Abstract
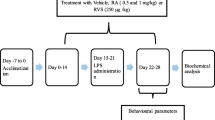
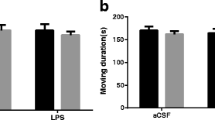
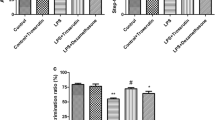
Hydrogen sulfide (H2S) is reported to have a neuroprotective activity; however, the role of H2S in neuroinflammation-induced neuronal damage is ambiguous. Here, we aimed to evaluate the underlying mechanisms for the neuroprotective effect of NaHS, a known H2S donor, against lipopolysaccharide (LPS)-induced memory impairment (MI). All the treatments were administered for 28 days, and LPS (0.25 mg/kg i.p.) was co-administered intermittently for 7 days from days 15 to 21. Morris water maze (MWM) and Y-maze tests were performed to evaluate MI. Neurodegeneration was histopathologically examined, and the brain homogenates were characterized for reduced glutathione (GSH), superoxide dismutase (SOD), malondialdehyde (MDA), tumor necrosis factor (TNF)-α, interleukin (IL)-6, caspase-3, c-Jun, and acetylcholinesterase (AChE) by biochemical analysis. H2S administration significantly improved spatial and working memory in MWM and Y-maze tasks, respectively. Exogenous H2S significantly reversed LPS-induced oxidative stress as evidenced by improved GSH, MDA, and SOD levels. H2S pretreatment significantly attenuated LPS-induced apoptosis and inflammation by decreasing c-Jun and caspase-3 levels and inhibiting TNF-α and IL-6, respectively. The decrease in these markers was supported by H&E and Nissl staining, which confirmed the anti-necrotic activity of H2S. However, there was no significant improvement in LPS-induced increase in AChE activity. These results indicate that chronic systemic inflammation leads to neurodegeneration and MI and H2S exerts its neuroprotective effect due to its anti-oxidative, anti-inflammatory, and anti-apoptotic potential via modulation of JNK and extrinsic apoptosis pathways.







Similar content being viewed by others
References
Aarsland D (2016) Cognitive impairment in Parkinson’s disease and dementia with Lewy bodies. Parkinsonism Relat Disord 22(Suppl 1):S144–S148. https://doi.org/10.1016/j.parkreldis.2015.09.034
Abe K, Kimura H (1996) The possible role of hydrogen sulfide as an endogenous neuromodulator. J Neurosci 16:1066–1071. https://doi.org/10.1523/JNEUROSCI.16-03-01066.1996
Alzheimer’s Association (2019) 2019 Alzheimer’s disease facts and figures includes a special report on Alzheimer’s detection in the primary care setting: connecting patients and physicians. Alzheimer’s Dement 15(3):321–387
Andreyev AY, Kushnareva YE, Starkov AA (2005) Mitochondrial metabolism of reactive oxygen species. Biochemistry (mosc) 70:200–214. https://doi.org/10.1007/s10541-005-0102-7
Bae J, Kumazoe M, Yamashita S, Tachibana H (2017) Hydrogen sulphide donors selectively potentiate a green tea polyphenol EGCG-induced apoptosis of multiple myeloma cells. Sci Rep 7:6665. https://doi.org/10.1038/s41598-017-06879-5
Banati RB, Gehrmann J, Schubert P, Kreutzberg GW (1993) Cytotoxicity of microglia. Glia 7:111–118. https://doi.org/10.1002/glia.440070117
Beheshti F, Hashemzehi M, Sabeti N et al (2019a) The effects of aminoguanidine on hippocampal cytokines, amyloid beta, brain-derived neurotrophic factor, memory and oxidative stress status in chronically lipopolysaccharide-treated rats. Cytokine 113:347–355. https://doi.org/10.1016/j.cyto.2018.10.005
Beheshti F, Hosseini M, Hashemzehi M et al (2019b) The effects of PPAR-γ agonist pioglitazone on hippocampal cytokines, brain-derived neurotrophic factor, memory impairment, and oxidative stress status in lipopolysaccharide-treated rats. Iran J Basic Med Sci 22:940–948. https://doi.org/10.22038/ijbms.2019.36165.8616
