Abstract
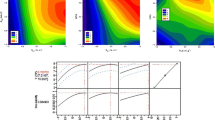
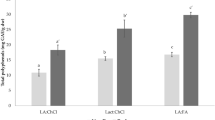
Apple peels were used as a model solid matrix for the comparative evaluation of aqueous mixtures of three bio-solvents, including ethanol, 2,3-butanediol (butanediol) and glycerol, with regard to polyphenol recovery. The evaluation process was based on a step-by-step optimisation of the extraction, taking into consideration basic factors, including solvent composition, liquid-to-solid ratio and temperature. A further detailed description of the extraction capacity of each solvent was carried out using kinetics. Optimal solvent concentrations were 50 % (v/v), 50 % (v/v) and 70 % (w/v) for ethanol, butanediol and glycerol, respectively, while optimal liquid-to-solid ratio and temperature were 100 mL g−1 and 80 °C for all solvents tested. The kinetic assay showed that extraction yields in total polyphenols obtained with these solvent systems displayed no statistical difference, although the overall extraction rate constant and diffusion coefficient were significantly higher for the extraction performed with 50 % (v/v) butanediol. The liquid chromatography-mass spectrometry examination of the extracts obtained under optimised conditions revealed that their polyphenolic profile was composed, in total, of 12 principal substances, but some qualitative differences among the extracts were also observed. The reducing power of the extract obtained with 50 % (v/v) ethanol was significantly higher, a finding attributed to possible synergistic effects among polyphenols.





Similar content being viewed by others
Abbreviations
- AAE:
-
Ascorbic acid equivalents
- APP:
-
Apple peels
- GAE:
-
Gallic acid equivalents
- TP:
-
Total polyphenols
- TPTZ:
-
2,4,6-Tripyridyl-s-triazine
- C solv :
-
Solvent concentration (%, v/v or w/v)
- C TP :
-
Total polyphenol concentration (mg GAE L−1)
- d :
-
Density (g cm−3)
- D e :
-
Effective diffusion coefficient (m2 s−1)
- h :
-
Initial extraction rate (mg g−1 min−1)
- k :
-
Extraction rate constant (g mg−1 min−1)
- PR :
-
Reducing power (μmol AAE g−1)
- R :
-
Particle radius (m)
- RL/S :
-
Liquid-to-solid ratio (mL g−1)
- t :
-
Time (min)
- t R :
-
Time required to enter the regular regime (min)
- T :
-
Temperature (°C)
- YTP :
-
Yield in total polyphenols (mg GAE g−1)
- YTP(s) :
-
Yield in total polyphenols at saturation (mg GAE g−1)
- ε :
-
Dielectric constant (dimensionless)
References
Mamma, D., Christakopoulos, P.: Biotransformation of citrus by-products into value added products. Waste Biomass Valoriz. 5, 529–549 (2014)
Ayala-Zavala, J., Vega-Vega, V., Rosas-Domínguez, C., Palafox-Carlos, H., Villa-Rodriguez, J., Siddiqui, M.W., Dávila-Aviña, J.E., González-Aguilar, G.E.: Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res. Int. 44, 1866–1874 (2011)
Djilas, S., Čanadanović-Brunet, J., Ćetković, G.: By-products of fruits processing as a source of phytochemicals. Chem. Ind. Chem. Eng. Q. 15, 191–202 (2009)
Kabir, F., Tow, W.W., Hamauzu, Y., Katayama, S., Tanaka, S., Nakamura, S.: Antioxidant and cytoprotective activities of extracts prepared from fruit and vegetable wastes and by-products. Food Chem. 167, 358–362 (2015)
Babbar, N., Oberoi, H.S., Sandhu, S.K.: Therapeutic and nutraceutical potential of bioactive compounds extracted from fruit residues. Crit. Rev. Food Sci. Nutr. 55, 319–337 (2015)
Dai, J., Mumper, R.J.: Plant phenolics: extraction, analysis and their antioxidant and anticancer properties. Molecules 15, 7313–7352 (2010)
Wijngaard, H., Hossain, M.B., Rai, D.K., Brunton, N.: Techniques to extract bioactive compounds from food by-products of plant origin. Food Res. Int. 46, 505–513 (2012)
