumab) [176]. Immune tolerance enhancement by differential PD-L1 regulation in normal and malignant tissues is an essential method for safer and more effective immunotherapy. In some cancer patients, PD-1/PD-L1 inhibitors can produce positive therapeutic outcomes. PD-1/PD-L1 inhibition-based combination therapies are available for a majority of cancer subtypes and may prolong patient survival [177]. Approximately 50% of individuals with cancer are candidates for ICI immune checkpoint inhibitor (ICI) therapy, and a large proportion of patients acquire sustained responses [178, 179]. Nonetheless, only a portion (20–40%) of patients respond well to this treatment, emphasizing the rising demand for the development of predictive biological markers [180]. Preliminary clinical trials of the anti-CTLA-4 antibody ipilimumab showed long-lasting responses and significant survival benefits in certain melanoma patients [181]. Patients who have advanced and otherwise incurable melanoma, NSCLC, and urothelial tumors responded similarly to PD-1 and PD-L1 blockade [182, 183]. Conversely, high-grade immune-related adverse events (irAEs), such as hepatitis, pneumonitis, thyroiditis, dermatitis, and colitis were linked to both CTLA-4 inhibition and PD-1/PD-L1 blocking [184,185,186]. These iAEs were most likely caused by the failure of self-tolerance caused by ICI [187, 188]. Combinatorial therapeutic targeting was made possible thanks to the complementing immunosuppressive properties of CTLA-4 and PD-1. Patients with RCC and melanoma attained an improved response rate after the administration of the ipilimumab + nivolumab (anti-PD-1) combination scheme [189, 190], even though it means experiencing higher-grade irAEs more frequently [186]. In the meantime, the practical usefulness of CPIs is hampered by toxicity associated with the therapy as well as tumor-specific or acquired tolerance to CPIs [191].
It has been demonstrated that hypoxia alters the levels of immune checkpoints like CTLA-4, PD-1/L-1, CD47, and TIM3 to modify the immune cell-induced anti-tumor response, thus suppressing immune surveillance [64]. Moreover, CD137 (4-1BB) is a member of the TNF receptor family that was first discovered on activated T lymphocytes. CD137 expression is greatly preferred in TILs as a result of a hypoxia-dependent HIF-1 response. Low doses of anti-CD137 mAb targeted TILs given within tumors have been proven in studies to have systemic therapeutic benefits and to function synergistically with systemic inhibition of the PD-1/B7-H1 (PD-L1) pathway [86]. HIFs targeting in immunotherapy is a fairly novel idea, with ample evidence of its viability provided by others [72, 73, 192]. Furthermore, a handful of reports have shown that CPIs coupled with various treatment modalities, including chemotherapy [193], radiation treatment, HIF inhibitors, and cancer vaccines can successfully overcome tumor resistance to ICI therapy.
9.2 Combination therapy of CPIs and HIF inhibitors
When the HIF-1α/PD-L1 axis in malignant cells is targeted, it leads to tumor rejection and the reactivation of tumor-infiltrating lymphocytes (TILs). For example, in vivo administration of PX-478 and anti-PD-1 antibodies inhibits tumor development and lengthens animal life, which is linked to decreased suppression of immunity, increased TIL homing, and decreased expression of EMT phenotypes [194]. Inhibiting the transcriptional activities of HIF-1α increases NK cells and CTLs mediated by CCL2- and CCL5 in the tumor bed in a murine melanoma model, thus enhancing the anticancer effects of peptide vaccine and anti-PD-1 blocking antibodies [69]. One study illustrated that HIF-1 suppression promoted PD-L1 overexpression in healthy regions while blocking PD-L1 in the tumor site [195]. According to Zandberg et al., mice with intra-tumoral hypoxia exhibited modified disease control rate (DCR) and survival in an animal model of HNSCC, making them non-responsive to anti-PD-1 rituximab [196]. Importantly, the proportion of mice that responded completely to anti-PD-1 CPI rose from 25 to 67% following treatment with the HIF inhibitor [160].
In preclinical studies, the topoisomerase 1 and HIF-1α dual inhibitor CRLX101 also displayed positive synergy with immunotherapeutic regimens [58]. Combining ICI (anti-CTLA-4/PD-1) with HIF-1-mediated ectoenzyme ENTPD2 inhibitors remarkably increased the levels of TILs and prolonged the lifespan of mice with tumors relative to ICI treatment alone [66]. Notably, hypoxia-triggered HIF pathways comprise a sophisticated network of several intersecting cascades, and the utilization of combo treatment calls for more research. Hence, combining HIF inhibitors and CPIs can be promising for increasing anticancer immune response while decreasing tumor cell plasticity and therapeutic resistance [197]. These results offer what we consider to be a novel approach to the development of immunotherapeutic drugs. HIF suppression could therefore be a useful ally for different CPIs.
9.3 HIF inhibitors decreased irAEs
Since CPIs operate to activate T-cell responses, it is not unexpected that these compounds might result in irAEs, some of which can be severe or perhaps deadly [187, 198]. The possible severity and mortality of ICI-related toxicities are becoming more well-recognized [198, 199]. Fatalities appear to occur randomly and often early following the initiation of therapy [198]. The prevention of autoimmune conditions may be aided by CTLA-4, a repressive factor controlling T-cell immune reactions. Nevertheless, blocking it with ipilimumab could result in irAEs like enterocolitis and colitis [200]. Correspondingly, monoclonal antibodies targeting PD-1 and PD-L1 are not as toxic as those against CTLA-4 [190]. Unfortunately, the present method, which resolves tumor escape from host defense, also impairs the immune tolerance checkpoint, resulting in severe irAEs, especially when combined with anti-CTLA-4 antibodies. Notably, the severity of fatal organ involvement varies across anti-CTLA-4 and anti-PD-1/PD-L1 interventions, with colitis being the most commonly observed in the former and neurotoxicity, hepatitis, and pneumonitis being recorded in the latter. [201,202,203]. Myocarditis and colitis are the most prevalent causes of death resulting from combination therapy (e.g., anti-PD-1/CTLA-4) [198].
According to the most current research data, the HIF-1 inhibitor echinomycin enhanced the anti-CTLA-4 treatment’s cancer immunotherapy effectiveness, with efficacy comparable to anti-CTLA-4 + anti-PD-1 antibodies [195]. Bailey et al. claim that inhibiting HIF-1 raises the immune tolerance checkpoint in healthy cells in addition to preventing immune escape in the TME [195]. Therefore, eliminating irAEs while maintaining the synergistic benefits of dual ICB is a key challenge for cancer immunotherapy.