Biradar SM, Joshi H, Chheda TK (2012) Neuropharmacological effect of Mangiferin on brain cholinesterase and brain biogenic amines in the management of Alzheimer’s disease. Eur J Pharmacol 683:140–147. https://doi.org/10.1016/j.ejphar.2012.02.042
Bloom GS (2014) Amyloid-β and tau: the trigger and bullet in Alzheimer disease pathogenesis. JAMA Neurol 71:505–508. https://doi.org/10.1001/jamaneurol.2013.5847
Bratton SB, Walker G, Srinivasula SM et al (2001) Recruitment, activation and retention of caspases-9 and -3 by Apaf-1 apoptosome and associated XIAP complexes. EMBO J 20:998–1009. https://doi.org/10.1093/emboj/20.5.998
Cervellati C, Cremonini E, Bosi C et al (2013) Systemic oxidative stress in older patients with mild cognitive impairment or late onset Alzheimer’s disease. Curr Alzheimer Res 10:365–372. https://doi.org/10.2174/1567205011310040003
Chelikani P, Fita I, Loewen PC (2004) Diversity of structures and properties among catalases. Cell Mol Life Sci 61:192–208. https://doi.org/10.1007/s00018-003-3206-5
Chen G-F, Xu T-H, Yan Y et al (2017) Amyloid beta: structure, biology and structure-based therapeutic development. Acta Pharmacol Sin 38:1205–1235. https://doi.org/10.1038/aps.2017.28
Chen P-L, Wang W-J, Rao Y-Q et al (2018a) Serum containing Gengnianchun formula suppresses amyloid β-induced inflammatory cytokines in BV-2 microglial cells by inhibiting the NF-κB and JNK signaling pathways. Mol Med Rep 17:5043–5048. https://doi.org/10.3892/mmr.2018.8524
Chen X, Guo C, Kong J (2012) Oxidative stress in neurodegenerative diseases. Neural Regen Res 7:376–385. https://doi.org/10.3969/j.issn.1673-5374.2012.05.009
Chen Y, Jiang M, Li L et al (2018b) DL-3-n-butylphthalide reduces microglial activation in lipopolysaccharide-induced Parkinson’s disease model mice. Mol Med Rep 17:3884–3890. https://doi.org/10.3892/mmr.2017.8332
Cheng X-J, Gu J-X, Pang Y-P et al (2019) Tacrine-hydrogen sulfide donor hybrid ameliorates cognitive impairment in the aluminum chloride mouse model of Alzheimer’s disease. ACS Chem Neurosci 10:3500–3509. https://doi.org/10.1021/acschemneuro.9b00120
Cheng Y-S, Dai D-Z, Dai Y et al (2016) Exogenous hydrogen sulphide ameliorates diabetic cardiomyopathy in rats by reversing disordered calcium-handling system in sarcoplasmic reticulum. J Pharm Pharmacol 68:379–388. https://doi.org/10.1111/jphp.12517
Chowdhury AA, Gawali NB, Bulani VD et al (2018a) In vitro antiglycating effect and in vivo neuroprotective activity of trigonelline in d-galactose induced cognitive impairment. Pharmacol Rep 70:372–377. https://doi.org/10.1016/j.pharep.2017.09.006
Chowdhury AA, Gawali NB, Munshi R, Juvekar AR (2018b) Trigonelline insulates against oxidative stress, proinflammatory cytokines and restores BDNF levels in lipopolysaccharide induced cognitive impairment in adult mice. Metab Brain Dis 33:681–691. https://doi.org/10.1007/s11011-017-0147-5
Chowdhury AA, Gawali NB, Shinde P et al (2018c) Imperatorin ameliorates lipopolysaccharide induced memory deficit by mitigating proinflammatory cytokines, oxidative stress and modulating brain-derived neurotropic factor. Cytokine 110:78–86. https://doi.org/10.1016/j.cyto.2018.04.018
Cisternas P, Taylor X, Lasagna-Reeves CA (2019) The amyloid-tau-neuroinflammation axis in the context of cerebral amyloid angiopathy. Int J Mol Sci 20:6319. https://doi.org/10.3390/ijms20246319
Consonni M, Dalla Bella E, Contarino VE et al (2020) Cortical thinning trajectories across disease stages and cognitive impairment in amyotrophic lateral sclerosis. Cortex. https://doi.org/10.1016/j.cortex.2020.07.007