Bai, X.L., Yue, T.L., Yuan, Y.H., Zhang, H.W.: Optimization of microwave-assisted extraction of polyphenols from apple pomace using response surface methodology and HPLC analysis. J. Sep. Sci. 33, 3751–3758 (2010)
Sekhon-Loodu, S., Warnakulasuriya, S.N., Rupasinghe, H.V., Shahidi, F.: Antioxidant ability of fractionated apple peel phenolics to inhibit fish oil oxidation. Food Chem. 140, 189–196 (2013)
Virot, M., Tomao, V., Le Bourvellec, C., Renard, C.M., Chemat, F.: Towards the industrial production of antioxidants from food processing by-products with ultrasound-assisted extraction. Ultrason. Sonochem. 17, 1066–1074 (2010)
Wijngaard, H.H., Brunton, N.: The optimisation of solid–liquid extraction of antioxidants from apple pomace by response surface methodology. J. Food Eng. 96, 134–140 (2010)
Ardi, M., Aroua, M., Hashim, N.A.: Progress, prospect and challenges in glycerol purification process: a review. Renew. Sustain. Energy Rev. 42, 1164–1173 (2015)
Apostolakis, A., Grigorakis, S., Makris, D.P.: Optimisation and comparative kinetics study of polyphenol extraction from olive leaves (Olea europaea) using heated water/glycerol mixtures. Sep. Purif. Technol. 128, 89–95 (2014)
Celińska, E., Grajek, W.: Biotechnological production of 2,3-butanediol—current state and prospects. Biotechnol. Adv. 27, 715–725 (2009)
Zeng, A.-P., Sabra, W.: Microbial production of diols as platform chemicals: recent progresses. Curr. Opin. Biotechnol. 22, 749–757 (2011)
Rabetafika, H.N., Bchir, B., Blecker, C., Richel, A.: Fractionation of apple by-products as source of new ingredients: current situation and perspectives. Trends Food Sci. Technol. 40, 99–114 (2014)
Makris, D.P., Kefalas, P.: Characterization of polyphenolic phytochemicals in red grape pomace. Int. J. Waste Resour. 126, (2013)
Makris, D.P., Boskou, G., Andrikopoulos, N.K.: Polyphenolic content and in vitro antioxidant characteristics of wine industry and other agri-food solid waste extracts. J. Food Compos. Anal. 20, 125–132 (2007)
Candrawinata, V., Golding, J., Roach, P., Stathopoulos, C.: Total phenolic content and antioxidant activity of apple pomace aqueous extract: effect of time, temperature and water to pomace ratio. Int. Food Res. J. 21, 2337–2344 (2014)
Cacace, J., Mazza, G.: Optimization of extraction of anthocyanins from black currants with aqueous ethanol. J. Food Sci. 68, 240–248 (2003)
Casazza, A.A., Aliakbarian, B., De Faveri, D., Fiori, L., Perego, P.: Antioxidants from winemaking wastes: a study on extraction parameters using response surface methodology. J. Food Biochem. 36, 28–37 (2012)
Rakotondramasy-Rabesiaka, L., Havet, J.-L., Porte, C.: Fauduet, H: estimation of effective diffusion and transfer rate during the protopine extraction process from Fumaria officinalis L. Sep. Purif. Technol. 76, 126–131 (2010)
Dubie, J., Stancik, A., Morra, M., Nindo, C.: Antioxidant extraction from mustard (Brassica juncea) seed meal using high-intensity ultrasound. J. Food Sci. 78, E542–E548 (2013)
Cacace, J., Mazza, G.: Mass transfer process during extraction of phenolic compounds from milled berries. J. Food Eng. 59, 379–389 (2003)
Khiari, Z., Makris, D.P., Kefalas, P.: An investigation on the recovery of antioxidant phenolics from onion solid wastes employing water/ethanol-based solvent systems. Food Bioprocess Technol. 2, 337–343 (2009)
Chan, S., Lee, C., Yap, C., Wan Aida, W., Ho, C.: Optimisation of extraction conditions for phenolic compounds from limau purut (Citrus hystrix) peels. Int. Food Res. J. 16, 203–213 (2009)