10 Discussions and perspectives
Identifying small chemical inhibitors that precisely target the HIF pathway has been attempted extensively to date (reviewed in [204]). Nonetheless, investigations of HIF-1 activities in cultured tumor cell lines are the basis for the majority of HIF inhibitors that have been found thus far. None of the inhibitors that are currently on the market appear to have the HIF-1 pathway as their only target [205]. Additionally, little is documented about their selectivity for other HIF-subunits (HIF-2/3 isoforms) and their capacity to influence the HIF switch. Currently, there is no medication available that only targets HIFs without interfering with other pathways. HIF inhibition or activation may have negative effects on living things, predicated on the therapeutic setting. Due to the potentially detrimental consequences of disrupting the HIF pathway, whether positively or negatively, developing strategies to precisely convey these medications to the targeted site while minimizing systemic exposure is an essential therapeutic consideration when exploring the use of HIF modulators.
Drug resistance is still a significant concern in anticancer therapy, and it is anticipated that HIF-1 inhibitors would have the same difficulty. Therefore, it must be determined if the on-specific effects of HIF inhibition are helpful for polypharmacy or harmful in terms of side effects. Multiple preclinical models of HIF-1-deficient tumors exhibit initially slow growth, followed by rapid growth, suggesting the presence of resistance and evasion mechanisms from HIF-1 inhibition [206,207,208,209]. By inhibiting tumor hypoxia, anti-hypoxia therapy reduces the expression of CD39 on the most terminally exhausted T cells, limiting its regulatory potential and promoting the improvement of CD8 + T cell effector function [210]. An increasing amount of evidence suggests that immunotherapy targeting immune checkpoints may be of greater significance after alleviating tumor hypoxia. It is crucial to comprehend the HIF-1-independent processes that may control these metabolic alterations in hypoxic cancerous cells to prevent the possible challenge of resistance to treatment.
The impressive anticancer effects for the majority of combinations are mostly restricted to animal tumor models. Choosing the best preclinical model to determine the activities of combination regimens is a major difficulty. Overall, compared to syngeneic murine models, which are often used, humanized models developed from patients may provide a more useful assessment of efficacy.This is undoubtedly the case for both combination immunotherapy and HIF inhibitor-based clinical anti-cancer treatment. A difficult task in the development of combination treatment is optimizing the delivery strategy, which includes dose, scheduling, and sequencing. Choosing the ideal medicine combination and finding the biomarkers that show treatment efficacy are other unsolved problems. Due to the heterogeneity and progression of tumors, a liquid biopsy could provide a real-time biological marker for guiding personalized immunotherapy by dynamically monitoring the immune milieu of the TME [211]. In summary, we believe that patients should get a customized mix of treatments based on immunological profile and other prognostic factors and HIF inhibitors may eventually offer better clinical cancer therapy options.
Data availability
Not applicable.
Abbreviations
- TFs:
-
Transcription factors
- HIFs:
-
Hypoxia-inducible factors
- CPIs:
-
Immune checkpoint inhibitors
- O2 :
-
Oxygen
- TFs:
-
Transcription factors
- EMT:
-
Epithelial-mesenchymal transition
- VHL:
-
Von Hippel-Lindau
- ARNT:
-
Aryl hydrocarbon receptor nuclear translocator
- PAS:
-
PER-ARNT-SIM
- bHLH:
-
Basic helix-loop-helix
- TADs:
-
Transactivation domains
- ODD:
-
Oxygen-dependent degradation domain
- FIH-1:
-
Factor-inhibiting hypoxia-inducible factor-1
- PHDs:
-
Prolinehydroxylases
- pVHL:
-
VHL tumour suppressor protein
- Fe2+ :
-
Iron ions
- CBP/CREBBP:
-
CREB-binding protein
- HRE:
-
Hypoxia response element
- SIRT1:
-
Sirtuin 1
- TME:
-
Tumor microenvironment
- MDSCs:
-
Myeloid-derived suppressor cells
- TAMs:
-
Tumor-associated macrophages
- MICA:
-
Major histocompatibility complex class I chain-related molecule
- HCC:
-
Hepatocellular carcinoma
- ENTPD2:
-
Ectonucleoside triphosphate diphosphohydrolase 2
- ICB:
-
Immune checkpoint blockade
- SOD3:
-
Superoxide dismutase
- EC:
-
Endothelial cells
- IFN:
-
Interferon
- VISTA:
-
V-domain Ig suppressor of T-cell activation
- CD47:
-
Cluster of differentiation 47
- HLA-G:
-
Human leukocyte antigen G
- ccRCC:
-
Clear cell renal cell carcinoma
- CAIX:
-
Carbonic anhydrase 9
- CRC:
-
Colorectal cancer
- DMOG:
-
Dimethyloxalylglycine
- TUBB3:
-
Tubulin beta-3 chain
- CSCs:
-
Cancer stem cells
- HAPs:
-
Hypoxia-activated prodrugs
- UPR:
-
Unfolded protein response
- HDAC:
-
Histone deacetylases
- ACT:
-
Adoptive cell transfer
- ICI:
-
Immune checkpoint inhibitor
- irAEs:
-
Immune-related adverse events
- TILs:
-
Tumor-infiltrating lymphocytes
References
Mckeown SR. Defining normoxia, physoxia and hypoxia in tumours-implications for treatment response. Br J Radiol. 2014;87(1035):20130676.
Span PN, Bussink J. Biology of hypoxia. Semin Nucl Med. 2015;45(2):101–9.
Semenza GL. Perspectives on oxygen sensing. Cell. 1999;98(3):281–4.
Semenza GL. Hypoxia-inducible factors in physiology and medicine. Cell. 2012;148(3):399–408.
Ratcliffe PJ. Oxygen sensing and hypoxia signalling pathways in animals: the implications of physiology for cancer. J Physiol. 2013;591(8):2027–42.
Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer. 2003;3(10):721–32.
Liao D, Johnson RS. Hypoxia: a key regulator of angiogenesis in cancer. Cancer Metastasis Rev. 2007;26(2):281–90.
Lee K, Zhang H, Qian DZ, et al. Acriflavine inhibits HIF-1 dimerization, tumor growth, and vascularization. Proc Natl Acad Sci USA. 2009;106(42):17910–5.
Barnhart BC, Simon MC. Metastasis and stem cell pathways. Cancer Metastasis Rev. 2007;26(2):261–71.
Suda T, Takubo K, Semenza GL. Metabolic regulation of hematopoietic stem cells in the hypoxic niche. Cell Stem Cell. 2011;9(4):298–310.
Wang Y, Liu Y, Malek SN, et al. Targeting HIF1α eliminates cancer stem cells in hematological malignancies. Cell Stem Cell. 2011;8(4):399–411.
Zhang H, Gao P, Fukuda R, et al. HIF-1 inhibits mitochondrial biogenesis and cellular respiration in VHL-deficient renal cell carcinoma by repression of C-MYC activity. Cancer Cell. 2007;11(5):407–20.
Luo W, Hu H, Chang R, et al. Pyruvate kinase M2 is a PHD3-stimulated coactivator for hypoxia-inducible factor 1. Cell. 2011;145(5):732–44.
Franovic A, Gunaratnam L, Smith K, et al. Translational up-regulation of the EGFR by tumor hypoxia provides a nonmutational explanation for its overexpression in human cancer. Proc Natl Acad Sci USA. 2007;104(32):13092–7.