Craft JM, Watterson DM, Van Eldik LJ (2006) Human amyloid beta-induced neuroinflammation is an early event in neurodegeneration. Glia 53:484–490. https://doi.org/10.1002/glia.20306
Dani M, Wood M, Mizoguchi R et al (2018) Microglial activation correlates in vivo with both tau and amyloid in Alzheimer’s disease. Brain 141:2740–2754. https://doi.org/10.1093/brain/awy188
Dhanasekaran DN, Reddy EP (2008) JNK signaling in apoptosis. Oncogene 27:6245–6251. https://doi.org/10.1038/onc.2008.301
Di Lorenzo F, Motta C, Bonnì S et al (2019) LTP-like cortical plasticity is associated with verbal memory impairment in Alzheimer’s disease patients. Brain Stimul 12:148–151. https://doi.org/10.1016/j.brs.2018.10.009
Distrutti E, Sediari L, Mencarelli A et al (2006) 5-amino-2-hydroxybenzoic acid 4-(5-thioxo-5H-[1,2]dithiol-3yl)-phenyl ester (ATB-429), a hydrogen sulfide-releasing derivative of mesalamine, exerts antinociceptive effects in a model of postinflammatory hypersensitivity. J Pharmacol Exp Ther 319:447–458. https://doi.org/10.1124/jpet.106.106435
Draper HH, Squires EJ, Mahmoodi H et al (1993) A comparative evaluation of thiobarbituric acid methods for the determination of malondialdehyde in biological materials. Free Radic Biol Med 15:353–363. https://doi.org/10.1016/0891-5849(93)90035-s
Du P, Li S-J, Ojcius DM et al (2018) A novel Fas-binding outer membrane protein and lipopolysaccharide of Leptospira interrogans induce macrophage apoptosis through the Fas/FasL-caspase-8/-3 pathway. Emerg Microbes Infect 7:135. https://doi.org/10.1038/s41426-018-0135-9
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77. https://doi.org/10.1016/0003-9861(59)90090-6
Ellman GL, Courtney KD, Andres VJ, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Eto K, Asada T, Arima K et al (2002) Brain hydrogen sulfide is severely decreased in Alzheimer’s disease. Biochem Biophys Res Commun 293:1485–1488. https://doi.org/10.1016/S0006-291X(02)00422-9
García-Ayllón M-S, Small DH, Avila J, Sáez-Valero J (2011) Revisiting the role of acetylcholinesterase in Alzheimer’s disease: cross-talk with p-tau and β-amyloid. Front Mol Neurosci 4:22. https://doi.org/10.3389/fnmol.2011.00022
Gawali NB, Bulani VD, Chowdhury AA et al (2016) Agmatine ameliorates lipopolysaccharide induced depressive-like behaviour in mice by targeting the underlying inflammatory and oxido-nitrosative mediators. Pharmacol Biochem Behav 149:1–8. https://doi.org/10.1016/j.pbb.2016.07.004
Ghods-Sharifi S, Haluk DM, Floresco SB (2008) Differential effects of inactivation of the orbitofrontal cortex on strategy set-shifting and reversal learning. Neurobiol Learn Mem 89:567–573. https://doi.org/10.1016/j.nlm.2007.10.007
Giuliani D, Ottani A, Zaffe D et al (2013) Hydrogen sulfide slows down progression of experimental Alzheimer’s disease by targeting multiple pathophysiological mechanisms. Neurobiol Learn Mem 104:82–91. https://doi.org/10.1016/j.nlm.2013.05.006
Gong Q-H, Wang Q, Pan L-L et al (2010) Hydrogen sulfide attenuates lipopolysaccharide-induced cognitive impairment: a pro-inflammatory pathway in rats. Pharmacol Biochem Behav 96:52–58. https://doi.org/10.1016/j.pbb.2010.04.006
González-Scarano F, Baltuch G (1999) Microglia as mediators of inflammatory and degenerative diseases. Annu Rev Neurosci 22:219–240. https://doi.org/10.1146/annurev.neuro.22.1.219
Guan R, Wang J, Li D et al (2020) Hydrogen sulfide inhibits cigarette smoke-induced inflammation and injury in alveolar epithelial cells by suppressing PHD2/HIF-1α/MAPK signaling pathway. Int Immunopharmacol 81:105979. https://doi.org/10.1016/j.intimp.2019.105979
