Abstract
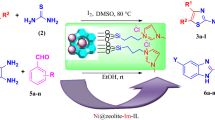
In this project, nano-MnO2@zeolite-Y (NMOZ) was successfully prepared via hydrothermal technique and its structure was confirmed using FT-IR, XRD, FE-SEM, EDX and BET analyses. MnO2 nanoparticles stabilized on zeolite-NaY were used as nanocatalyst for efficient solventless preparation of 3,4-disubstituted isoxazole-5(4H)-one scaffolds via one-pot three-component reaction of ethyl acetoacetate, hydroxylamine hydrochloride, and aromatic aldehydes at 100 °C. One of the advantages of this new method is the non-use of a metal oxidant such as KMnO4 (greener method) and the stabilization of MnO2 to the zeolite-Y surface via ion bonding. This causes the MnO2 not be leached from the zeolite during the processes of nanocatalyst recycling in organic reactions. This simple approach also has promising features as like as safe, inexpensive and recyclable nanocatalyst, solvent-free reaction, short reaction times, high to excellent yields, and easy isolation of the products.
Similar content being viewed by others
References
Moshoeshoe M, Nadiye-Tabbiruka M. S. Obuseng V (2017) Am J Mater Sci 7:196–221
Tontisirin S (2015). J Porous Mater 22:437–445
Kalhor M, Banibairami S, Mirshokraie SA (2018). Green Chem Lett Rev 11:334–344
Kalhor M, Khodaparast N (2015). Res Chem Intermed 41:3235–3242
Liang J, Liang Z, Zou R, Zhao Y (2017) Adv mater 29:1701139
Perot G, Guisnet M (1990). J Mol Catal 61:173–196
Lima CGS, Moreira NM, Paixão MW, Corrêa AG (2019). Curr Opin Green Sustain Chem 15:7–12
Safari J, Zarnegar Z, Borjian Borujeni M (2013). Chem Pap 67:688–695
Zhu Y, Shen M, Xia Y, Lu M (2015). Catal Commun 64:37–43
Saputra E, Muhammad S, Sun H, Ang HM, Tade MO, Wang S (2013). Environ Sci Technol 47:5882–5887
Zhang QX, Peng D, Huang XJ (2013). Electrochem Commun 34:270–273
Suib SL (2008) Porous manganese oxide octahedral molecular sieves and octahedral layered materials. Acc Chem Res 41:479–487
Liu J, Meng L, Fei Z, Dyson PJ, Jing X, Liu X (2017) MnO2 nanosheets as an artificial enzyme to mimic oxidase for rapid and sensitive detection of glutathione. Biosens Bioelectron 90:69–74
Chabre Y, Pannetier J (1995). Prog Solid State Chem 23:1–130
Zhang XY, Han LQ, Sun S, Wang CY, Chen MM (2015). J Alloys Compd 653:539–545
Zhang R, Yu X, Nam K. W, Ling C, Arthur T. S, Song W, Knapp A. M, Ehrlich S. N, Yang X. Q, Matsui M (2012) Electrochem Commun 23:110–113
Julien C, Massot M (2002). Phys Chem Chem Phys 4:4226–4235
Zsadon Jr B, Papp J, Czárán E, Papp Jr J (1990). J Mol Catal 62:321–330
Bentrup U, Brückner A, Richter M, Fricke R (2001). Appl Catal B 32:229–241
Richter M, Berndt H, Eckelt R, Schneider M, Fricke R (1999). Catal Today 54:531–545
Endang PS, Rahadian AR, Ulva TIM, Alvin RW, Rendy MI, Nurul W (2019). Mater. Sci. Forum 964:199–208
Han J, Guo H, Wang XG, Pang ML, Meng JB (2007). Chin J Chem 25:129–131
Aret E, Meekes H, Vlieg E, Deroover G (2007). Dyes Pigments 72:339–344
Biju S, Reddy MLP, Freire RO (2007). Inorg Chem Commun 10:393–396
Zhang XH, Zhan YH, Chen D, Wang F, Wang LY (2012). Dyes Pigments 93:1408–1415
Clemens Lamberth J (2018). J Heterocycl Chem 55:2035–2045
Santos MM, Faria N, Iley J (2010) Reaction of naphthoquinones with substituted nitromethanes. Facile synthesis and antifungal activity of naphtho[2,3-d]isoxazole-4,9-diones. Bioorg Med Chem Lett 20:193–195
Karabasanagouda T, Adhikari AV, Girisha M (2009). Indian J Chem 48B:430–437