Lau CK, Yang ZF, Ho DW, et al. An Akt/hypoxia-inducible factor-1alpha/platelet-derived growth factor-BB autocrine loop mediates hypoxia-induced chemoresistance in liver cancer cells and tumorigenic hepatic progenitor cells. Clin Cancer Res. 2009;15(10):3462–71.
Wu MZ, Tsai YP, Yang MH, et al. Interplay between HDAC3 and WDR5 is essential for hypoxia-induced epithelial-mesenchymal transition. Mol Cell. 2011;43(5):811–22.
Esteban MA, Tran MG, Harten SK, et al. Regulation of E-cadherin expression by VHL and hypoxia-inducible factor. Can Res. 2006;66(7):3567–75.
Krishnamachary B, Zagzag D, Nagasawa H, et al. Hypoxia-inducible factor-1-dependent repression of E-cadherin in von Hippel-Lindau tumor suppressor-null renal cell carcinoma mediated by TCF3, ZFHX1A, and ZFHX1B. Can Res. 2006;66(5):2725–31.
Sullivan R, Graham CH. Hypoxia-driven selection of the metastatic phenotype. Cancer Metastasis Rev. 2007;26(2):319–31.
Wong CC, Gilkes DM, Zhang H, et al. Hypoxia-inducible factor 1 is a master regulator of breast cancer metastatic niche formation. Proc Natl Acad Sci USA. 2011;108(39):16369–74.
Zhang H, Wong CC, Wei H, et al. HIF-1-dependent expression of angiopoietin-like 4 and L1CAM mediates vascular metastasis of hypoxic breast cancer cells to the lungs. Oncogene. 2012;31(14):1757–70.
Moeller BJ, Richardson RA, Dewhirst MW. Hypoxia and radiotherapy: opportunities for improved outcomes in cancer treatment. Cancer Metastasis Rev. 2007;26(2):241–8.
Rohwer N, Cramer T. Hypoxia-mediated drug resistance: novel insights on the functional interaction of HIFs and cell death pathways. Drug Resist Updat. 2011;14(3):191–201.
Semenza GL. Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene. 2010;29(5):625–34.
Majmundar AJ, Wong WJ, Simon MC. Hypoxia-inducible factors and the response to hypoxic stress. Mol Cell. 2010;40(2):294–309.
Kaelin WG Jr, Ratcliffe PJ. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol Cell. 2008;30(4):393–402.
Wang GL, Jiang BH, Rue EA, et al. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci USA. 1995;92(12):5510–4.
Kallio PJ, Okamoto K, O’brien S, et al. Signal transduction in hypoxic cells: inducible nuclear translocation and recruitment of the CBP/p300 coactivator by the hypoxia-inducible factor-1alpha. EMBO J. 1998;17(22):6573–86.
Loboda A, Jozkowicz A, Dulak J. HIF-1 versus HIF-2–is one more important than the other? Vascul Pharmacol. 2012;56(5–6):245–51.
Koh MY, Powis G. Passing the baton: the HIF switch. Trends Biochem Sci. 2012;37(9):364–72.
Pasanen A, Heikkilä M, Rautavuoma K, et al. Hypoxia-inducible factor (HIF)-3alpha is subject to extensive alternative splicing in human tissues and cancer cells and is regulated by HIF-1 but not HIF-2. Int J Biochem Cell Biol. 2010;42(7):1189–200.
Maynard MA, Evans AJ, Hosomi T, et al. Human HIF-3alpha4 is a dominant-negative regulator of HIF-1 and is down-regulated in renal cell carcinoma. FASEB J. 2005;19(11):1396–406.
Zhang P, Yao Q, Lu L, et al. Hypoxia-inducible factor 3 is an oxygen-dependent transcription activator and regulates a distinct transcriptional response to hypoxia. Cell Rep. 2014;6(6):1110–21.
Janaszak-Jasiecka A, Bartoszewska S, Kochan K, et al. miR-429 regulates the transition between hypoxia-inducible factor (HIF)1A and HIF3A expression in human endothelial cells. Sci Rep. 2016;6:22775.
Talks KL, Turley H, Gatter KC, et al. The expression and distribution of the hypoxia-inducible factors HIF-1alpha and HIF-2alpha in normal human tissues, cancers, and tumor-associated macrophages. Am J Pathol. 2000;157(2):411–21.
Iommarini L, Porcelli AM, Gasparre G, et al. Non-canonical mechanisms regulating hypoxia-inducible factor 1 alpha in cancer. Front Oncol. 2017;7:286.
Maxwell PH, Wiesener MS, Chang GW, et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature. 1999;399(6733):271–5.
Appelhoff RJ, Tian YM, Raval RR, et al. Differential function of the prolyl hydroxylases PHD1, PHD2, and PHD3 in the regulation of hypoxia-inducible factor. J Biol Chem. 2004;279(37):38458–65.
Berra E, Benizri E, Ginouvès A, et al. HIF prolyl-hydroxylase 2 is the key oxygen sensor setting low steady-state levels of HIF-1alpha in normoxia. EMBO J. 2003;22(16):4082–90.
Keith B, Johnson RS, Simon MC. HIF1α and HIF2α: sibling rivalry in hypoxic tumour growth and progression. Nat Rev Cancer. 2011;12(1):9–22.
Lando D, Peet DJ, Whelan DA, et al. Asparagine hydroxylation of the HIF transactivation domain a hypoxic switch. Science. 2002;295(5556):858–61.
Ruas JL, Poellinger L, Pereira T. Role of CBP in regulating HIF-1-mediated activation of transcription. J Cell Sci. 2005;118(Pt 2):301–11.
Mahon PC, Hirota K, Semenza GL. FIH-1: a novel protein that interacts with HIF-1alpha and VHL to mediate repression of HIF-1 transcriptional activity. Genes Dev. 2001;15(20):2675–86.
Ziello JE, Jovin IS, Huang Y. Hypoxia-Inducible Factor (HIF)-1 regulatory pathway and its potential for therapeutic intervention in malignancy and ischemia. Yale J Biol Med. 2007;80(2):51–60.
Wang GL, Semenza GL. General involvement of hypoxia-inducible factor 1 in transcriptional response to hypoxia. Proc Natl Acad Sci USA. 1993;90(9):4304–8.
Hubbi ME, Semenza GL. Regulation of cell proliferation by hypoxia-inducible factors. Am J Physiol Cell Physiol. 2015;309(12):C775-782.
Yang MH, Wu MZ, Chiou SH, et al. Direct regulation of TWIST by HIF-1alpha promotes metastasis. Nat Cell Biol. 2008;10(3):295–305.
Liao D, Corle C, Seagroves TN, et al. Hypoxia-inducible factor-1alpha is a key regulator of metastasis in a transgenic model of cancer initiation and progression. Can Res. 2007;67(2):563–72.
Hiraga T, Kizaka-Kondoh S, Hirota K, et al. Hypoxia and hypoxia-inducible factor-1 expression enhance osteolytic bone metastases of breast cancer. Can Res. 2007;67(9):4157–63.