Cavdarova, M., Makris, D.P.: Extraction kinetics of phenolics from carob (Ceratonia siliqua L.) kibbles using environmentally benign solvents. Waste Biomass Valoriz. 5, 773–779 (2014)
Tzima, K., Kallithraka, S., Kotseridis, Y., Makris, D.P.: Kinetic modelling for flavanol extraction from red grape (Vitis vinifera L.) pomace using aqueous organic acid solutions. Int. Food Res. J. 21, 1919–1924 (2014)
Seikova, I., Simeonov, E., Ivanova, E.: Protein leaching from tomato seed—experimental kinetics and prediction of effective diffusivity. J. Food Eng. 61, 165–171 (2004)
Ho, C.H., Cacace, J.E., Mazza, G.: Mass transfer during pressurized low polarity water extraction of lignans from flaxseed meal. J. Food Eng. 89, 64–71 (2008)
Herodež, Š.S., Hadolin, M., Škerget, M., Knez, Ž.: Solvent extraction study of antioxidants from Balm (Melissa officinalis L.) leaves. Food Chem. 80, 275–282 (2003)
Tao, Y., Wu, D., Zhang, Q.-A., Sun, D.-W.: Ultrasound-assisted extraction of phenolics from wine lees: modeling, optimization and stability of extracts during storage. Ultrason. Sonochem. 21, 706–715 (2014)
Cacace, J., Mazza, G.: Extraction of anthocyanins and other phenolics from black currants with sulfured water. J. Agric. Food Chem. 50, 5939–5946 (2002)
Lameiras, P., Boudesocque, L., Mouloungui, Z., Renault, J.-H., Wieruszeski, J.-M., Lippens, G., Nuzillard, J.-M.: Glycerol and glycerol carbonate as ultraviscous solvents for mixture analysis by NMR. J. Magn. Reson. 212, 161–168 (2011)
Swami, M., Pawar, V., Hudge, P., Kumbharkhane, A.: Dielectric relaxation study of amines in 2, 3-butanediol mixture using picosecond time domain reflectometry technique. J. Mol. Liq. 190, 178–184 (2014)
Galanakis, C., Goulas, V., Tsakona, S., Manganaris, G., Gekas, V.: A knowledge base for the recovery of natural phenols with different solvents. Int. J. Food Prop. 16, 382–396 (2013)
Tsakona, S., Galanakis, C.M., Gekas, V.: Hydro-ethanolic mixtures for the recovery of phenols from Mediterranean plant materials. Food Bioprocess Technol. 5, 1384–1393 (2012)
Kefalas, P., Makris, D.P.: Liquid chromatography-mass spectrometry techniques in flavonoid analysis: recent advances. In: Boskou, D., Gerothanasis, I., Kefalas, P. (eds.) Antioxidant Plant Phenols: Sources, Structure-Activity Relationship, Current Trends in Analysis and Characterization, pp. 69–123. RESEARCH SIGNPOST Publ., Kerala (2006)
Bi, X., Zhang, J., Chen, C., Zhang, D., Li, P., Ma, F.: Anthocyanin contributes more to hydrogen peroxide scavenging than other phenolics in apple peel. Food Chem. 152, 205–209 (2014)
Ramirez-Ambrosi, M., Abad-Garcia, B., Viloria-Bernal, M., Garmon-Lobato, S., Berrueta, L., Gallo, B.: A new ultrahigh performance liquid chromatography with diode array detection coupled to electrospray ionization and quadrupole time-of-flight mass spectrometry analytical strategy for fast analysis and improved characterization of phenolic compounds in apple products. J. Chromatogr. A 1316, 78–91 (2013)
Karvela, E., Makris, D.P.: Assessment of the reducing effects in mixtures of grape (Vitis vinifera) seed extracts with α-tocopherol using response surface methodology. J. Microbiol. Biotechnol. Food Sci. 2, 771–787 (2012)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blidi, S., Bikaki, M., Grigorakis, S. et al. A Comparative Evaluation of Bio-solvents for the Efficient Extraction of Polyphenolic Phytochemicals: Apple Waste Peels as a Case Study. Waste Biomass Valor 6, 1125–1133 (2015). https://doi.org/10.1007/s12649-015-9410-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-015-9410-3