Haider S, Saleem S, Perveen T et al (2014) Age-related learning and memory deficits in rats: role of altered brain neurotransmitters, acetylcholinesterase activity and changes in antioxidant defense system. Age (dordr) 36:9653. https://doi.org/10.1007/s11357-014-9653-0
Hakimi Z, Salmani H, Marefati N et al (2020) Protective effects of carvacrol on brain tissue inflammation and oxidative stress as well as learning and memory in lipopolysaccharide-challenged rats. Neurotox Res 37:965–976. https://doi.org/10.1007/s12640-019-00144-5
Hansen DV, Hanson JE, Sheng M (2018) Microglia in Alzheimer’s disease. J Cell Biol 217:459–472. https://doi.org/10.1083/jcb.201709069
He M, Xiang Z, Xu L et al (2019) Lipopolysaccharide induces human olfactory ensheathing glial apoptosis by promoting mitochondrial dysfunction and activating the JNK-Bnip3-Bax pathway. Cell Stress Chaperones 24:91–104. https://doi.org/10.1007/s12192-018-0945-7
Hopperton KE, Trépanier M-O, Giuliano V, Bazinet RP (2016) Brain omega-3 polyunsaturated fatty acids modulate microglia cell number and morphology in response to intracerebroventricular amyloid-β 1–40 in mice. J Neuroinflammation 13:257. https://doi.org/10.1186/s12974-016-0721-5
Hu L-F, Wong PT-H, Moore PK, Bian J-S (2007) Hydrogen sulfide attenuates lipopolysaccharide-induced inflammation by inhibition of p38 mitogen-activated protein kinase in microglia. J Neurochem 100:1121–1128. https://doi.org/10.1111/j.1471-4159.2006.04283.x
Huang W, Li Z, Zhao L, Zhao W (2017) Simvastatin ameliorate memory deficits and inflammation in clinical and mouse model of Alzheimer’s disease via modulating the expression of miR-106b. Biomed Pharmacother 92:46–57. https://doi.org/10.1016/j.biopha.2017.05.060
Jang YJ, Kim J, Shim J et al (2013) Decaffeinated coffee prevents scopolamine-induced memory impairment in rats. Behav Brain Res 245:113–119. https://doi.org/10.1016/j.bbr.2013.02.003
Ju IG, Choi JG, Kim N et al (2018) Peucedani Japonici Radix ameliorates lipopolysaccharide-induced neuroinflammation by regulating microglial responses. Neurosci Lett 686:161–167. https://doi.org/10.1016/j.neulet.2018.09.010
Julayanont P, McFarland NR, Heilman KM (2020) Mild cognitive impairment and dementia in motor manifest Huntington’s disease: classification and prevalence. J Neurol Sci 408:116523. https://doi.org/10.1016/j.jns.2019.116523
Kumar M, Arora P, Sandhir R (2019) Hydrogen sulfide suppresses homocysteine-induced glial activation and inflammatory response. Nitric Oxide 90:15–28. https://doi.org/10.1016/j.niox.2019.05.008
Kumar M, Arora P, Sandhir R (2020) Hydrogen sulfide reverses LPS-induced behavioral deficits by suppressing microglial activation and promoting M2 polarization. J Neuroimmune Pharmacol. https://doi.org/10.1007/s11481-020-09920-z
Kimura Y, Kimura H (2004) Hydrogen sulfide protects neurons from oxidative stress. FASEB J off Publ Fed Am Soc Exp Biol 18:1165–1167. https://doi.org/10.1096/fj.04-1815fje
Le XT, Pham HTN, Do PT et al (2013) Bacopa monnieri ameliorates memory deficits in olfactory bulbectomized mice: possible involvement of glutamatergic and cholinergic systems. Neurochem Res 38:2201–2215. https://doi.org/10.1007/s11064-013-1129-6
Lee JW, Lee YK, Yuk DY et al (2008) Neuro-inflammation induced by lipopolysaccharide causes cognitive impairment through enhancement of beta-amyloid generation. J Neuroinflammation 5:37. https://doi.org/10.1186/1742-2094-5-37
Lee M, Schwab C, Yu S et al (2009) Astrocytes produce the antiinflammatory and neuroprotective agent hydrogen sulfide. Neurobiol Aging 30:1523–1534. https://doi.org/10.1016/j.neurobiolaging.2009.06.001