Semenza GL, Roth PH, Fang HM, et al. Transcriptional regulation of genes encoding glycolytic enzymes by hypoxia-inducible factor 1. J Biol Chem. 1994;269(38):23757–63.
Seagroves TN, Ryan HE, Lu H, et al. Transcription factor HIF-1 is a necessary mediator of the pasteur effect in mammalian cells. Mol Cell Biol. 2001;21(10):3436–44.
Wykoff CC, Beasley NJ, Watson PH, et al. Hypoxia-inducible expression of tumor-associated carbonic anhydrases. Can Res. 2000;60(24):7075–83.
Jiang BH, Agani F, Passaniti A, et al. V-SRC induces expression of hypoxia-inducible factor 1 (HIF-1) and transcription of genes encoding vascular endothelial growth factor and enolase 1: involvement of HIF-1 in tumor progression. Can Res. 1997;57(23):5328–35.
Chen KF, Lai YY, Sun HS, et al. Transcriptional repression of human cad gene by hypoxia inducible factor-1alpha. Nucleic Acids Res. 2005;33(16):5190–8.
Mole DR, Blancher C, Copley RR, et al. Genome-wide association of hypoxia-inducible factor (HIF)-1alpha and HIF-2alpha DNA binding with expression profiling of hypoxia-inducible transcripts. J Biol Chem. 2009;284(25):16767–75.
Lim JH, Lee YM, Chun YS, et al. Sirtuin 1 modulates cellular responses to hypoxia by deacetylating hypoxia-inducible factor 1alpha. Mol Cell. 2010;38(6):864–78.
Koh MY, Sayegh N, Agarwal N. Seeing the forest for the trees-single-cell atlases link CD8(+) T cells and macrophages to disease progression and treatment response in kidney cancer. Cancer Cell. 2021;39(5):594–6.
Wang B, Zhao Q, Zhang Y, et al. Targeting hypoxia in the tumor microenvironment: a potential strategy to improve cancer immunotherapy. J Exp Clin Cancer Res. 2021;40(1):24.
Baginska J, Viry E, Berchem G, et al. Granzyme B degradation by autophagy decreases tumor cell susceptibility to natural killer-mediated lysis under hypoxia. Proc Natl Acad Sci USA. 2013;110(43):17450–5.
Noman MZ, Buart S, Romero P, et al. Hypoxia-inducible miR-210 regulates the susceptibility of tumor cells to lysis by cytotoxic T cells. Can Res. 2012;72(18):4629–41.
Noman MZ, Janji B, Kaminska B, et al. Blocking hypoxia-induced autophagy in tumors restores cytotoxic T-cell activity and promotes regression. Can Res. 2011;71(18):5976–86.
Ren L, Yu Y, Wang L, et al. Hypoxia-induced CCL28 promotes recruitment of regulatory T cells and tumor growth in liver cancer. Oncotarget. 2016;7(46):75763–73.
Facciabene A, Peng X, Hagemann IS, et al. Tumour hypoxia promotes tolerance and angiogenesis via CCL28 and T(reg) cells. Nature. 2011;475(7355):226–30.
Hu M, Li Y, Lu Y, et al. The regulation of immune checkpoints by the hypoxic tumor microenvironment. PeerJ. 2021;9:e11306.
Torres N, Regge MV, Secchiari F, et al. Restoration of antitumor immunity through anti-MICA antibodies elicited with a chimeric protein. J Immunother Cancer. 2020;8(1):e000233.
Chiu DK, Tse AP, Xu IM, et al. Hypoxia inducible factor HIF-1 promotes myeloid-derived suppressor cells accumulation through ENTPD2/CD39L1 in hepatocellular carcinoma. Nat Commun. 2017;8(1):517.
Carmona-Rodríguez L, Martínez-Rey D, Fernández-Aceñero MJ, et al. SOD3 induces a HIF-2α-dependent program in endothelial cells that provides a selective signal for tumor infiltration by T cells. J Immunother Cancer. 2020;8(1):e000432.
Sun X, Kanwar JR, Leung E, et al. Gene transfer of antisense hypoxia inducible factor-1 alpha enhances the therapeutic efficacy of cancer immunotherapy. Gene Ther. 2001;8(8):638–45.
Lequeux A, Noman MZ, Xiao M, et al. Targeting HIF-1 alpha transcriptional activity drives cytotoxic immune effector cells into melanoma and improves combination immunotherapy. Oncogene. 2021;40(28):4725–35.
Kheshtchin N, Arab S, Ajami M, et al. Inhibition of HIF-1α enhances anti-tumor effects of dendritic cell-based vaccination in a mouse model of breast cancer. Cancer Immunol Immunother. 2016;65(10):1159–67.
Messai Y, Gad S, Noman MZ, et al. Renal cell carcinoma programmed death-ligand 1, a new direct target of hypoxia-inducible factor-2 alpha, is regulated by von Hippel-Lindau gene mutation status. Eur Urol. 2016;70(4):623–32.
Noman MZ, Desantis G, Janji B, et al. PD-L1 is a novel direct target of HIF-1α, and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J Exp Med. 2014;211(5):781–90.
Barsoum IB, Smallwood CA, Siemens DR, et al. A mechanism of hypoxia-mediated escape from adaptive immunity in cancer cells. Can Res. 2014;74(3):665–74.
Pinato DJ, Black JR, Trousil S, et al. Programmed cell death ligands expression in phaeochromocytomas and paragangliomas: relationship with the hypoxic response, immune evasion and malignant behavior. Oncoimmunology. 2017;6(11):e1358332.
Curigliano G, Criscitiello C, Gelao L, et al. Molecular pathways: human leukocyte antigen G (HLA-G). Clin Cancer Res. 2013;19(20):5564–71.
Garziera M, Scarabel L, Toffoli G. Hypoxic modulation of HLA-G expression through the metabolic sensor HIF-1 in human cancer cells. J Immunol Res. 2017;2017:4587520.
Mouillot G, Marcou C, Zidi I, et al. Hypoxia modulates HLA-G gene expression in tumor cells. Hum Immunol. 2007;68(4):277–85.
Yaghi L, Poras I, Simoes RT, et al. Hypoxia inducible factor-1 mediates the expression of the immune checkpoint HLA-G in glioma cells through hypoxia response element located in exon 2. Oncotarget. 2016;7(39):63690–707.
Sasaki T, Kanaseki T, Shionoya Y, et al. Microenvironmental stresses induce HLA-E/Qa-1 surface expression and thereby reduce CD8(+) T-cell recognition of stressed cells. Eur J Immunol. 2016;46(4):929–40.
Logtenberg MEW, Scheeren FA, Schumacher TN. The CD47-SIRPα immune checkpoint. Immunity. 2020;52(5):742–52.
Zhang H, Lu H, Xiang L, et al. HIF-1 regulates CD47 expression in breast cancer cells to promote evasion of phagocytosis and maintenance of cancer stem cells. Proc Natl Acad Sci USA. 2015;112(45):E6215-6223.