Kamal A, Bharathi EV, Reddy JS (2011) Synthesis and biological evaluation of 3,5-diaryl isoxazoline/isoxazole linked 2,3-dihydroquinazolinone hybrids as anticancer agents. Eur J Med Chem 46:691–703
Kan H, Adachi I, Kido R, Hirose K (1967). J Med Chem 10:411–418
Diana P, Carbone A, Barraja P, Kelter G, Fiebig HH, Cirrincione G (2010) Synthesis and antitumor activity of 2,5-bis(3'-indolyl)-furans and 3,5-bis(3'-indolyl)-isoxazoles, nortopsentin analogues. Bioorg Med Chem 18:4524–4529
Ishioka T, Tanatani A, Nagasawa K, Hashimoto Y (2003). bioorg med Chem Lett 13:2655–2658
Ishioka T, Kubo A, Koiso Y, Nagasawa K, Itai A, Hashimoto Y (2002). Bioorg Med Chem 10:1555–1566
Kafle B, Aher NG, Khadka D, Park H, Cho H (2011) Isoxazol-5(4H)one derivatives as PTP1B inhibitors showing an anti-obesity effect. Chem Asian J 6:2073–2079
Panathur N, Gokhale N, Dalimba U, Koushik PV, Yogeeswari P, Sriram D (2015) New indole-isoxazolone derivatives: Synthesis, characterisation and in vitro SIRT1 inhibition studies. Bioorg Med Chem Lett 25:2768–2772
Villemin D, Martin B, Garrigues B (1993). Synth Commun 23:2251–2257
Nakamura I, Okamoto M, Terada M (2010). Org Lett 12:2453–2245
Donleavy JJ, Gilbert EE (1937). J Am Chem Soc 59:1072–1076
Lowe DB, Magnuson S, Qi N (2004) In vitro SAR of (5-(2H)-isoxazolonyl) ureas, potent inhibitors of hormone-sensitive lipase. Bioorg Med Chem Lett 14:3155–3159
Safari J, Ahmadzadeh M, Zarnegar Z (2016). Catal Commun 86:91–95
Saikh F, Das J, Ghosh S (2013). Tetrahedron Lett 54:4679–4682
Fozooni S, Gholam Hosseinzadeh N, Hamidianc H, Akhgarb MR (2013). J Braz Chem Soc 24:1649–1655
Kiyani H, Ghorbani F (2013). Heteroletters 3:145–153
Amol Khandebharad U, Sarda Swapnil R, Gill Charansingh H, Agrawal Brijmohan R (2015). Res J Chem Sci 5:27–32
Irannegad-Gheshlaghchaei N, Zare A, Sajadikhah SS, Banaei A (2018). Res Chem Intermed 44:6253–6266
Ferouani G, Nacer A, Ameur N, Bachir R, Ziani-Cherif C (2017). J Chin Chem Soc 65:459–464
laroum R, debache A (2018) Syn Commun 48:1876–1882
Kiyani H, Ghorbani F (2016). Res Chem Intermed 42:6831–6844
Vekariya RH, Patel KD, Patel HD (2016). Res Chem Intermed 42:7559–7579
Liu Q, Zhang YN (2011). Bull Kor Chem Soc 32:3559–3560
Rikani A, Setamdideh D (2016). Orient J Chem 32:1433–1437
Patil MS, Mudaliar C, Chaturbhu GU (2017). Tetrahedron Lett 58:3256–3261
Kiyani H, Kanaani A, Ajloo D, Ghorbani F, Vakili M (2015). Res Chem Intermed 41:7739–7773
Khandebharad AU, Sarda SR, Gill CH, Agrawal BR (2015). Res J Chem Sci 5:27–32
Chavan AP, Pinjari AB, Mhaske PC (2015). J Heterocycl Chem 52:1911–1915
Cheng QF, Liu XY, Wang QF, Liu LS, Liu WJ, Lin Q, Yang XJ (2009). Chin J Org Chem 29:1267–1271
Liu Q, Hou X (2012). Phosphorus Sulfur Silicon Relat Elem 187:448–453
Kiyani H, Ghorbani F (2013). Org Chem 58:14948–14950
Ablajan K, Xiamuxi H (2011). Chin Chem Lett 22:151–154
Acknowledgments
The gratitude of authors goes to the research commute of Chemistry Department of Payame Noor University who provided financial and technical supports for this project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kalhor, M., Samiei, S. & Mirshokraie, S.A. MnO2@Zeolite-Y Nanoporous: Preparation and Application as a High Efficient Catalyst for Multi-Component Synthesis of 4-Arylidene-Isoxazolidinones. Silicon 13, 201–210 (2021). https://doi.org/10.1007/s12633-020-00413-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-020-00413-5