Lee M, Tazzari V, Giustarini D et al (2010) Effects of hydrogen sulfide-releasing L-DOPA derivatives on glial activation: potential for treating Parkinson disease. J Biol Chem 285:17318–17328. https://doi.org/10.1074/jbc.M110.115261
Li G-Z, Tao H-L, Zhou C et al (2018a) Midazolam prevents motor neuronal death from oxidative stress attack mediated by JNK-ERK pathway. Hum Cell 31:64–71. https://doi.org/10.1007/s13577-017-0184-8
Li H-W, Xiao F-Y (2020) Effect of hydrogen sulfide on cardiomyocyte apoptosis in rats with myocardial ischemia-reperfusion injury via the JNK signaling pathway. Eur Rev Med Pharmacol Sci 24:2054–2061. https://doi.org/10.26355/eurrev_202002_20383
Li T, Liu H, Xue H, Zhang J, Han X, Yan S, Bo S, Liu S, Yuan L, Deng L, Li G, Wang Z (2017) Neuroprotective Effects of Hydrogen Sulfide Against Early Brain Injury and Secondary Cognitive Deficits Following Subarachnoid Hemorrhage. Brain Pathol 27(1):51-63
Li X, Lin Y, Lin Y et al (2018b) Hydrogen sulfide protects against acetaminophen-induced acute liver injury by inhibiting apoptosis via the JNK/MAPK signaling pathway. J Cell Biochem 120:4385–4397. https://doi.org/10.1002/jcb.27724
Li Y, Zhang L, Tang J et al (2019) Role of toll-like receptor 4 in the regulation of the cell death pathway and neuroinflammation. Brain Res Bull 148:79–90. https://doi.org/10.1016/j.brainresbull.2019.03.015
Liou G-Y, Storz P (2010) Reactive oxygen species in cancer. Free Radic Res 44:479–496. https://doi.org/10.3109/10715761003667554
Liu H, Bai X-B, Shi S, Cao Y-X (2009) Hydrogen sulfide protects from intestinal ischaemia-reperfusion injury in rats. J Pharm Pharmacol 61:207–212. https://doi.org/10.1211/jpp/61.02.0010
Liu L, Wang J, Wang H (2020) Hydrogen sulfide alleviates oxidative stress injury and reduces apoptosis induced by MPP+ in Parkinson’s disease cell model. Mol Cell Biochem 472:231–240. https://doi.org/10.1007/s11010-020-03801-y
Ma MX, Chen YM, He J et al (2007) Effects of morphine and its withdrawal on Y-maze spatial recognition memory in mice. Neuroscience 147:1059–1065. https://doi.org/10.1016/j.neuroscience.2007.05.020
Marefati N, Beheshti F, Memarpour S et al (2020) The effects of acetyl-11-keto-β-boswellic acid on brain cytokines and memory impairment induced by lipopolysaccharide in rats. Cytokine 131:155107. https://doi.org/10.1016/j.cyto.2020.155107
Merrill JE, Koyanagi Y, Zack J et al (1992) Induction of interleukin-1 and tumor necrosis factor alpha in brain cultures by human immunodeficiency virus type 1. J Virol 66:2217–2225
Navarro V, Sanchez-Mejias E, Jimenez S et al (2018) Microglia in Alzheimer’s disease: activated, dysfunctional or degenerative. Front Aging Neurosci 10:140. https://doi.org/10.3389/fnagi.2018.00140
Ohno N, Ikenaka K (2019) Axonal and neuronal degeneration in myelin diseases. Neurosci Res 139:48–57. https://doi.org/10.1016/j.neures.2018.08.013
Pearson AG, Byrne UTE, MacGibbon GA et al (2006) Activated c-Jun is present in neurofibrillary tangles in Alzheimer’s disease brains. Neurosci Lett 398:246–250. https://doi.org/10.1016/j.neulet.2006.01.031
Rinaldi L, Gobbi G, Pambianco M et al (2006) Hydrogen sulfide prevents apoptosis of human PMN via inhibition of p38 and caspase 3. Lab Invest 86:391–397. https://doi.org/10.1038/labinvest.3700391
Salmani H, Hosseini M, Beheshti F et al (2018) Angiotensin receptor blocker, losartan ameliorates neuroinflammation and behavioral consequences of lipopolysaccharide injection. Life Sci 203:161–170. https://doi.org/10.1016/j.lfs.2018.04.033
Savage MJ, Lin Y-G, Ciallella JR et al (2002) Activation of c-Jun N-terminal kinase and p38 in an Alzheimer’s disease model is associated with amyloid deposition. J Neurosci 22:3376–3385. https://doi.org/10.1523/JNEUROSCI.22-09-03376.2002