Michaels AD, Newhook TE, Adair SJ, et al. CD47 blockade as an adjuvant immunotherapy for resectable pancreatic cancer. Clin Cancer Res. 2018;24(6):1415–25.
Noman MZ, Hasmim M, Lequeux A, et al. Improving cancer immunotherapy by targeting the hypoxic tumor microenvironment: new opportunities and challenges. Cells. 2019;8(9):1083.
Dysthe M, Parihar R. Myeloid-derived suppressor cells in the tumor microenvironment. Adv Exp Med Biol. 2020;1224:117–40.
Deng J, Li J, Sarde A, et al. Hypoxia-induced VISTA promotes the suppressive function of myeloid-derived suppressor cells in the tumor microenvironment. Cancer Immunol Res. 2019;7(7):1079–90.
Palazón A, Martínez-Forero I, Teijeira A, et al. The HIF-1α hypoxia response in tumor-infiltrating T lymphocytes induces functional CD137 (4–1BB) for immunotherapy. Cancer Discov. 2012;2(7):608–23.
Wang X, Zhang T, Song Z, et al. Tumor CD73/A2aR adenosine immunosuppressive axis and tumor-infiltrating lymphocytes in diffuse large B-cell lymphoma: correlations with clinicopathological characteristics and clinical outcome. Int J Cancer. 2019;145(5):1414–22.
Leone RD, Emens LA. Targeting adenosine for cancer immunotherapy. J Immunother Cancer. 2018;6(1):57.
Zhang T, Liu H, Jiao L, et al. Genetic characteristics involving the PD-1/PD-L1/L2 and CD73/A2aR axes and the immunosuppressive microenvironment in DLBCL. J Immunother Cancer. 2022;10(4):e004114.
Chen Q, Yin H, He J, et al. Tumor microenvironment responsive CD8(+) T cells and myeloid-derived suppressor cells to trigger CD73 inhibitor AB680-based synergistic therapy for pancreatic cancer. Adv Sci. 2023. https://doi.org/10.1002/advs.202302498.
Samain R, Maiques O, Monger J, et al. CD73 controls Myosin II-driven invasion, metastasis, and immunosuppression in amoeboid pancreatic cancer cells. Sci Adv. 2023;9(42):eadi0244.
Ploeg EM, Samplonius DF, Xiong X, et al. Bispecific antibody CD73xEGFR more selectively inhibits the CD73/adenosine immune checkpoint on cancer cells and concurrently counteracts pro-oncogenic activities of CD73 and EGFR. J Immunother Cancer. 2023;11(9):e006837.
Masjedi A, Hassannia H, Atyabi F, et al. Downregulation of A2AR by siRNA loaded PEG-chitosan-lactate nanoparticles restores the T cell mediated anti-tumor responses through blockage of PKA/CREB signaling pathway. Int J Biol Macromol. 2019;133:436–45.
Yuan CS, Teng Z, Yang S, et al. Reshaping hypoxia and silencing CD73 via biomimetic gelatin nanotherapeutics to boost immunotherapy. J Controll Releas. 2022;351:255–71.
Chambers AM, Matosevic S. Immunometabolic dysfunction of natural killer cells mediated by the hypoxia-CD73 axis in solid tumors. Front Mol Biosci. 2019;6:60.
Cosin-Roger J, Simmen S, Melhem H, et al. Hypoxia ameliorates intestinal inflammation through NLRP3/mTOR downregulation and autophagy activation. Nat Commun. 2017;8(1):98.
Barrett TD, Palomino HL, Brondstetter TI, et al. Pharmacological characterization of 1-(5-chloro-6-(trifluoromethoxy)-1H-benzoimidazol-2-yl)-1H-pyrazole-4-carboxylic acid (JNJ-42041935), a potent and selective hypoxia-inducible factor prolyl hydroxylase inhibitor. Mol Pharmacol. 2011;79(6):910–20.
Robinson A, Keely S, Karhausen J, et al. Mucosal protection by hypoxia-inducible factor prolyl hydroxylase inhibition. Gastroenterology. 2008;134(1):145–55.
Kobayashi H, Ohyama T, Kitamura-Miyazaki M, et al. Studies on novel HIF activators, A-503451sII: biological activities of A-503451A. J Antibiot. 2016;69(10):754–8.
Ozgür B, Helms HCC, Tornabene E, et al. Hypoxia increases expression of selected blood-brain barrier transporters GLUT-1, P-gp, SLC7A5 and TFRC, while maintaining barrier integrity, in brain capillary endothelial monolayers. Fluids Barriers CNS. 2022;19(1):1.
Knowles HJ, Tian YM, Mole DR, et al. Novel mechanism of action for hydralazine: induction of hypoxia-inducible factor-1alpha, vascular endothelial growth factor, and angiogenesis by inhibition of prolyl hydroxylases. Circ Res. 2004;95(2):162–9.
Peng G, Liu Y. Hypoxia-inducible factors in cancer stem cells and inflammation. Trends Pharmacol Sci. 2015;36(6):374–83.
Terzuoli E, Puppo M, Rapisarda A, et al. Aminoflavone, a ligand of the aryl hydrocarbon receptor, inhibits HIF-1alpha expression in an AhR-independent fashion. Can Res. 2010;70(17):6837–48.
Shackelford DB, Vasquez DS, Corbeil J, et al. mTOR and HIF-1alpha-mediated tumor metabolism in an LKB1 mouse model of Peutz-Jeghers syndrome. Proc Natl Acad Sci USA. 2009;106(27):11137–42.
Liu YV, Baek JH, Zhang H, et al. RACK1 competes with HSP90 for binding to HIF-1alpha and is required for O(2)-independent and HSP90 inhibitor-induced degradation of HIF-1alpha. Mol Cell. 2007;25(2):207–17.
Molineaux SM. Molecular pathways: targeting proteasomal protein degradation in cancer. Clin Cancer Res. 2012;18(1):15–20.
Befani CD, Vlachostergios PJ, Hatzidaki E, et al. Bortezomib represses HIF-1α protein expression and nuclear accumulation by inhibiting both PI3K/Akt/TOR and MAPK pathways in prostate cancer cells. J Mol Med. 2012;90(1):45–54.
Wallace EM, Rizzi JP, Han G, et al. A small-molecule antagonist of HIF2α is efficacious in preclinical models of renal cell carcinoma. Can Res. 2016;76(18):5491–500.
Raspaglio G, Petrillo M, Martinelli E, et al. Sox9 and Hif-2α regulate TUBB3 gene expression and affect ovarian cancer aggressiveness. Gene. 2014;542(2):173–81.
Warnecke C, Griethe W, Weidemann A, et al. Activation of the hypoxia-inducible factor-pathway and stimulation of angiogenesis by application of prolyl hydroxylase inhibitors. FASEB J. 2003;17(9):1186–8.
Ivan M, Kaelin WG Jr. The EGLN-HIF O(2)-sensing system: multiple inputs and feedbacks. Mol Cell. 2017;66(6):772–9.
Kim WY, Safran M, Buckley MR, et al. Failure to prolyl hydroxylate hypoxia-inducible factor alpha phenocopies VHL inactivation in vivo. EMBO J. 2006;25(19):4650–62.
Rodriguez J, Pilkington R, Garcia Munoz A, et al. Substrate-trapped interactors of PHD3 and FIH cluster in distinct signaling pathways. Cell Rep. 2016;14(11):2745–60.
Zhu ZY, Wang F, Jia CH, et al. Apigenin-induced HIF-1α inhibitory effect improves abnormal glucolipid metabolism in AngII/hypoxia-stimulated or HIF-1α-overexpressed H9c2 cells. Phytomedicine. 2019;62:152713.
Zhang H, Qian DZ, Tan YS, et al. Digoxin and other cardiac glycosides inhibit HIF-1alpha synthesis and block tumor growth. Proc Natl Acad Sci USA. 2008;105(50):19579–86.
Carbonaro M, O’brate A, Giannakakou P. Microtubule disruption targets HIF-1alpha mRNA to cytoplasmic P-bodies for translational repression. J Cell Biol. 2011;192(1):83–99.
Kummar S, Raffeld M, Juwara L, et al. Multihistology, target-driven pilot trial of oral topotecan as an inhibitor of hypoxia-inducible factor-1α in advanced solid tumors. Clin Cancer Res. 2011;17(15):5123–31.
Creighton-Gutteridge M, Cardellina JH 2nd, Stephen AG, et al. Cell type-specific, topoisomerase II-dependent inhibition of hypoxia-inducible factor-1alpha protein accumulation by NSC 644221. Clin Cancer Res. 2007;13(3):1010–8.
Greenberger LM, Horak ID, Filpula D, et al. A RNA antagonist of hypoxia-inducible factor-1alpha, EZN-2968, inhibits tumor cell growth. Mol Cancer Ther. 2008;7(11):3598–608.
Sun HL, Liu YN, Huang YT, et al. YC-1 inhibits HIF-1 expression in prostate cancer cells: contribution of Akt/NF-kappaB signaling to HIF-1alpha accumulation during hypoxia. Oncogene. 2007;26(27):3941–51.
Schwartz DL, Bankson JA, Lemos R Jr, et al. Radiosensitization and stromal imaging response correlates for the HIF-1 inhibitor PX-478 given with or without chemotherapy in pancreatic cancer. Mol Cancer Ther. 2010;9(7):2057–67.
Coltella N, Valsecchi R, Ponente M, et al. Synergistic leukemia eradication by combined treatment with retinoic acid and HIF inhibition by EZN-2208 (PEG-SN38) in preclinical models of PML-RARα and PLZF-RARα-driven leukemia. Clin Cancer Res. 2015;21(16):3685–94.
Lee SH, Jee JG, Bae JS, et al. A group of novel HIF-1α inhibitors, glyceollins, blocks HIF-1α synthesis and decreases its stability via inhibition of the PI3K/AKT/mTOR pathway and Hsp90 binding. J Cell Physiol. 2015;230(4):853–62.
Kinget L, Roussel E, Verbiest A, et al. MicroRNAs targeting HIF-2α, VEGFR1 and/or VEGFR2 as potential predictive biomarkers for VEGFR tyrosine kinase and HIF-2α inhibitors in metastatic clear-cell renal cell carcinoma. Cancers. 2021;13(12):3099.
Samuni Y, Cook JA, Choudhuri R, et al. Inhibition of adipogenesis by Tempol in 3T3-L1 cells. Free Radical Biol Med. 2010;49(4):667–73.
Gao P, Zhang H, Dinavahi R, et al. HIF-dependent antitumorigenic effect of antioxidants in vivo. Cancer Cell. 2007;12(3):230–8.
Ahmad F, Dixit D, Joshi SD, et al. G9a inhibition induced PKM2 regulates autophagic responses. Int J Biochem Cell Biol. 2016;78:87–95.
Kim YH, Coon A, Baker AF, et al. Antitumor agent PX-12 inhibits HIF-1α protein levels through an Nrf2/PMF-1-mediated increase in spermidine/spermine acetyl transferase. Cancer Chemother Pharmacol. 2011;68(2):405–13.
Qian DZ, Kachhap SK, Collis SJ, et al. Class II histone deacetylases are associated with VHL-independent regulation of hypoxia-inducible factor 1 alpha. Can Res. 2006;66(17):8814–21.
Guan Y, Reddy KR, Zhu Q, et al. G-rich oligonucleotides inhibit HIF-1alpha and HIF-2alpha and block tumor growth. Mol Ther. 2010;18(1):188–97.
Hamsa TP, Kuttan G. Harmine inhibits tumour specific neo-vessel formation by regulating VEGF, MMP, TIMP and pro-inflammatory mediators both in vivo and in vitro. Eur J Pharmacol. 2010;649(1–3):64–73.
Chintala S, Tóth K, Cao S, et al. Se-methylselenocysteine sensitizes hypoxic tumor cells to irinotecan by targeting hypoxia-inducible factor 1alpha. Cancer Chemother Pharmacol. 2010;66(5):899–911.
Harada H, Itasaka S, Zhu Y, et al. Treatment regimen determines whether an HIF-1 inhibitor enhances or inhibits the effect of radiation therapy. Br J Cancer. 2009;100(5):747–57.
Xie C, Yagai T, Luo Y, et al. Activation of intestinal hypoxia-inducible factor 2α during obesity contributes to hepatic steatosis. Nat Med. 2017;23(11):1298–308.
Chen W, Hill H, Christie A, et al. Targeting renal cell carcinoma with a HIF-2 antagonist. Nature. 2016;539(7627):112–7.
Jonasch E, Donskov F, Iliopoulos O, et al. Belzutifan for renal cell carcinoma in von hippel-lindau disease. N Engl J Med. 2021;385(22):2036–46.
Ban HS, Kim BK, Lee H, et al. The novel hypoxia-inducible factor-1α inhibitor IDF-11774 regulates cancer metabolism, thereby suppressing tumor growth. Cell Death Dis. 2017;8(6):e2843.
Lee K, Ban HS, Naik R, et al. Identification of malate dehydrogenase 2 as a target protein of the HIF-1 inhibitor LW6 using chemical probes. Angew Chem Int Ed Engl. 2013;52(39):10286–9.
Huang YC, Huang FI, Mehndiratta S, et al. Anticancer activity of MPT0G157, a derivative of indolylbenzenesulfonamide, inhibits tumor growth and angiogenesis. Oncotarget. 2015;6(21):18590–601.
Kim KH, Kim D, Park JY, et al. NNC 55–0396, a T-type Ca2+ channel inhibitor, inhibits angiogenesis via suppression of hypoxia-inducible factor-1α signal transduction. J Mol Med. 2015;93(5):499–509.
Dilly AK, Lee YJ, Zeh HJ, et al. Targeting hypoxia-mediated mucin 2 production as a therapeutic strategy for mucinous tumors. Transl Res. 2016;169:19-30.e11.