Selkoe DJ, Hardy J (2016) The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol Med 8:595–608. https://doi.org/10.15252/emmm.201606210
Sestito S, Nesi G, Pi R, Macchia M, Rapposelli S (2017) Hydrogen sulfide: a worthwhile tool in the design of new multitarget drugs. Front Chem 27(5):72. https://doi.org/10.3389/fchem.2017.00072
Sestito S, Pruccoli L, Runfola M et al (2019) Design and synthesis of H2S-donor hybrids: a new treatment for Alzheimer’s disease? Eur J Med Chem 184:111745. https://doi.org/10.1016/j.ejmech.2019.111745
Suryavanshi PS, Ugale RR, Yilmazer-Hanke D et al (2014) GluN2C/GluN2D subunit-selective NMDA receptor potentiator CIQ reverses MK-801-induced impairment in prepulse inhibition and working memory in Y-maze test in mice. Br J Pharmacol 171:799–809. https://doi.org/10.1111/bph.12518
Talesa VN (2001) Acetylcholinesterase in Alzheimer’s disease. Mech Ageing Dev 122:1961–1969. https://doi.org/10.1016/s0047-6374(01)00309-8
Tyagi E, Agrawal R, Nath C, Shukla R (2008) Influence of LPS-induced neuroinflammation on acetylcholinesterase activity in rat brain. J Neuroimmunol 205:51–56. https://doi.org/10.1016/j.jneuroim.2008.08.015
Varatharaj A, Galea I (2017) The blood-brain barrier in systemic inflammation. Brain Behav Immun 60:1–12. https://doi.org/10.1016/j.bbi.2016.03.010
Vasconcellos LFR, Pereira JS, Charchat-Fichman H et al (2019) Mild cognitive impairment in Parkinson’s disease: characterization and impact on quality of life according to subtype. Geriatr Gerontol Int 19:497–502. https://doi.org/10.1111/ggi.13649
Wang CY, Zou W, Liang XY et al (2017) Hydrogen sulfide prevents homocysteine-induced endoplasmic reticulum stress in PC12 cells by upregulating SIRT-1. Mol Med Rep 16:3587–3593. https://doi.org/10.3892/mmr.2017.7004
Wu W, Hou C-L, Mu X-P et al (2017) H(2)S donor NaHS changes the production of endogenous H(2)S and NO in D-galactose-induced accelerated ageing. Oxid Med Cell Longev 2017:5707830. https://doi.org/10.1155/2017/5707830
Yarza R, Vela S, Solas M, Ramirez MJ (2015) c-Jun N-terminal kinase (JNK) signaling as a therapeutic target for Alzheimer’s disease. Front Pharmacol 6:321. https://doi.org/10.3389/fphar.2015.00321
Yoshida H, Hastie CJ, McLauchlan H et al (2004) Phosphorylation of microtubule-associated protein tau by isoforms of c-Jun N-terminal kinase (JNK). J Neurochem 90:352–358. https://doi.org/10.1111/j.1471-4159.2004.02479.x
Zhao FL, Fang F, Qiao PF, Yan N, Gao D, Yan Y (2016) AP39, a mitochondria-targeted hydrogen sulfide donor, supports cellular bioenergetics and protects against Alzheimer’s disease by preserving mitochondrial function in APP/PS1 mice and neurons. Oxid Med Cell Longev 2016:8360738. https://doi.org/10.1155/2016/8360738
Zheng Y-F, Dai D-Z, Dai Y (2010) NaHS ameliorates diabetic vascular injury by correcting depressed connexin 43 and 40 in the vasculature in streptozotocin-injected rats. J Pharm Pharmacol 62:615–621. https://doi.org/10.1211/jpp.62.05.0009
Acknowledgements
The authors would like to acknowledge the help of Dr. S. J. Pawar (M.V.Sc. Veterinary pathology) for histopathological examinations of brain samples.
Funding
The research was supported by the All India Council for Technical Education (AICTE), New Delhi, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kshirsagar, V., Thingore, C., Gursahani, M. et al. Hydrogen Sulfide Ameliorates Lipopolysaccharide-Induced Memory Impairment in Mice by Reducing Apoptosis, Oxidative, and Inflammatory Effects. Neurotox Res 39, 1310–1322 (2021). https://doi.org/10.1007/s12640-021-00374-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-021-00374-6