Liu R, Yang H, Chen Z, et al. Design, synthesis and biological evaluation of (R)-5-methylpyrrolidin-2-ones as p300 bromodomain inhibitors with anti-tumor activities in multiple tumor lines. Bioorg Chem. 2022;124:105803.
Hede K. Low-dose anthracyclines may block HIF-1 and stop tumor growth. J Natl Cancer Inst. 2009;101(6):368–70.
Shah RR. Safety and tolerability of histone deacetylase (HDAC) inhibitors in oncology. Drug Saf. 2019;42(2):235–45.
Qiu GZ, Jin MZ, Dai JX, et al. Reprogramming of the tumor in the hypoxic niche: the emerging concept and associated therapeutic strategies. Trends Pharmacol Sci. 2017;38(8):669–86.
Bosco MC, D’orazi G, Del Bufalo D. Targeting hypoxia in tumor: a new promising therapeutic strategy. J Exp Clin Cancer Res. 2020;39(1):8.
Semenza GL. Pharmacologic targeting of hypoxia-inducible factors. Annu Rev Pharmacol Toxicol. 2019;59:379–403.
Fallah J, Rini BI. HIF inhibitors: status of current clinical development. Curr Oncol Rep. 2019;21(1):6.
Terry S, Faouzi Zaarour R, Hassan Venkatesh G, et al. Role of hypoxic stress in regulating tumor immunogenicity, resistance and plasticity. Int J Mol Sci. 2018;19(10):3044.
Lee SH, Golinska M, Griffiths JR. HIF-1-independent mechanisms regulating metabolic adaptation in hypoxic cancer cells. Cells. 2021;10(9):2371.
Fallah J, Brave MH, Weinstock C, et al. FDA approval summary: belzutifan for von hippel-lindau disease-associated tumors. Clin Cancer Res. 2022;28(22):4843–8.
Motzer RJ, Schmidinger M, Eto M, et al. LITESPARK-011: belzutifan plus lenvatinib vs cabozantinib in advanced renal cell carcinoma after anti-PD-1/PD-L1 therapy. Future Oncol. 2023;19(2):113–21.
Choueiri TK, Mcdermott DF, Merchan J, et al. Belzutifan plus cabozantinib for patients with advanced clear cell renal cell carcinoma previously treated with immunotherapy: an open-label, single-arm, phase 2 study. Lancet Oncol. 2023. https://doi.org/10.1016/S1470-2045(23)00097-9.
Gebrael G, Sahu KK, Agarwal N, et al. Update on combined immunotherapy for the treatment of advanced renal cell carcinoma. Hum Vaccin Immunother. 2023;19(1):2193528.
Garcia Garcia CJ, Huang Y, Fuentes NR, et al. Stromal HIF2 regulates immune suppression in the pancreatic cancer microenvironment. Gastroenterology. 2022;162(7):2018–31.
Zhao T, Ren H, Jia L, et al. Correction: Inhibition of HIF-1α by PX-478 enhances the anti-tumor effect of gemcitabine by inducing immunogenic cell death in pancreatic ductal adenocarcinoma. Oncotarget. 2019;10(53):5569–70.
Zhu Y, Zang Y, Zhao F, et al. Inhibition of HIF-1α by PX-478 suppresses tumor growth of esophageal squamous cell cancer in vitro and in vivo. Am J Cancer Res. 2017;7(5):1198–212.
Abd-Aziz N, Stanbridge EJ, Shafee N. Bortezomib attenuates HIF-1- but not HIF-2-mediated transcriptional activation. Oncol Lett. 2015;10(4):2192–6.
Schönberger T, Fandrey J, Prost-Fingerle K. Ways into understanding HIF inhibition. Cancers. 2021;13(1):159.
Salman S, Meyers DJ, Wicks EE, et al. HIF inhibitor 32–134D eradicates murine hepatocellular carcinoma in combination with anti-PD1 therapy. J Clin Invest. 2022. https://doi.org/10.1172/JCI156774.
Shannon AM, Bouchier-Hayes DJ, Condron CM, et al. Tumour hypoxia, chemotherapeutic resistance and hypoxia-related therapies. Cancer Treat Rev. 2003;29(4):297–307.
Colliez F, Gallez B, Jordan BF. Assessing tumor oxygenation for predicting outcome in radiation oncology: a review of studies correlating tumor hypoxic status and outcome in the preclinical and clinical settings. Front Oncol. 2017;7:10.
Ebos JM, Lee CR, Cruz-Munoz W, et al. Accelerated metastasis after short-term treatment with a potent inhibitor of tumor angiogenesis. Cancer Cell. 2009;15(3):232–9.
Loges S, Mazzone M, Hohensinner P, et al. Silencing or fueling metastasis with VEGF inhibitors: antiangiogenesis revisited. Cancer Cell. 2009;15(3):167–70.
Pàez-Ribes M, Allen E, Hudock J, et al. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell. 2009;15(3):220–31.
Tanne JH. FDA cancels approval for bevacizumab in advanced breast cancer. BMJ. 2011;343:d7684.
Zhang H, Wong CCL, Wei H, et al. Correction: HIF-1-dependent expression of angiopoietin-like 4 and L1CAM mediates vascular metastasis of hypoxic breast cancer cells to the lungs. Oncogene. 2021;40(8):1552–3.
Zhao F, Mancuso A, Bui TV, et al. Imatinib resistance associated with BCR-ABL upregulation is dependent on HIF-1alpha-induced metabolic reprograming. Oncogene. 2010;29(20):2962–72.
Rapisarda A, Shoemaker RH, Melillo G. Antiangiogenic agents and HIF-1 inhibitors meet at the crossroads. Cell cycle. 2009;8(24):4040–3.
Hamilton PT, Anholt BR, Nelson BH. Tumour immunotherapy: lessons from predator-prey theory. Nat Rev Immunol. 2022;22(12):765–75.
Sadeghi Rad H, Monkman J, Warkiani ME, et al. Understanding the tumor microenvironment for effective immunotherapy. Med Res Rev. 2021;41(3):1474–98.
Cai X, Zhan H, Ye Y, et al. Current progress and future perspectives of immune checkpoint in cancer and infectious diseases. Front Genet. 2021;12:785153.
Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12(4):252–64.
Nishimura H, Okazaki T, Tanaka Y, et al. Autoimmune dilated cardiomyopathy in PD-1 receptor-deficient mice. Science. 2001;291(5502):319–22.
Seidel JA, Otsuka A, Kabashima K. Anti-PD-1 and Anti-CTLA-4 therapies in cancer: mechanisms of action, efficacy, and limitations. Front Oncol. 2018;8:86.
Liebl MC, Hofmann TG. Identification of responders to immune checkpoint therapy: which biomarkers have the highest value? J Eur Acad Dermatol Venereol. 2019;33(Suppl 8):52–6.
Yi M, Zheng X, Niu M, et al. Combination strategies with PD-1/PD-L1 blockade: current advances and future directions. Mol Cancer. 2022;21(1):28.
Kalbasi A, Ribas A. Tumour-intrinsic resistance to immune checkpoint blockade. Nat Rev Immunol. 2020;20(1):25–39.
Galluzzi L, Chan TA, Kroemer G, et al. The hallmarks of successful anticancer immunotherapy. Sci Transl Med. 2018. https://doi.org/10.1126/scitranslmed.aat7807.
Paucek RD, Baltimore D, Li G. The cellular immunotherapy revolution: arming the immune system for precision therapy. Trends Immunol. 2019;40(4):292–309.
Hodi FS, O’day SJ, Mcdermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. New Engl J Med. 2010;363(8):711–23.
Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366(26):2443–54.
Brahmer JR, Tykodi SS, Chow LQ, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012;366(26):2455–65.
Robert C, Schachter J, Long GV, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372(26):2521–32.
Mangan BL, Mcalister RK, Balko JM, et al. Evolving insights into the mechanisms of toxicity associated with immune checkpoint inhibitor therapy. Br J Clin Pharmacol. 2020;86(9):1778–89.
Johnson DB, Reynolds KL, Sullivan RJ, et al. Immune checkpoint inhibitor toxicities: systems-based approaches to improve patient care and research. Lancet Oncol. 2020;21(8):e398–404.
Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med. 2018;378(2):158–68.
Johnson DB, Chandra S, Sosman JA. Immune checkpoint inhibitor toxicity in 2018. JAMA. 2018;320(16):1702–3.
Postow MA, Chesney J, Pavlick AC, et al. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med. 2015;372(21):2006–17.
Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2019;381(16):1535–46.
Bajorin DF, Witjes JA, Gschwend JE, et al. Adjuvant nivolumab versus placebo in muscle-invasive urothelial carcinoma. N Engl J Med. 2021;384(22):2102–14.
Hatfield SM, Kjaergaard J, Lukashev D, et al. Immunological mechanisms of the antitumor effects of supplemental oxygenation. Sci Transl Med. 2015;7(277):277ra230.
Reck M, Rodríguez-Abreu D, Robinson AG, et al. Updated analysis of KEYNOTE-024: pembrolizumab versus platinum-based chemotherapy for advanced non-small-cell lung cancer with PD-L1 tumor proportion score of 50% or greater. J Clin Oncol. 2019;37(7):537–46.
Luo F, Lu FT, Cao JX, et al. HIF-1α inhibition promotes the efficacy of immune checkpoint blockade in the treatment of non-small cell lung cancer. Cancer Lett. 2022;531:39–56.
Bailey CM, Liu Y, Liu M, et al. Targeting HIF-1α abrogates PD-L1-mediated immune evasion in tumor microenvironment but promotes tolerance in normal tissues. J Clin Invest. 2022. https://doi.org/10.1172/JCI150846.
Zandberg DP, Menk AV, Velez M, et al. Tumor hypoxia is associated with resistance to PD-1 blockade in squamous cell carcinoma of the head and neck. J Immunother Cancer. 2021;9(5):e002088.
Shurin MR, Umansky V. Cross-talk between HIF and PD-1/PD-L1 pathways in carcinogenesis and therapy. J Clin Invest. 2022. https://doi.org/10.1172/JCI159473.
Wang DY, Salem JE, Cohen JV, et al. Fatal toxic effects associated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Oncol. 2018;4(12):1721–8.
Herrmann J. Adverse cardiac effects of cancer therapies: cardiotoxicity and arrhythmia. Nat Rev Cardiol. 2020;17(8):474–502.
Fecher LA, Agarwala SS, Hodi FS, et al. Ipilimumab and its toxicities: a multidisciplinary approach. Oncologist. 2013;18(6):733–43.
Moey MYY, Gougis P, Goldschmidt V, et al. Increased reporting of fatal pneumonitis associated with immune checkpoint inhibitors: a WHO pharmacovigilance database analysis. Eur Respir J. 2020;55(6):2000038.
Vozy A, De Martin E, Johnson DB, et al. Increased reporting of fatal hepatitis associated with immune checkpoint inhibitors. Eur J Cancer. 2019;123:112–5.
Johnson DB, Manouchehri A, Haugh AM, et al. Neurologic toxicity associated with immune checkpoint inhibitors: a pharmacovigilance study. J Immunother Cancer. 2019;7(1):134.
Xia Y, Choi HK, Lee K. Recent advances in hypoxia-inducible factor (HIF)-1 inhibitors. Eur J Med Chem. 2012;49:24–40.
Maxwell PH, Eckardt KU. HIF prolyl hydroxylase inhibitors for the treatment of renal anaemia and beyond. Nat Rev Nephrol. 2016;12(3):157–68.
Maxwell PH, Dachs GU, Gleadle JM, et al. Hypoxia-inducible factor-1 modulates gene expression in solid tumors and influences both angiogenesis and tumor growth. Proc Natl Acad Sci USA. 1997;94(15):8104–9.
Williams KJ, Telfer BA, Airley RE, et al. A protective role for HIF-1 in response to redox manipulation and glucose deprivation: implications for tumorigenesis. Oncogene. 2002;21(2):282–90.
Griffiths JR, Mcsheehy PM, Robinson SP, et al. Metabolic changes detected by in vivo magnetic resonance studies of HEPA-1 wild-type tumors and tumors deficient in hypoxia-inducible factor-1beta (HIF-1beta): evidence of an anabolic role for the HIF-1 pathway. Can Res. 2002;62(3):688–95.
Golinska M, Troy H, Chung YL, et al. Adaptation to HIF-1 deficiency by upregulation of the AMP/ATP ratio and phosphofructokinase activation in hepatomas. BMC Cancer. 2011;11:198.
Vignali PDA, Depeaux K, Watson MJ, et al. Hypoxia drives CD39-dependent suppressor function in exhausted T cells to limit antitumor immunity. Nat Immunol. 2023;24(2):267–79.
Zhang T, Agarwal A, Almquist RG, et al. Expression of immune checkpoints on circulating tumor cells in men with metastatic prostate cancer. Biomark Res. 2021;9(1):14.
Funding
The study was supported by Major project of Wuxi Science and Technology Bureau (N20201006), Wuxi Double-Hundred Talent Fund Project (BJ2020076), Wuxi Health Commission Precision Medicine Project (J202106), Jiangsu Provincial Maternal and Child Health Research Project (F202034) and Jiangsu Provincial Six Talent Peaks Project (No. YY-124) and Graduate Research and Innovation Projects of Jiangsu Province (KYCX22_2435).
Author information
Authors and Affiliations
Contributions
LJY and JY conceived and designed the review. CLY and QZW wrote the manuscript. ZY participated in the whole process of chart review and article proofreading. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, J., Jiang, Y., Chen, L. et al. Associations between HIFs and tumor immune checkpoints: mechanism and therapy. Discov Onc 15, 2 (2024). https://doi.org/10.1007/s12672-023-00836-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12672-023-00836-7